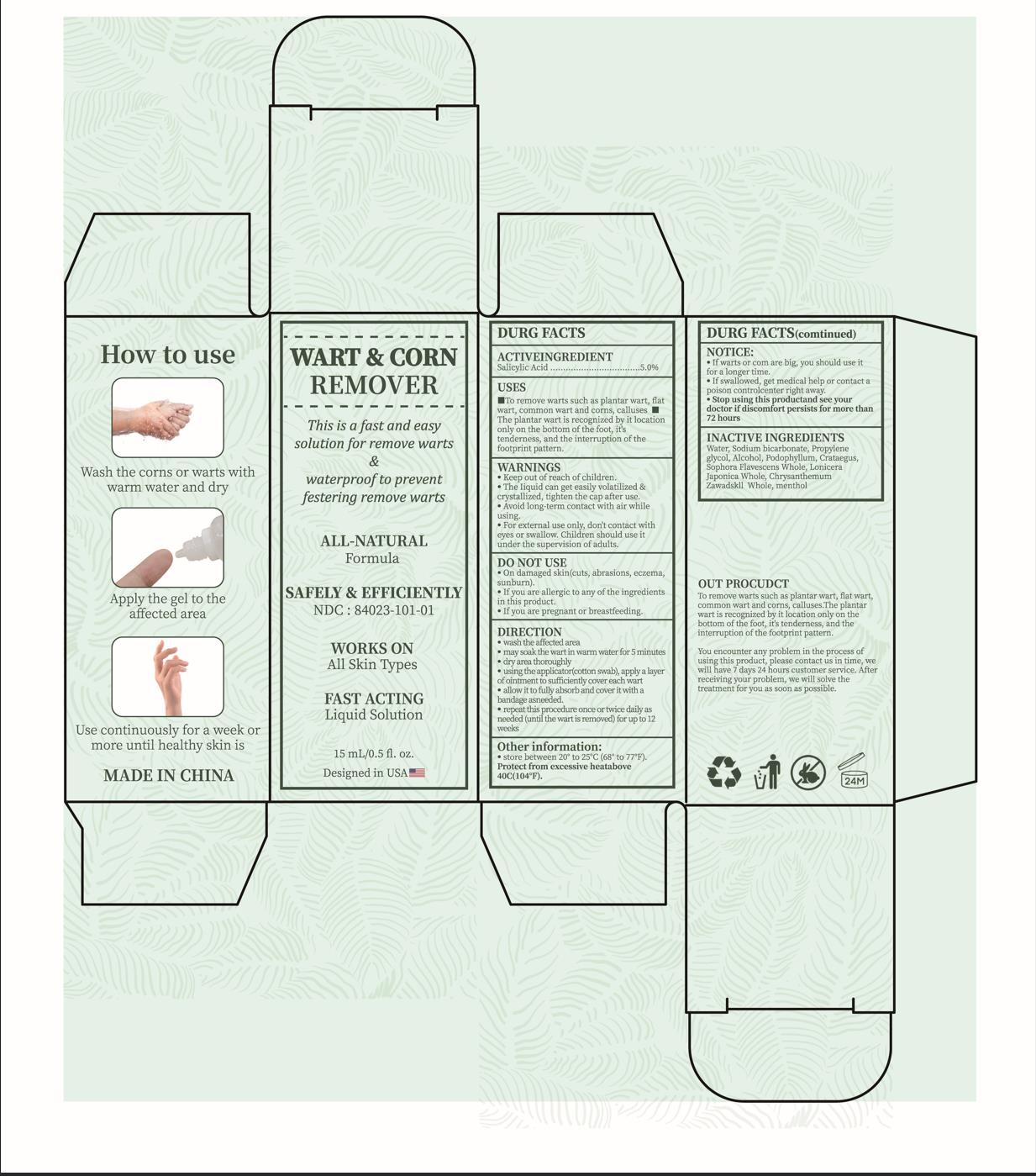
Label: WART CORN REMOVER- salicylic acid liquid
- NDC Code(s): 84023-101-01
- Packager: Shenzhen Yangan Technology Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
-
INDICATIONS & USAGE
Uses for the removal of common warts.The common wart is easily recognized by the rough cauliflower likeappearance of the surface.
The plantar wart is recognized by it slocation only on the bottom of the foot, its tenderness,and the interruption of the footprint pattern.
for removal of plantar warts on the bottom of the foot. - WARNINGS
- DO NOT USE
-
WHEN USING
wash the affected area.
may soak the wart in warmn water for 5 minutes.
dry area thoroughly.
using the applicator(cotton swab), apply a layer of ointment to sufficiently cover each wart.
allow it to fully absorb and cover it with a bandage asneeded.
repeat this procedure once or twice dailyas needed (until the wartis removed) for up to 12 weeks. - STOP USE
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
WART CORN REMOVER
salicylic acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84023-101 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 5 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) MENTHOL (UNII: L7T10EIP3A) CHRYSANTHEMUM ZAWADSKII WHOLE (UNII: 8Z1G3DJ591) LONICERA JAPONICA WHOLE (UNII: UV4X7MQQ43) SOPHORA FLAVESCENS WHOLE (UNII: X8KX602M5L) HAWTHORN LEAF WITH FLOWER (UNII: 6OM09RPY36) PODOPHYLLUM (UNII: 2S713A4VP3) ALCOHOL (UNII: 3K9958V90M) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM BICARBONATE (UNII: 8MDF5V39QO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84023-101-01 1 in 1 BOX 01/25/2024 1 15 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M028 01/25/2024 Labeler - Shenzhen Yangan Technology Co., Ltd. (419283765) Establishment Name Address ID/FEI Business Operations Shenzhen Yangan Technology Co., Ltd. 419283765 manufacture(84023-101)