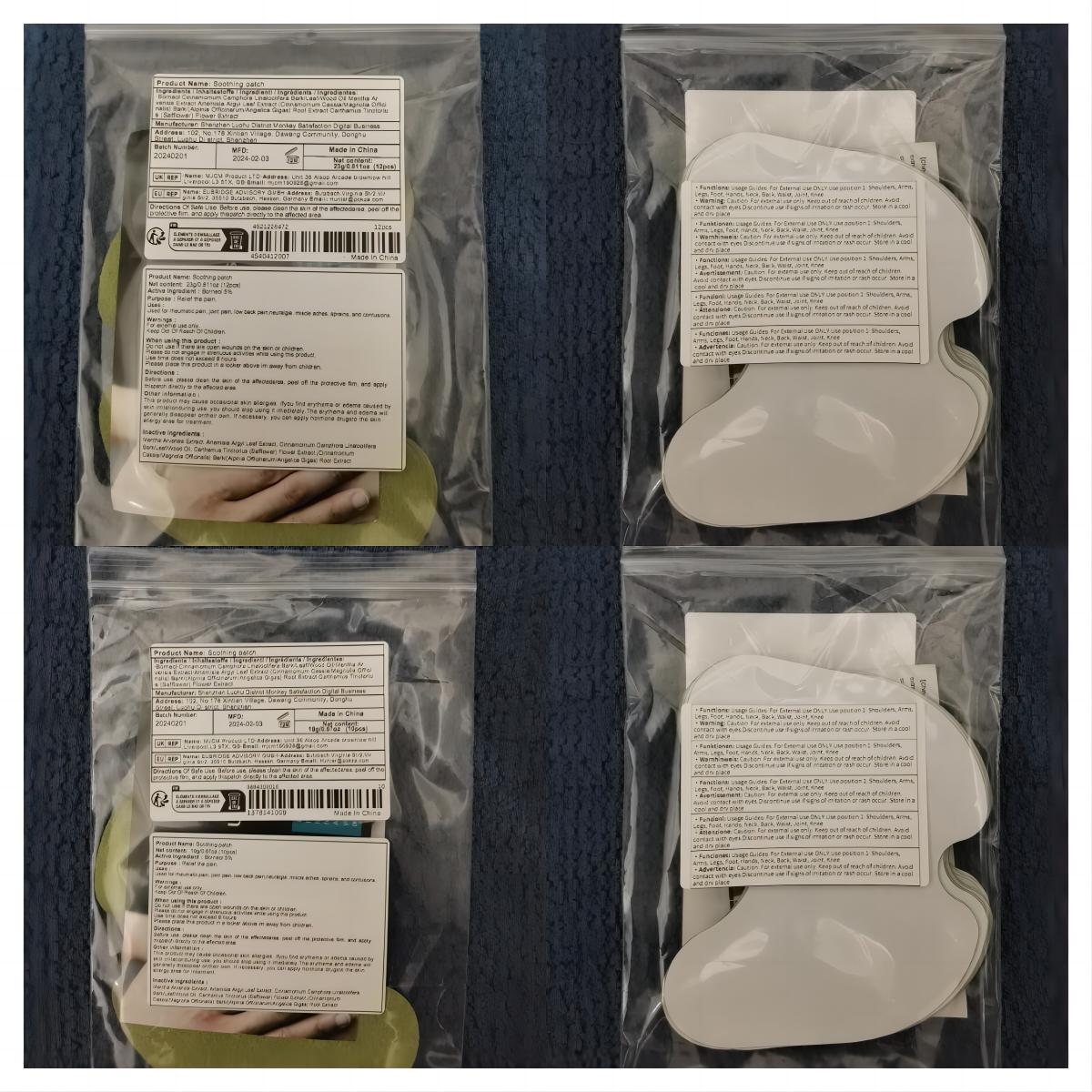
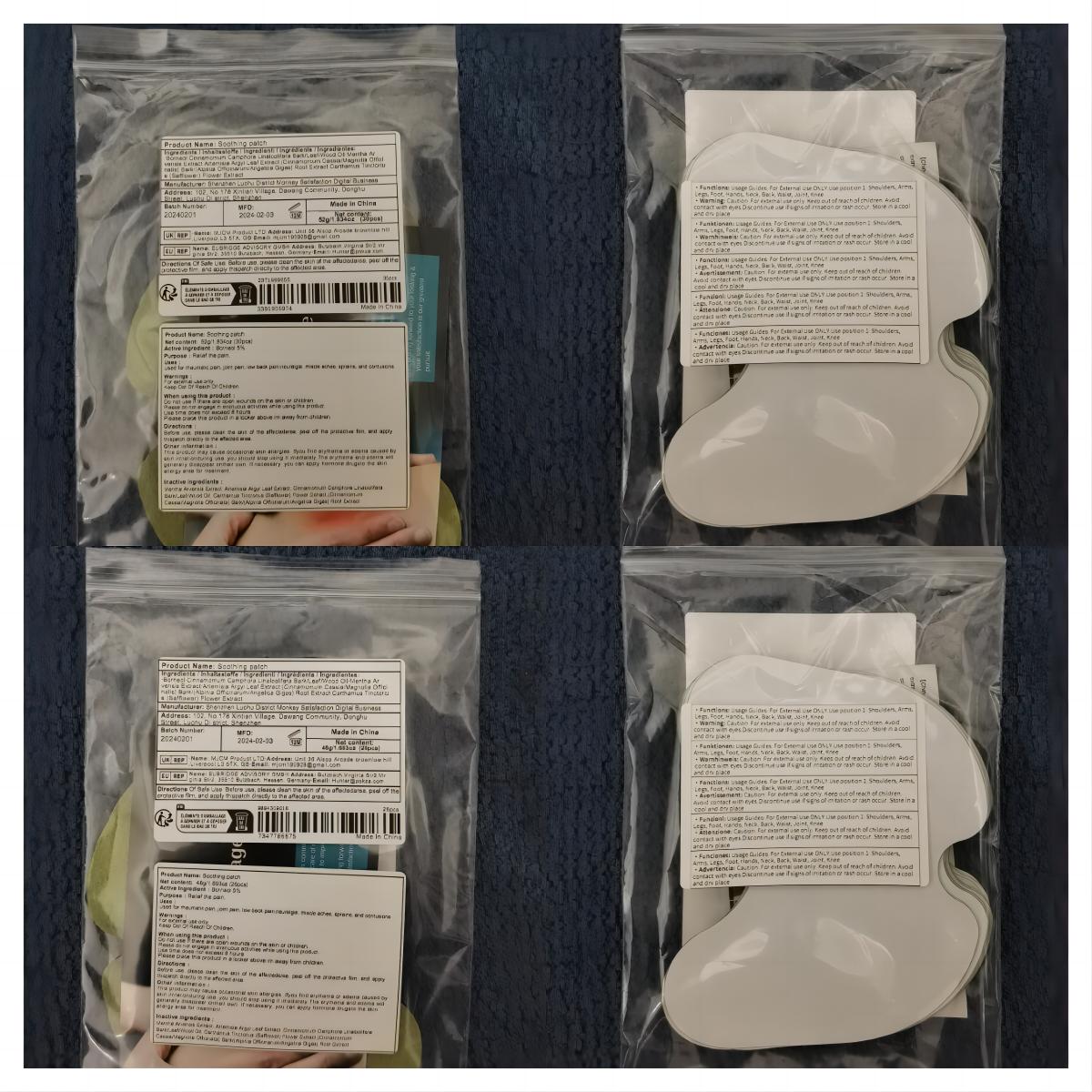
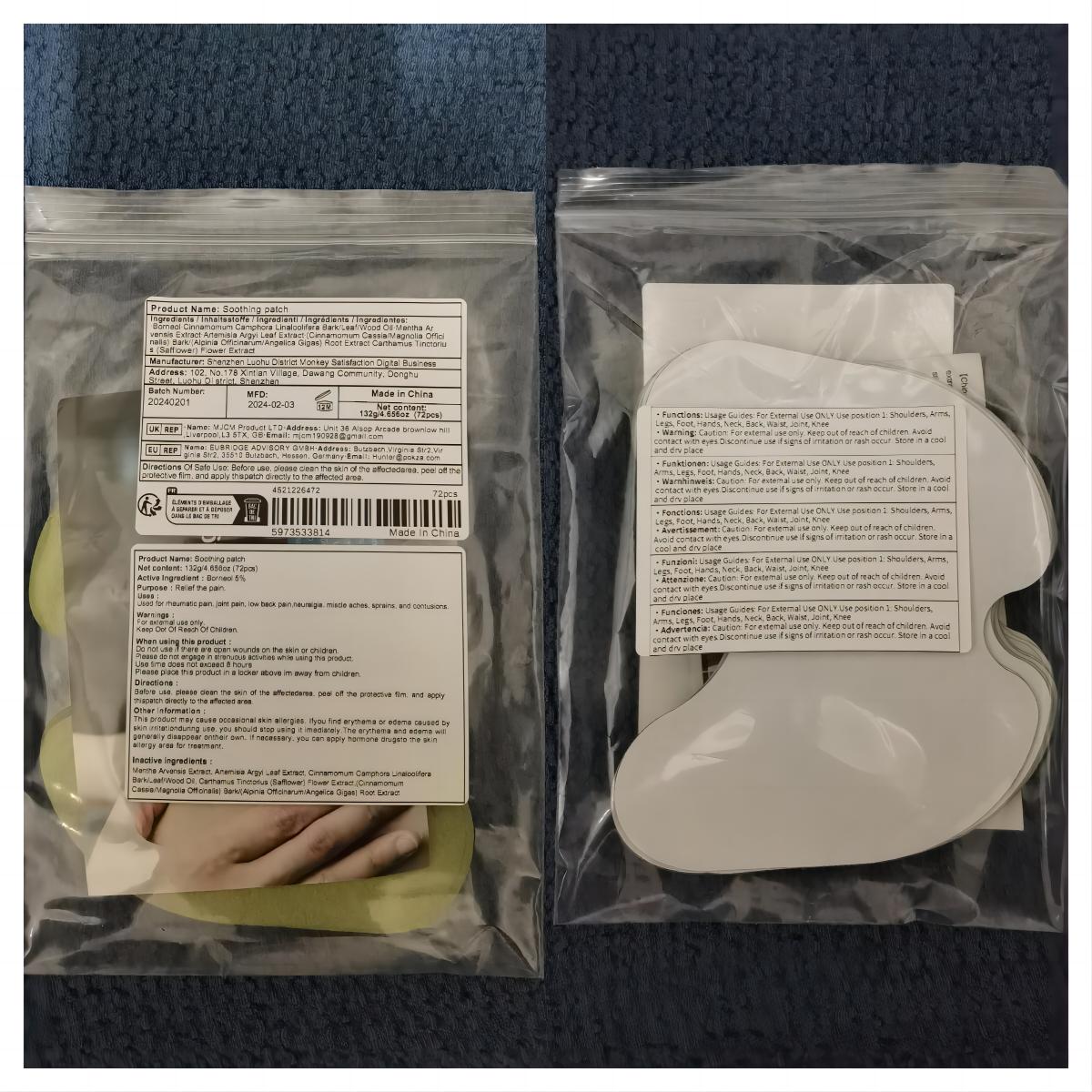
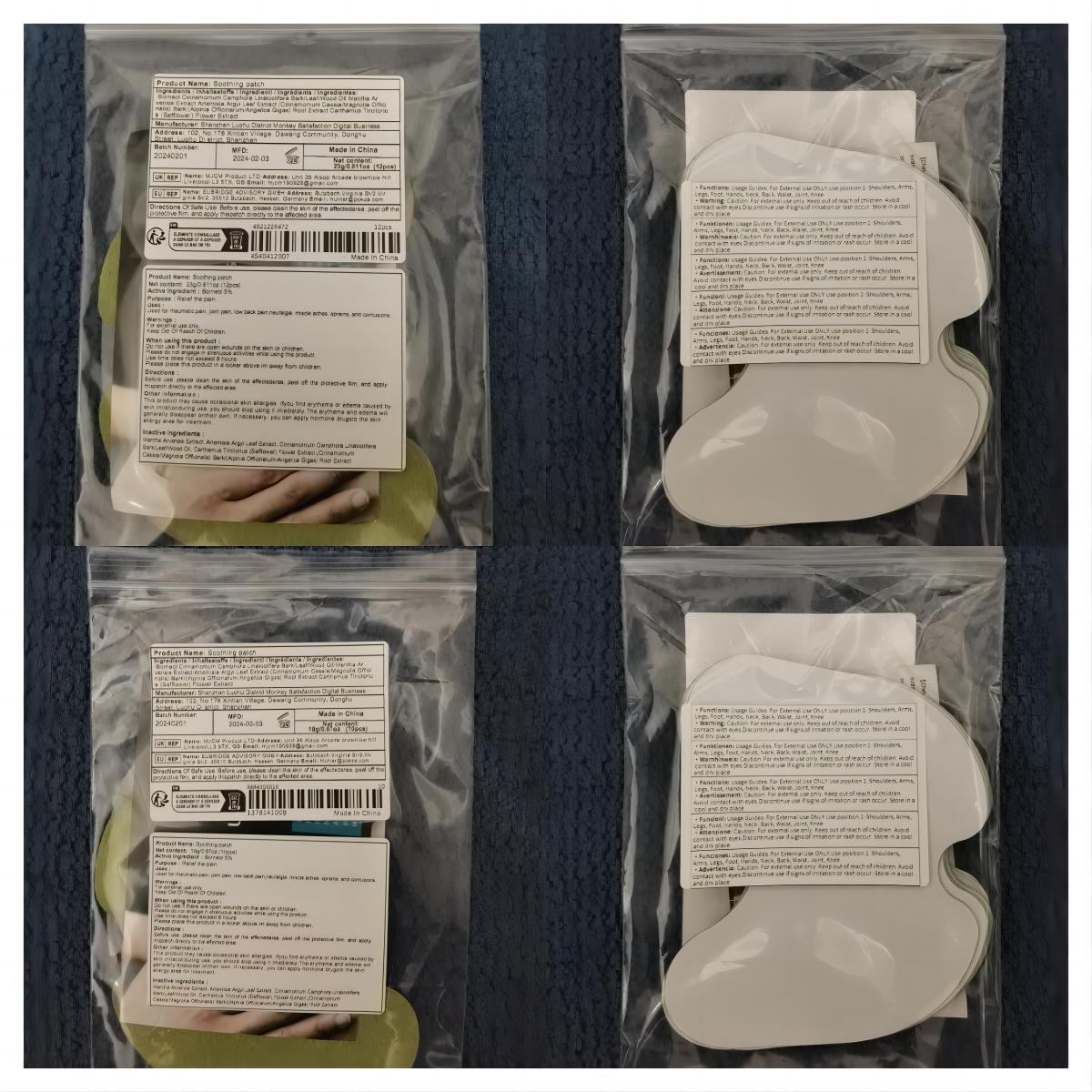
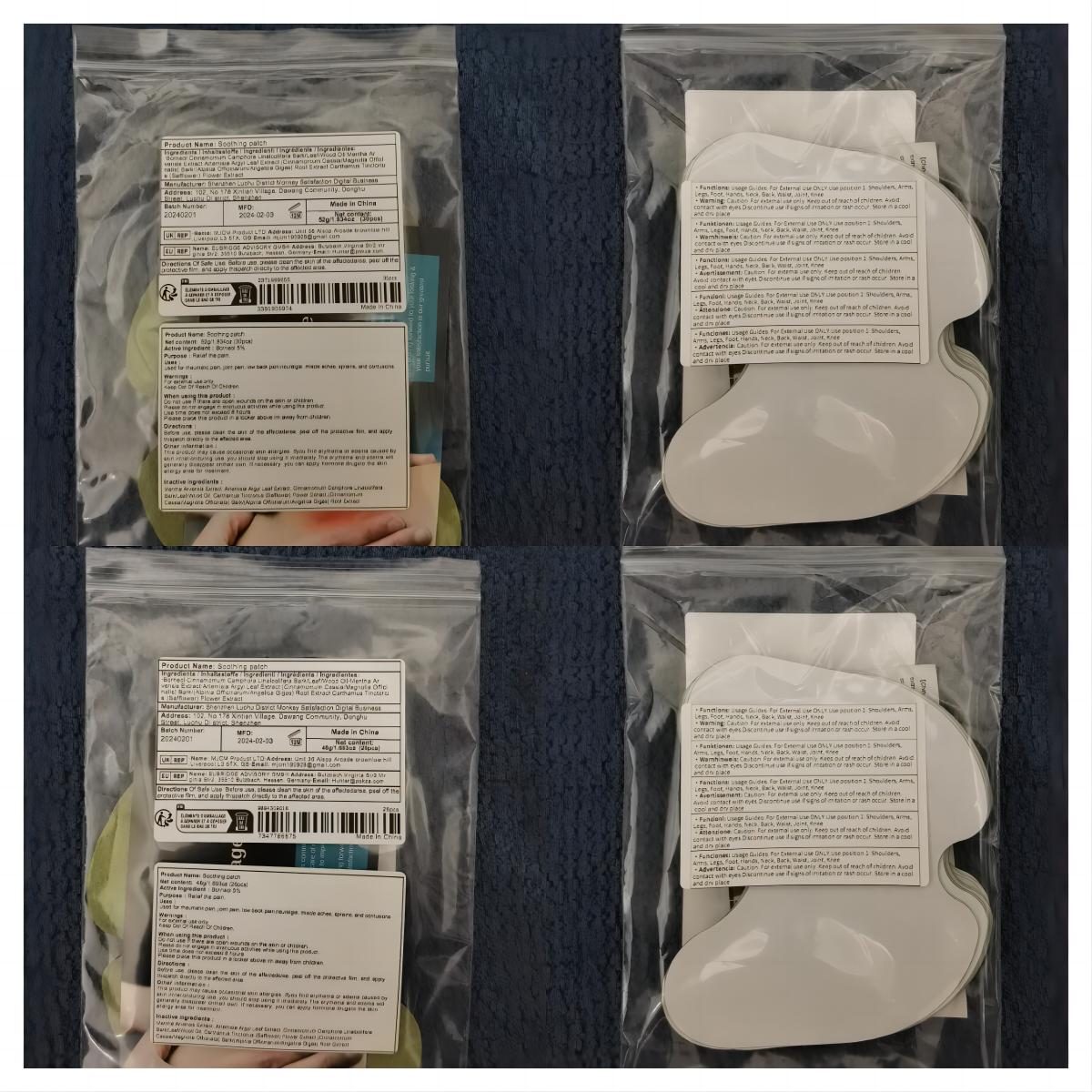
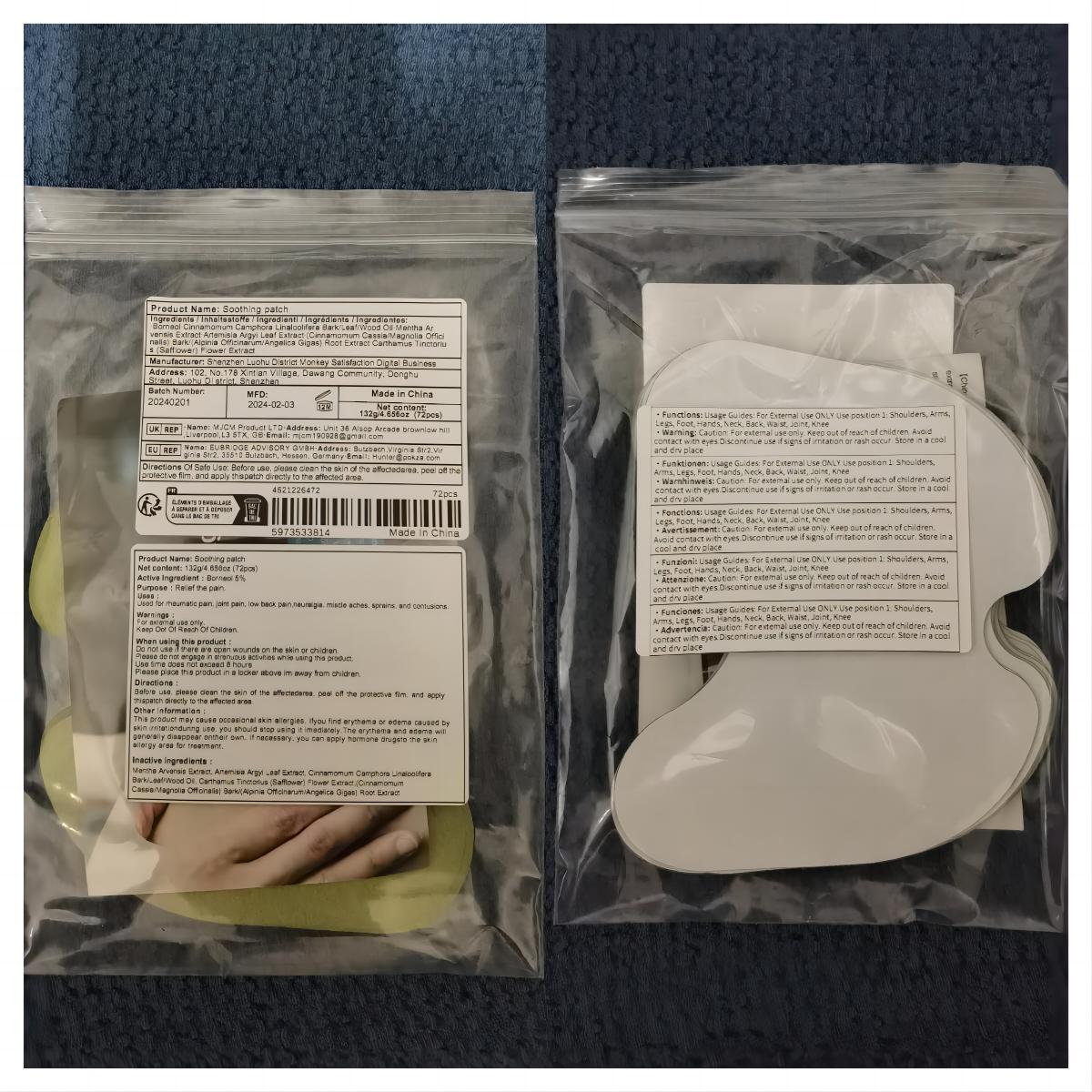
Label: SOOTHING PATCH patch
-
NDC Code(s):
83822-002-01,
83822-002-02,
83822-002-03,
