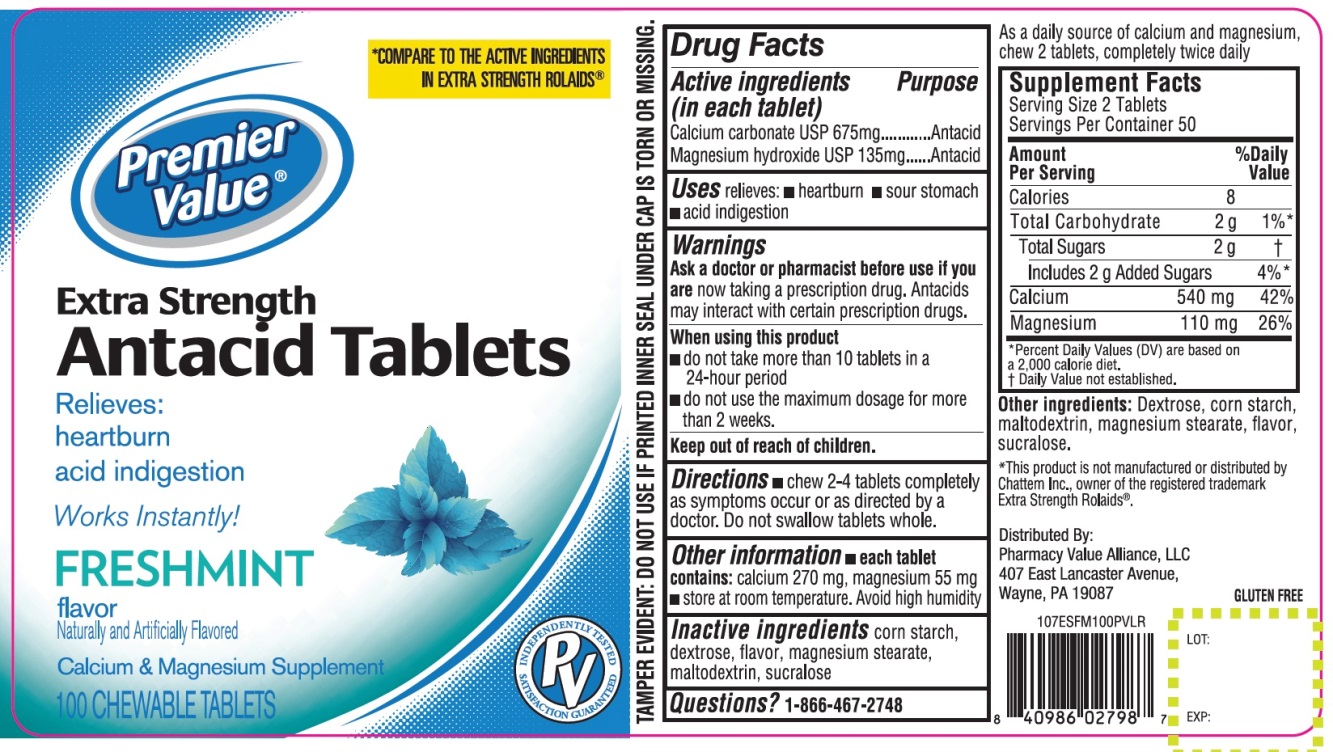
Label: PREMIER VALUE EXTRA STRENGTH FRESHMINT FLAVOR- calcium carbonate ,magnesium hydroxide tablet, chewable
- NDC Code(s): 68016-678-00
- Packager: CHAIN DRUG CONSORTIUM, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 3, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients (in each tablet)
- Purpose
- Uses
- Warnings
- Directions
-
Other information
- ▪
- each tablet contains: calcium 270 mg, magnesium 55 mg
- ▪
- store room temperature
- ▪
- Avoid high humidity.
DO NOT USE IF PRINTED INNER SEAL IS BROKEN OR MISSING
CALCIUM SUPPLEMENT
USES
As a daily source of calcium and magnesium, chew 2 tablets, completely twice daily
Supplement Facts
Serving Size: 2 Tablets
Servings Per Container: 50
Amount Per Serving
% Daily Value
Calories 8
Total Carbohydrate 2g
1%
Total Sugars 2g
†
Includes 2 g Added Sugar
4%
Calcium 540 mg
42%
Magnesium 110 mg
26%
*Percent Daily Values (DV) are based on a 2,000 calories diet.
†Daily Value not established.
- Inactive ingredients
-
PRINCIPAL DISPLAY PANEL
Premier Value®
NDC# 68016-678-00
COMPARE TO ACTIVE INGREDIENTS IN ROLAIDS® EXTRA STRENGTH*
Extra Strength
Antacid Tablets
Freshmint Flavor
Relieves:
- •
- Heartburn
- •
- Acid Indigestion
Antacid/Calcium Supplement
100 CHEWABLE TABLETS
GLUTEN FREE
TAMPER EVIDENT: DO NOT USE IF PRINTED INNER SEAL IS BROKEN OR MISSING
*This product is not manufactured or distributed by Chattem Inc., owner of the registered trademark Extra Strength Rolaids®
Distributed by:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue,
Wayne, PA 19087
-
INGREDIENTS AND APPEARANCE
PREMIER VALUE EXTRA STRENGTH FRESHMINT FLAVOR
calcium carbonate ,magnesium hydroxide tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-678 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CARBONATE 675 mg MAGNESIUM HYDROXIDE (UNII: NBZ3QY004S) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM HYDROXIDE 135 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) DEXTROSE, UNSPECIFIED FORM (UNII: IY9XDZ35W2) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color WHITE Score no score Shape ROUND Size 16mm Flavor MINT Imprint Code RP107 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-678-00 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/18/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 04/18/2019 Labeler - CHAIN DRUG CONSORTIUM, LLC (101668460)