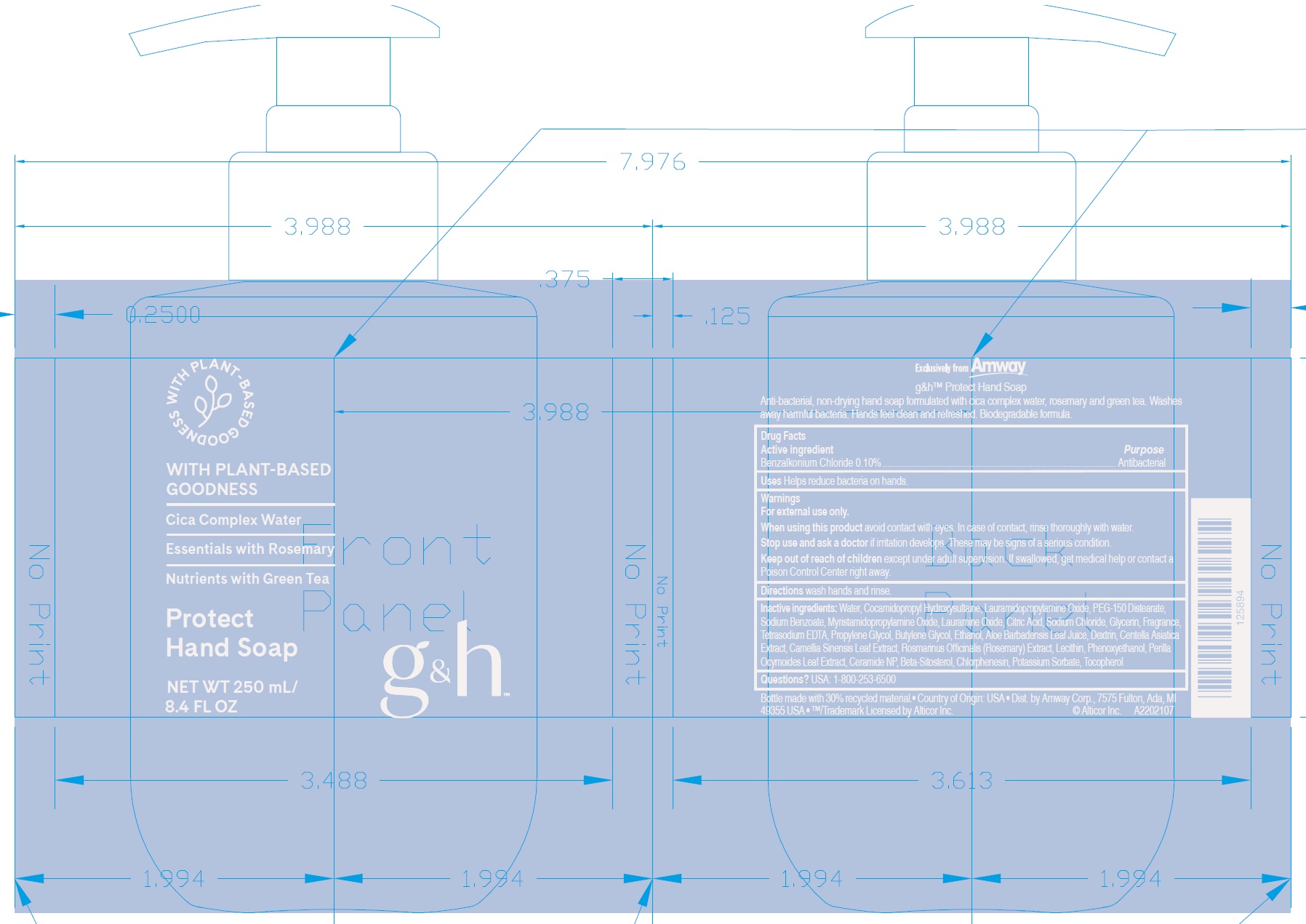
Label: G AND H PROTECT HAND- benzalkonium chloride soap
- NDC Code(s): 10056-026-00
- Packager: Access Business Group LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 23, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Uses
- Warnings
- Directions
-
Inactive ingredients:
Water, Cocamidopropyl Hydroxysultaine, Lauramidopropylamine Oxide, PEG-150 Distearate, Sodium Benzoate, Myristamidopropylamine Oxide, Lauramine Oxide, Citric Acid, Sodium Chloride, Glycerin, Fragrance, Tetrasodium EDTA, Propylene Glycol, Butylene Glycol, Ethanol, Aloe Barbadensis Leaf Juice, Dextrin, Centella Asiatica Extract, Camellia Sinensis Leaf Extract, Rosmarinus Officinalis (Rosemary) Extract, Lecithin, Phenoxyethanol, Perilla Ocymoides Leaf Extract, Ceramide NP, Beta-Sitosterol, Chlorphenesin, Potassium Sorbate, Tocopherol
- Questions?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
G AND H PROTECT HAND
benzalkonium chloride soapProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10056-026 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) COCAMIDOPROPYL HYDROXYSULTAINE (UNII: 62V75NI93W) LAURAMIDOPROPYLAMINE OXIDE (UNII: I6KX160QTV) PEG-150 DISTEARATE (UNII: 6F36Q0I0AC) SODIUM BENZOATE (UNII: OJ245FE5EU) MYRISTAMIDOPROPYLAMINE OXIDE (UNII: 3HSF539C9T) LAURAMINE OXIDE (UNII: 4F6FC4MI8W) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM CHLORIDE (UNII: 451W47IQ8X) GLYCERIN (UNII: PDC6A3C0OX) EDETATE SODIUM (UNII: MP1J8420LU) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ALCOHOL (UNII: 3K9958V90M) ALOE VERA LEAF (UNII: ZY81Z83H0X) CENTELLA ASIATICA TRITERPENOIDS (UNII: 4YS74Q4G4J) GREEN TEA LEAF (UNII: W2ZU1RY8B0) ROSEMARY (UNII: IJ67X351P9) PHENOXYETHANOL (UNII: HIE492ZZ3T) PERILLA FRUTESCENS LEAF (UNII: T4L5881Y68) CERAMIDE NP (UNII: 4370DF050B) .BETA.-SITOSTEROL (UNII: S347WMO6M4) CHLORPHENESIN (UNII: I670DAL4SZ) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) TOCOPHEROL (UNII: R0ZB2556P8) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10056-026-00 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/19/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 04/19/2023 Labeler - Access Business Group LLC (839830713)