Label: LIDOGEL- lidocaine hcl gel
- NDC Code(s): 80425-0372-1
- Packager: Advanced Rx Pharmacy of Tennessee, LLC
- This is a repackaged label.
- Source NDC Code(s): 59088-466
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 18, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Contains Lidocaine HCl 2.8% in a mild acidic vehicle. Lidocaine is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl), and has the following structure:
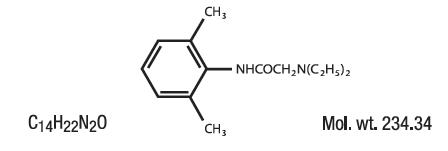
INGREDIENTS:Each gram of Lidogel™ 2.8% Gelcontains Lidocaine HCl USP 28 mg. Inactive Ingredients include: Aloe Barbadensis (Aloe Vera) Leaf Juice, Citric Acid, Hydroxyethylcellulose, Methylparaben, PEG-4, Propylene Glycol, Propylparaben, Purified Water.
-
CLINICAL PHARMACOLOGY:
MECHANISM OF ACTION:
Lidogel™ 2.8% Gelreleases Lidocaine from a mild acidic vehicle to stabilize the neuronal membrane by inhibiting the ionic fluxes
required for initiation and conduction of impulses, thereby affecting local anesthetic action. A mild acidic vehicle lowers pH to increase protection against alkaline irritants and to provide a favorable environment for healing.PHARMACOKINETICS:
Lidocaine may be absorbed following topical administration to mucous membranes, its rate and extent of absorption depending upon the specific site of application, duration of exposure, concentration, and total dosage. In general, the rate of absorption of local anesthetic agents following topical application occurs most rapidly after intratracheal administration. Lidocaine is also well-absorbed from the gastrointestinal tract, but little intact drug appears in the circulation because of biotransformation in the liver.
Lidocaine is metabolized rapidly by the liver and metabolites and unchanged drugs are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to but less potent than, those of Lidocaine. Approximately 90% of Lidocaine administered is excreted in the form of various metabolites and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2, 6-dimethylaniline. The plasma binding of Lidocaine is dependent on drug concentration and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 g of free base per mL, 60 to 80 percent of Lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-1-acid glycoprotein. Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion. Studies of Lidocaine metabolism following intravenous bolus injections have shown that the elimination half-life of this agent is typically 1.5 to 2 hours. Because of the rapid rate at which Lidocaine is metabolized, any condition that affects liver function may alter Lidocaine kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction. Renal dysfunction does not affect Lidocaine kinetics but may increase the accumulation of metabolites. Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of Lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 g free base per mL. In the rhesus monkey, arterial blood levels of 18-21 g/mL have been shown to be threshold for convulsive activity.
- CONTRAINDICATIONS:
- WARNINGS:
- PRECAUTIONS:
- CARCINOGENESIS, MUTAGENESIS, AND IMPAIRMENT OF FERTILITY:
-
USE IN PREGNANCY
Teratogenic Effects; Pregnancy Category B. Reproduction studies have been performed for Lidocaine in rats at doses up to 6.6 times the human dose and have revealed no evidence of harm to the fetus caused by Lidocaine. There are, however, no adequate and well-controlled studies in pregnant women. Animal reproduction studies are not always predictive of human response. General consideration should be given to this fact before administering Lidocaine to women of childbearing potential, especially during early pregnancy when maximum organogenesis takes place.
- NURSING MOTHERS:
- PEDIATRIC USE:
- ADVERSE REACTIONS:
- DOSAGE AND ADMINISTRATION:
- HOW SUPPLIED:
- STORAGE AND HANDLING:
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LIDOGEL
lidocaine hcl gelProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:80425-0372(NDC:59088-466) Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 28 mg in 1 g Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) HYDROXYETHYL CELLULOSE, UNSPECIFIED (UNII: T4V6TWG28D) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) POLYETHYLENE GLYCOL 200 (UNII: R95B8J264J) ALOE VERA LEAF (UNII: ZY81Z83H0X) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80425-0372-1 100 g in 1 TUBE; Type 0: Not a Combination Product 01/18/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 01/18/2023 Labeler - Advanced Rx Pharmacy of Tennessee, LLC (117023142) Establishment Name Address ID/FEI Business Operations Advanced Rx Pharmacy of Tennessee, LLC 117023142 repack(80425-0372)


