Label: vasocon- Naphazoline hydrochloride solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 58768-844-15 - Packager: CIBA Vision
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated August 31, 2006
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- N/A - Section Title Not Found In Database
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Naphazoline HCl, an ocular vasoconstrictor, is an imidazoline derivative sympathomimetic amine. The active ingredient is represented by the structural formula:
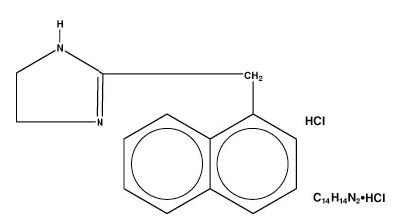
Chemical Name:
2-(1-naphthylmethyl)-2-imidazoline monohydrochloride
VASOCON REGULAR ophthalmic solution is a sterile solution containing 1 mg/mL naphazoline hydrochloride in an isotonic solution containing polyethylene glycol 8000, sodium chloride, polyvinyl alcohol, edetate disodium and purified water; preserved with benzalkonium chloride. Hydrochloric acid and/or sodium hydroxide added to adjust pH. It has a pH of 5.5 to 7.0.
-
CLINICAL PHARMACOLOGY
Naphazoline constricts the vascular system of the conjunctiva. It is presumed that this effect is due to direct stimulation action of the drug upon the alpha adrenergic receptors in the arterioles of the conjunctiva resulting in decreased conjunctival congestion. Naphalozine belongs to the imidazoline class of sympathomimetics.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
- WARNINGS
-
PRECAUTIONS
General
Use with caution in the presence of hypertension, cardiovascular abnormalities, hyperglycemia (diabetes), hyperthyroidism, ocular infection or injury and when other medications are being used.
Patient Information
Patients should be advised to discontinue the drug and consult a physician if relief is not obtained within 48 hours of therapy, if irritation, blurring, or redness persists or increases, or if symptoms of systemic absorption occur, i.e., dizziness, headache, nausea, decrease in body temperature, or drowsiness.
To prevent contaminating the dropper tip and solution, do not touch the eyelids or the surrounding area with the dropper tip of the bottle. If solution changes color or becomes cloudy, do not use.
Drug Interactions
Concurrent use of maprotiline or tricyclic antidepressants and naphazoline may potentiate the pressor effect of naphazoline. Patients under therapy with MAO inhibitors may experience a severe hypertensive crisis if given a sympathomimetic drug. (See WARNINGS).
Pregnancy Category C
Animal reproduction studies have not been conducted with naphazoline. It is also not known whether naphazoline can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Naphazoline should be given to a pregnant woman only if clearly needed.
- ADVERSE REACTIONS
- DOSAGE AND ADMINISTRATION
-
HOW SUPPLIED
VASOCON REGULAR (naphazoline hydrochloride ophthalmic solution USP, 0.1%): 15 mL plastic squeeze bottle with dropper tip. NDC 58768-844-15 To be dispensed only in original, unopened container. Store at controlled room temperature 15°-30°C (59°-86°F).
CAUTION: Federal law prohibits dispensing without prescription.
Mfd. by OMJ Pharmaceuticals, Inc.,
San Germán, P.R., 00683 for:
CIBA Vision
Ophthalmics®
___________________
Atlanta, Georgia 30155
6072-A
-
INGREDIENTS AND APPEARANCE
VASOCON
naphazoline hydrochloride solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:58768-844 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength naphazoline hydrochloride (UNII: MZ1131787D) (naphazoline - UNII:H231GF11BV) 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength benzalkonium chloride () edetate disodium (UNII: 7FLD91C86K) hydrochloric acid (UNII: QTT17582CB) polyethylene glycol 8000 () polyvinyl alcohol () water (UNII: 059QF0KO0R) sodium chloride (UNII: 451W47IQ8X) sodium hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58768-844-15 15 mL in 1 BOTTLE, DROPPER Labeler - CIBA Vision