83822-002-04, view more83822-002-05, 83822-002-06, 83822-002-07, 83822-002-08, 83822-002-09
- Packager: Fuzhou Ayimong Trading Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 25, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
- When Using
- Stop Use
- Keep Oot Of Reach Of Children
- Directions
- Other information
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SOOTHING PATCH
soothing patch patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83822-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BORNEOL (UNII: M89NIB437X) (BORNEOL - UNII:M89NIB437X) BORNEOL 5 g in 100 Inactive Ingredients Ingredient Name Strength ARTEMISIA ARGYI LEAF (UNII: 2JYC99Q0WZ) MENTHOL (UNII: L7T10EIP3A) CINNAMOMUM CAMPHORA WHOLE (UNII: 0B27814T7X) CINNAMOMUM CASSIA TWIG (UNII: 4UD6VQR2UP) SAFFLOWER (UNII: 4VBL71TY4Y) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83822-002-01 10 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 2 NDC:83822-002-02 12 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 3 NDC:83822-002-03 15 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 4 NDC:83822-002-04 24 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 5 NDC:83822-002-05 26 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 6 NDC:83822-002-06 30 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 7 NDC:83822-002-07 48 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 8 NDC:83822-002-08 60 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 9 NDC:83822-002-09 72 in 1 BAG; Type 0: Not a Combination Product 02/26/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 02/26/2024 Labeler - Fuzhou Ayimong Trading Co., Ltd. (416068159) Establishment Name Address ID/FEI Business Operations Fuzhou Ayimong Trading Co., Ltd. 416068159 manufacture(83822-002) , label(83822-002)