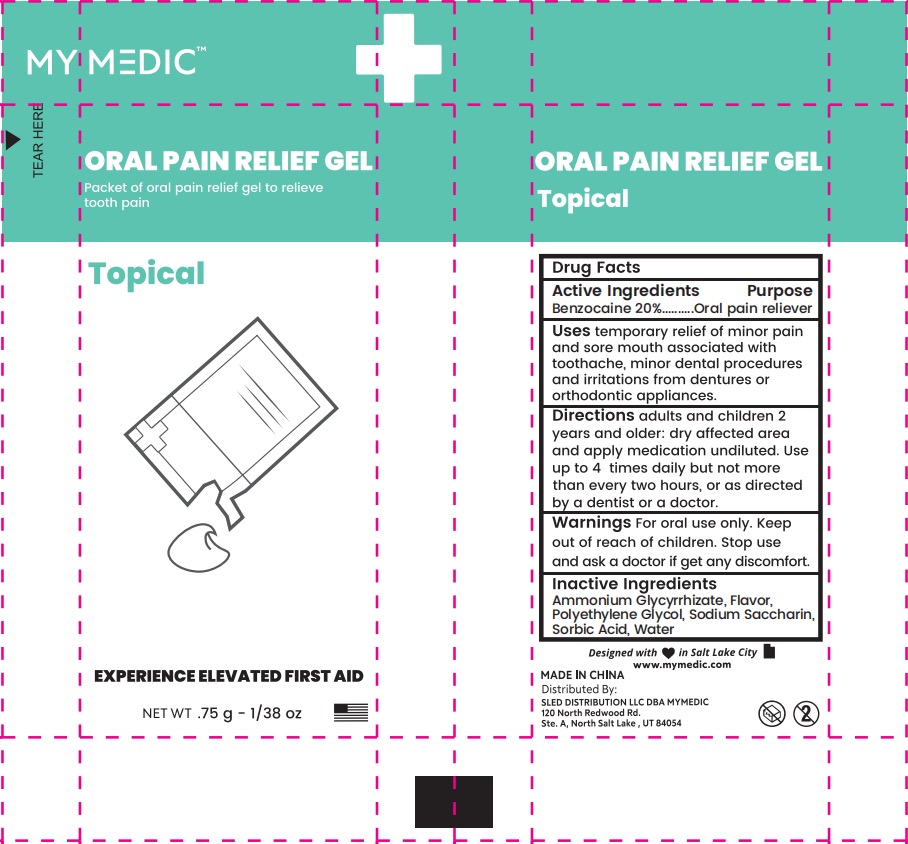
Label: MYMEDIC ORAL PAIN RELIEF GEL- benzocaine gel
- NDC Code(s): 43473-306-01
- Packager: Nantong Health & Beyond Hygienic Products Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 16, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DOSAGE & ADMINISTRATION
- WARNINGS
- INACTIVE INGREDIENT
- INDICATIONS & USAGE
- KEEP OUT OF REACH OF CHILDREN
- PURPOSE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MYMEDIC ORAL PAIN RELIEF GEL
benzocaine gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:43473-306 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 20 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SACCHARIN SODIUM (UNII: SB8ZUX40TY) AMMONIUM GLYCYRRHIZATE (UNII: 3VRD35U26C) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) SORBIC ACID (UNII: X045WJ989B) Product Characteristics Color Score Shape Size 65mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:43473-306-01 0.75 g in 1 POUCH; Type 0: Not a Combination Product 01/16/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 01/16/2024 Labeler - Nantong Health & Beyond Hygienic Products Inc. (421280161) Registrant - Nantong Health & Beyond Hygienic Products Inc. (421280161) Establishment Name Address ID/FEI Business Operations Nantong Health & Beyond Hygienic Products Inc. 421280161 manufacture(43473-306)