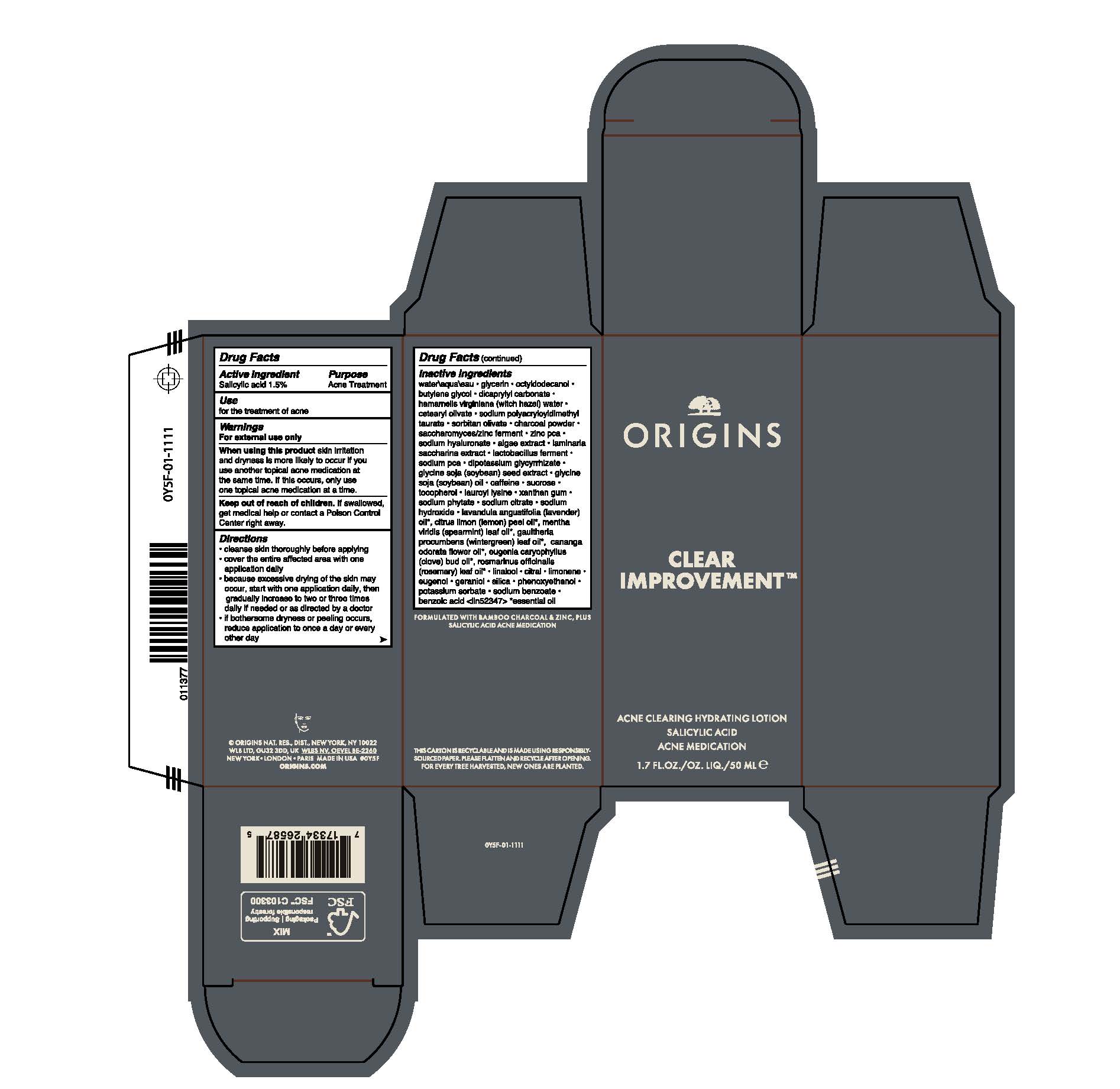
Label: CLEAR IMPROVEMENT ACNE CLEARING HYDRATING LTN- salicylic acid lotion
- NDC Code(s): 59427-117-01, 59427-117-02, 59427-117-03
- Packager: ORIGINS NATURAL RESOURCES INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 5, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
-
Directions
Cleanse skin throughly before applying
Cover the entire affected area with one application daily
Because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor.
