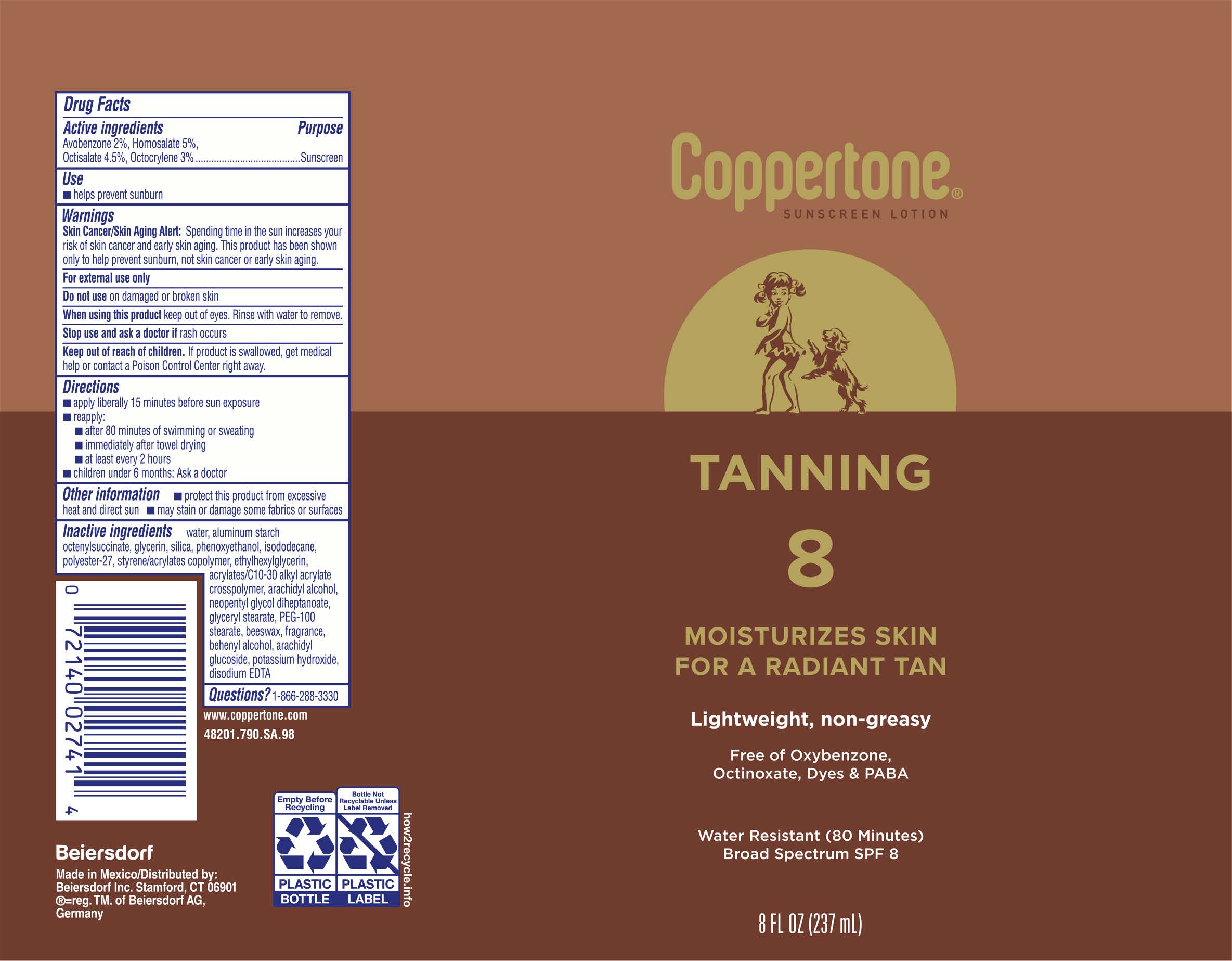
Label: COPPERTONE TANNING SUNSCREEN SPF 8- avobenzone 2%, homosalate 5%, octisalate 4.5%, octocrylene 3% lotion
- NDC Code(s): 66800-7034-8
- Packager: Beiersdorf Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 16, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients
- Purpose
- Use
- Warnings
- DO NOT USE
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
-
Inactive ingredients
water, aluminum starch octenylsuccinate, glycerin, silica, phenoxyethanol, isododecane, polyester-27, styrene/acrylates copolymer, ethylhexylglycerin, acrylates/C10-30 alkyl acrylate crosspolymer, arachidyl alcohol, neopentyl glycol diheptanoate, glyceryl stearate, PEG-100 stearate, beeswax, fragrance, behenyl alcohol, arachidyl glucoside, potassium hydroxide, disodium EDTA
- Questions?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
COPPERTONE TANNING SUNSCREEN SPF 8
avobenzone 2%, homosalate 5%, octisalate 4.5%, octocrylene 3% lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66800-7034 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 4.5 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 3 g in 100 g AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 2 g in 100 g HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 5 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) STYRENE/ACRYLAMIDE COPOLYMER (500000 MW) (UNII: 5Z4DPO246A) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) ARACHIDYL GLUCOSIDE (UNII: 6JVW35JOOJ) NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) FRAGRANCE FLORAL ORC0902236 (UNII: R66Z4YW3X0) DISODIUM EDTA-COPPER (UNII: 6V475AX06U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) POLYESTER-7 (UNII: 0841698D2F) PEG-100 STEARATE (UNII: YD01N1999R) SYNTHETIC BEESWAX (UNII: 08MNR5YE2R) ALUMINUM STARCH OCTENYLSUCCINATE (UNII: I9PJ0O6294) GLYCERIN (UNII: PDC6A3C0OX) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) PHENOXYETHANOL (UNII: HIE492ZZ3T) ISODODECANE (UNII: A8289P68Y2) ACRYLATES/C10-30 ALKYL ACRYLATE CROSSPOLYMER (60000 MPA.S) (UNII: 8Z5ZAL5H3V) ARACHIDYL ALCOHOL (UNII: 1QR1QRA9BU) DOCOSANOL (UNII: 9G1OE216XY) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66800-7034-8 237 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 01/31/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/31/2024 Labeler - Beiersdorf Inc (001177906)