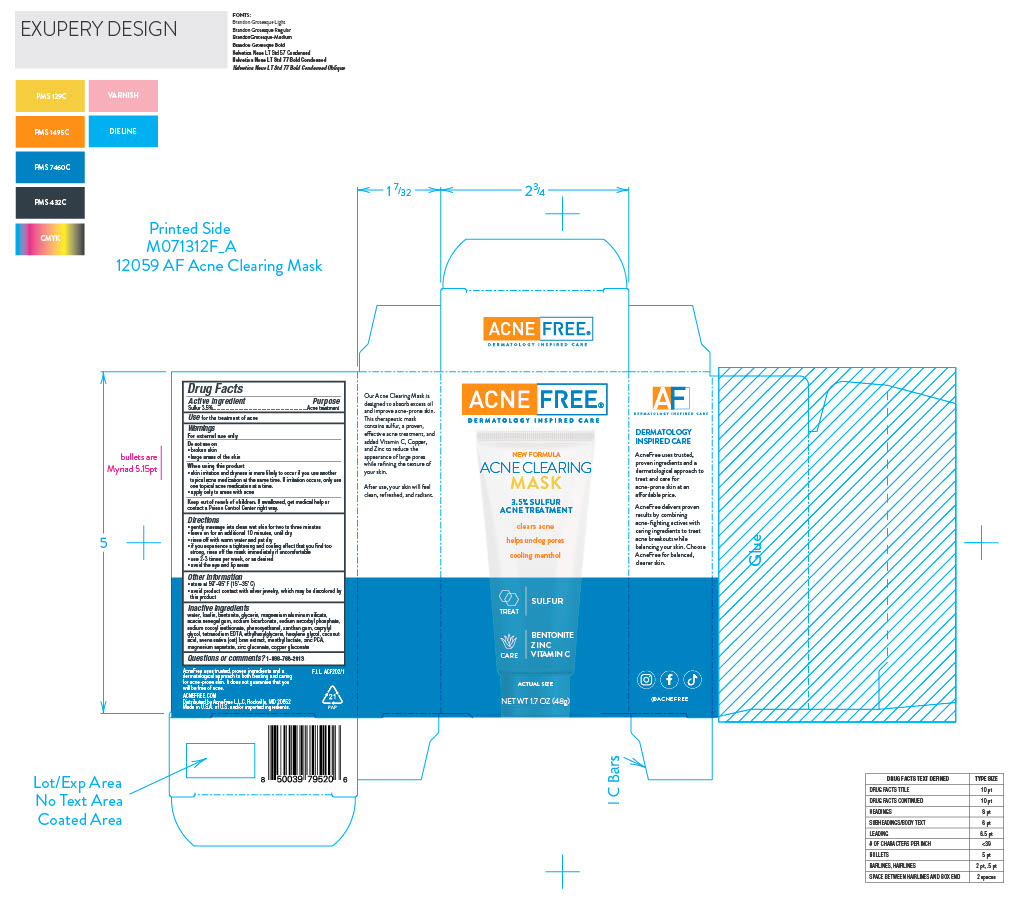
Label: SULFUR ACNE TREATMENT- sulfur cream
- NDC Code(s): 80861-116-01
- Packager: Acne Free
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
-
Directions
Directions • gently massage into clean wet skin for two to three minutes • leave on for an additional 10 minutes, until dry • rinse off with warm water and pat dry • if you experience a tightening and cooling effect that you find too strong, rinse off the mask immediately if uncomfortable • use 2-3 times per week, or as desired • avoid the eye and lip areas
- Other information
-
INACTIVE INGREDIENT
Inactive ingredients water, kaolin, bentonite, glycerin, magnesium aluminum silicate, acacia senegal gum, sodium bicarbonate, sodium ascorbyl phosphate, sodium cocoyl isethionate, phenoxyethanol, xanthan gum, caprylyl glycol, tetrasodium EDTA, ethylhexylglycerin, hexylene glycol, coconut acid, avena sativa (oat) bran extract, menthyl lactate, zinc PCA, magnesium aspartate, zinc gluconate, copper gluconate
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SULFUR ACNE TREATMENT
sulfur creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80861-116 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SULFUR (UNII: 70FD1KFU70) (SULFUR - UNII:70FD1KFU70) SULFUR 0.035 g in 1 g Inactive Ingredients Ingredient Name Strength BENTONITE (UNII: A3N5ZCN45C) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80861-116-01 1 g in 1 TUBE; Type 0: Not a Combination Product 02/05/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 02/02/2024 Labeler - Acne Free (122237140) Establishment Name Address ID/FEI Business Operations Paklab 177711082 manufacture(80861-116)