If bothersome dryness or peeling occurs, reduce application to once a day or every other day
-
Inactive Ingredients
water\aqua\eau,glycerin,octyldodecanol,butylene glycol,dicaprylyl carbonate,hamamelis virginiana (witch hazel) water,cetearyl olivate,sodium polyacryloyldimethyl taurate,sorbitan olivate,charcoal powder,saccharomyces/zinc ferment,zinc pca,sodium hyaluronate,algae extract,laminaria saccharina extract,lactobacillus ferment,sodium pca,dipotassium glycyrrhizate,glycine soja (soybean) seed extract,glycine soja (soybean) oil,caffeine,sucrose,tocopherol,lauroyl lysine,xanthan gum,sodium phytate,sodium citrate,sodium hydroxide,lavandula angustifolia (lavender) oil*, citrus limon (lemon) peel oil*, mentha viridis (spearmint) leaf oil*, gaultheria procumbens (wintergreen) leaf oil*, cananga odorata flower oil*, eugenia caryophyllus (clove) bud oil*, rosmarinus officinalis (rosemary) leaf oil*,linalool,citral,limonene,eugenol,geraniol,silica,phenoxyethanol,potassium sorbate,sodium benzoate,benzoic acid <iln52347>
* essential oil - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CLEAR IMPROVEMENT ACNE CLEARING HYDRATING LTN
salicylic acid lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59427-117 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 15 mg in 1 mL Inactive Ingredients Ingredient Name Strength SACCHARINA LATISSIMA (UNII: 68CMP2MB55) GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) CETEARYL OLIVATE (UNII: 58B69Q84JO) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) LAUROYL LYSINE (UNII: 113171Q70B) CAFFEINE (UNII: 3G6A5W338E) TOCOPHEROL (UNII: R0ZB2556P8) METHYL SALICYLATE (UNII: LAV5U5022Y) ROSEMARY OIL (UNII: 8LGU7VM393) LEMON OIL (UNII: I9GRO824LL) SPEARMINT OIL (UNII: C3M81465G5) CANANGA OIL (UNII: 8YOY78GNNX) OCTYLDODECANOL (UNII: 461N1O614Y) HAMAMELIS VIRGINIANA TOP WATER (UNII: NT00Y05A2V) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SUCROSE (UNII: C151H8M554) XANTHAN GUM (UNII: TTV12P4NEE) PHENOXYETHANOL (UNII: HIE492ZZ3T) WATER (UNII: 059QF0KO0R) SOYBEAN OIL (UNII: 241ATL177A) LIMONENE, (+)- (UNII: GFD7C86Q1W) GERANIOL (UNII: L837108USY) PORPHYRIDIUM PURPUREUM (UNII: K2P8K2558N) SODIUM PYRROLIDONE CARBOXYLATE (UNII: 469OTG57A2) CLOVE OIL (UNII: 578389D6D0) CITRAL (UNII: T7EU0O9VPP) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) GLYCERIN (UNII: PDC6A3C0OX) SODIUM POLYACRYLOYLDIMETHYL TAURATE (UNII: NG5NG5733T) SORBITAN OLIVATE (UNII: MDL271E3GR) ACTIVATED CHARCOAL (UNII: 2P3VWU3H10) ZINC PIDOLATE (UNII: C32PQ86DH4) HYALURONATE SODIUM (UNII: YSE9PPT4TH) SOYBEAN (UNII: L7HT8F1ZOD) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM HYDROXIDE (UNII: 55X04QC32I) LAVENDER OIL (UNII: ZBP1YXW0H8) LINALOOL, (+/-)- (UNII: D81QY6I88E) EUGENOL (UNII: 3T8H1794QW) SODIUM BENZOATE (UNII: OJ245FE5EU) BENZOIC ACID (UNII: 8SKN0B0MIM) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59427-117-01 1 in 1 CARTON 02/05/2024 1 50 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:59427-117-02 15 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/05/2024 3 NDC:59427-117-03 5 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/05/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 02/05/2024 Labeler - ORIGINS NATURAL RESOURCES INC. (611716283) Registrant - Estee Lauder Companies Inc. (790802086) Establishment Name Address ID/FEI Business Operations The Estee Lauder Inc 802599436 manufacture(59427-117) , label(59427-117) , pack(59427-117)