Label: CLOSYS SILVER HEALTHY GUMS- cetylpyridinium chloride liquid
- NDC Code(s): 58578-0407-1
- Packager: Rowpar Pharmaceuticals, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- PURPOSE
- Uses
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- Other Information
- INACTIVE INGREDIENT
- Questions or comments?
-
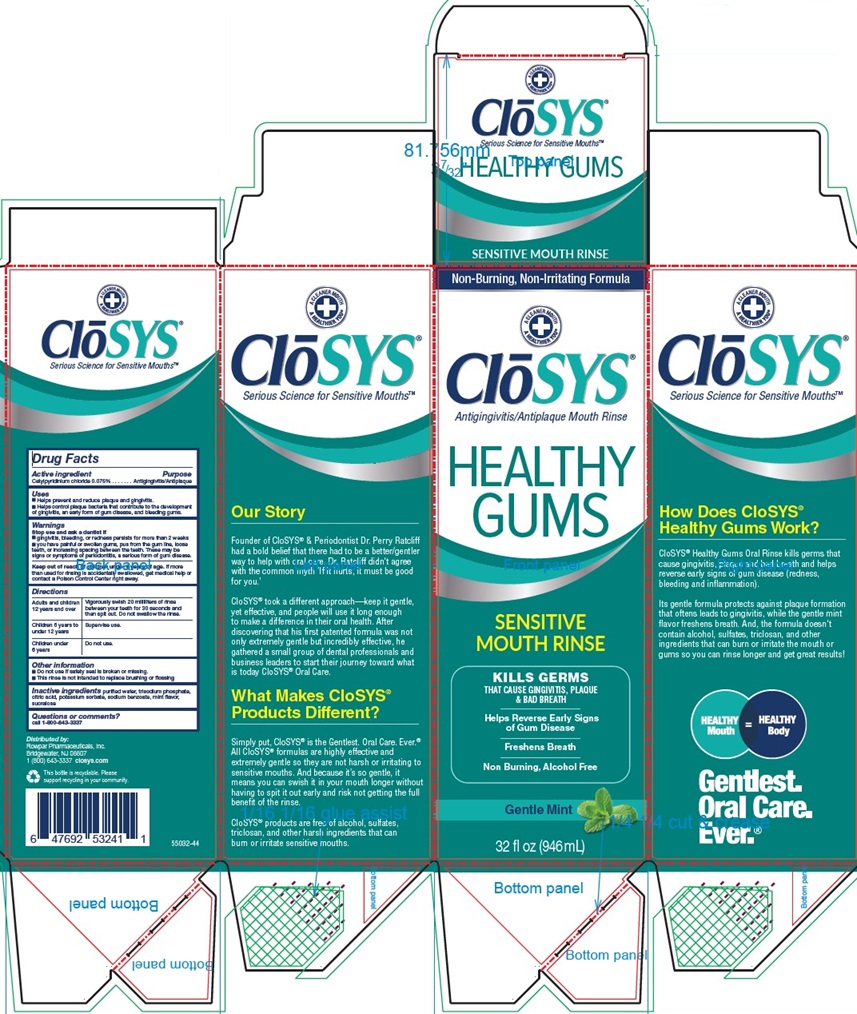
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Non-Burning, Non-Irritating Formula
A CLEANER MOUTH
A HEALTHIER YOU®
ClōSYS®
Antigingivitis/Antiplaque Mouth Rinse
HEALTHY
GUMS
SENSITIVE
MOUTH RINSE
KILLS GERMS
THAT CAUSE GINGIVITIS, PLAQUE
& BAD BREATH
Helps Reverse Early Signs
of Gum Disease
Freshens Breath
Non Burning, Alcohol Free
Gentle Mint
32 fl oz (946 mL)
Our Story
Founder of CloSYS® & Periodontist Dr. Perry Ratcliff
had a bold belief that there had to be a better/gentler
way to help with oral care. Dr. Ratcliff didn’t agree
with the common myth ‘If it hurts, it must be good
for you.’
CloSYS® took a different approach―keep it gentle,
yet effective, and people will use it long enough
to make a difference in their oral health. After
discovering that his first patented formula was not
only extremely gentle but incredibly effective, he
gathered a small group of dental professionals and
business leaders to start their journey toward what
is today Founder of CloSYS® Oral Care.
What Makes CloSYS®
Products Different?
Simply put, CloSYS® is the Gentlest, Oral Care. Ever.®
All CloSYS® formulas are highly effective and
extremely gentle so they are not harsh or irritating to
sensitive mouths. And because it’s so gentle, it
means you can swish it in your mouth longer without
having to spit it out early and risk not getting the full
benefit of the rinse.
CloSYS® products are free of alcohol, sulfates,
triclosan, and other harsh ingredients that can
burn or irritate sensitive mouths.
How Does CloSYS®
Healthy Gums Work?
CloSYS® Healthy Gums Oral Rinse kills germs that
cause gingivitis, plaque and bad breath and helps
reverse early signs of gum disease (redness,
bleeding and inflammation).
Its gentle formula protects against plaque formation
that oftens leads to gingivitis, while the gentle mint
flavor freshens breath. And, the formula doesn’t
contain alcohol, sulfates, triclosan, and other
ingredients that can burn or irritate the mouth or
gums so you can rinse longer and get great results!
HEALTHY Mouth= HEALTHY Body
Gentlest.
Oral Care.
Ever.®Distributed by:
Rowpar Pharmaceuticals, Inc.
Bridgewater, NJ 08807
1(800) 643-3337 closys.com
This bottle is recyclable. Please
support recycling in your community.55002-44
A CLEANER MOUTH
A HEALTHIER YOU®
ClōSYS
Antigingivitis/Antiplaque Mouth Rinse
HEALTHY
GUMS
SENSITIVE
MOUTH RINSE
KILLS GERMS
THAT CAUSE GINGIVITIS, PLAQUE
& BAD BREATH
Helps Reverse Early Signs
of Gum Disease
Freshens Breath
Non Burning, Alcohol Free
Gentle Mint
32 fl oz (946 mL)
52532-85
Distributed by: Rowpar Pharmaceuticals, Inc.
Bridgewater, NJ 08807
1(800) 643-3337 closys.com
This bottle is recyclable. Please
support recycling in your community.
52532-86

-
INGREDIENTS AND APPEARANCE
CLOSYS SILVER HEALTHY GUMS
cetylpyridinium chloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58578-0407 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Cetylpyridinium Chloride (UNII: D9OM4SK49P) (Cetylpyridinium - UNII:CUB7JI0JV3) Cetylpyridinium Chloride 0.75 g in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Potassium Sorbate (UNII: 1VPU26JZZ4) Sodium Phosphate, Tribasic, Dodecahydrate (UNII: B70850QPHR) Anhydrous Citric Acid (UNII: XF417D3PSL) Sodium Benzoate (UNII: OJ245FE5EU) Mint (UNII: FV98Z8GITP) Sucralose (UNII: 96K6UQ3ZD4) Benzoic Acid (UNII: 8SKN0B0MIM) Product Characteristics Color Score Shape Size Flavor MINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58578-0407-1 1 in 1 CARTON 03/15/2023 1 946 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M022 03/15/2023 Labeler - Rowpar Pharmaceuticals, Inc. (783704661)