Label: MEDIKOII BIO ACTIVE VITAL SHOT AMPOULE- niacinamide, adenosine gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 72695-0011-1, 72695-0011-2 - Packager: THERANOTICS CO.,LTD.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated May 29, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
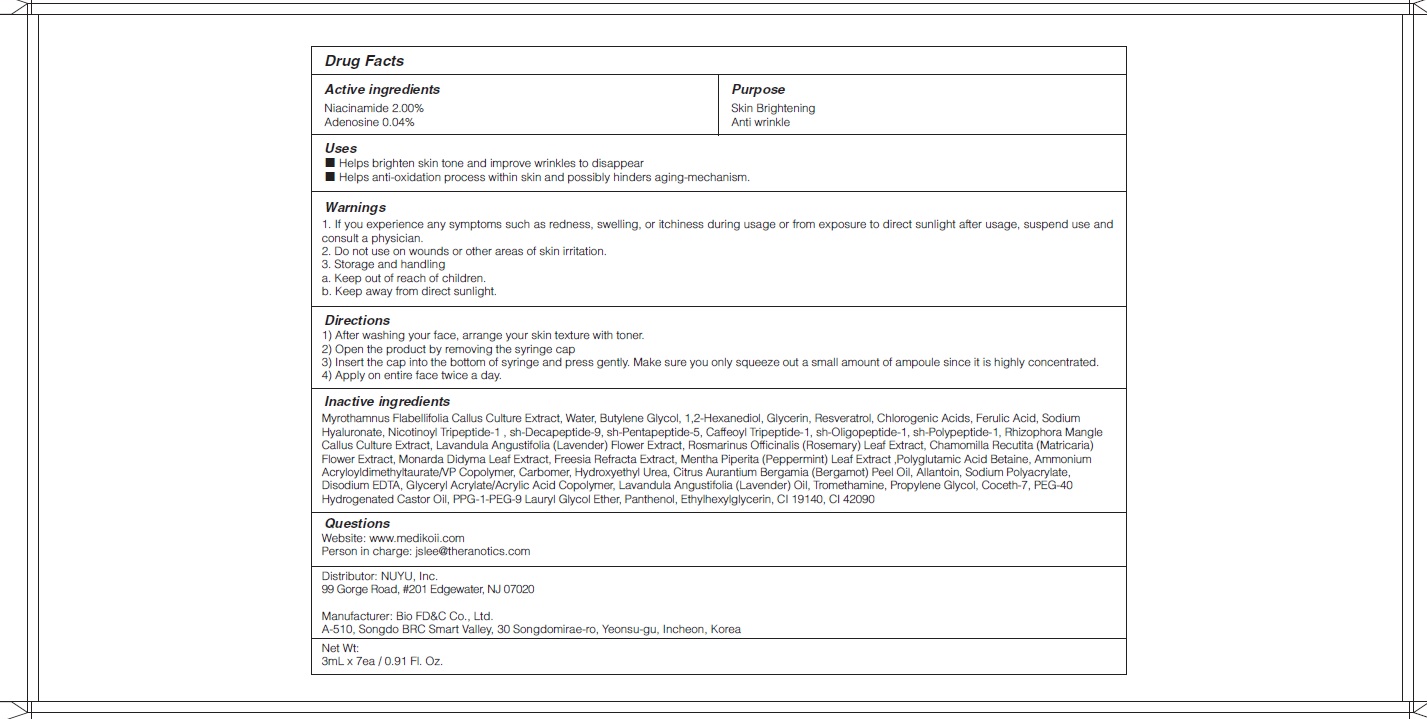
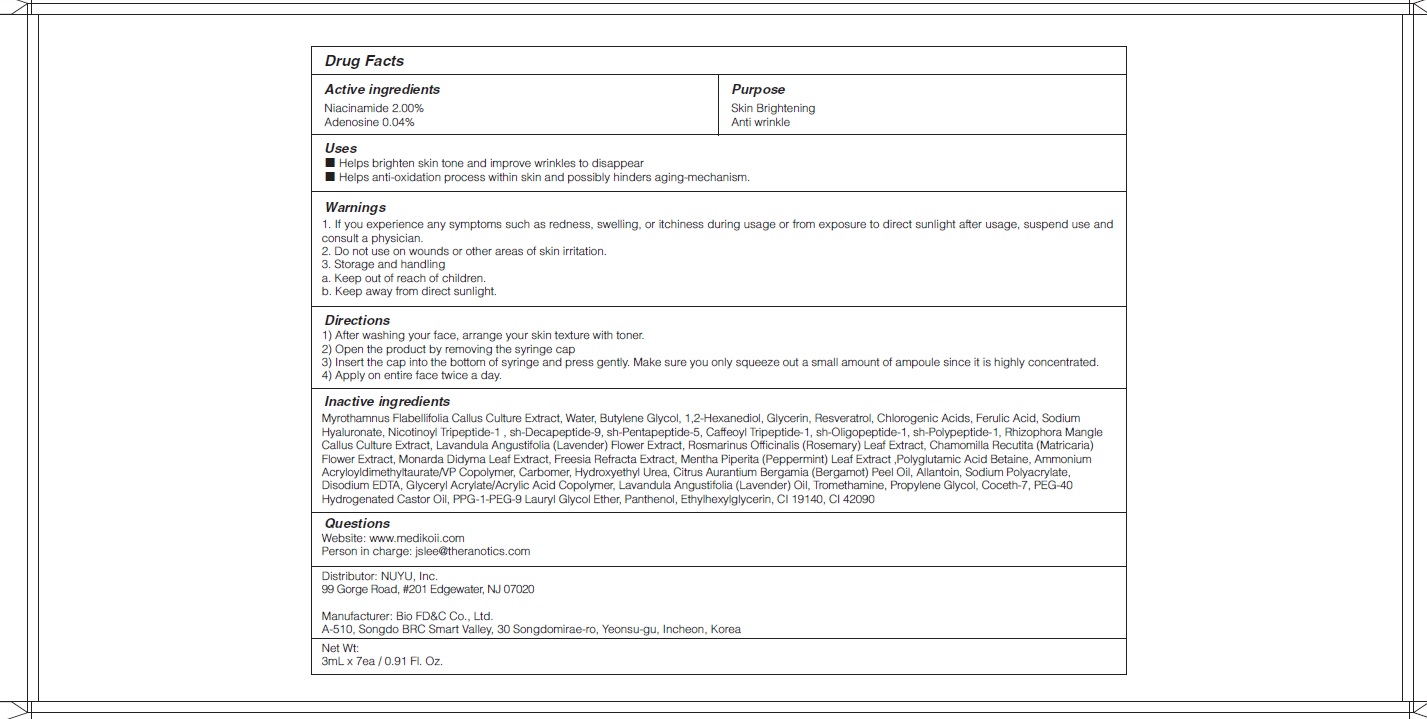
- ACTIVE INGREDIENTS
-
INACTIVE INGREDIENTS
Myrothamnus Flabellifolia Callus Culture Extract, Water, Butylene Glycol, 1,2-Hexanediol, Glycerin, Resveratrol, Chlorogenic Acids, Ferulic Acid,
Sodium Hyaluronate, Nicotinoyl Tripeptide-1 , sh-Decapeptide-9, sh-Pentapeptide-5, Caffeoyl Tripeptide-1, sh-Oligopeptide-1, sh-Polypeptide-1,
Rhizophora Mangle Callus Culture Extract, Lavandula Angustifolia (Lavender) Flower Extract, Rosmarinus Officinalis (Rosemary) Leaf Extract, Chamomilla Recutita (Matricaria) Flower Extract, Monarda Didyma Leaf Extract, Freesia Refracta Extract, Mentha Piperita (Peppermint) Leaf Extract ,Polyglutamic Acid, Betaine, Ammonium Acryloyldimethyltaurate/VP Copolymer, Carbomer, Hydroxyethyl Urea, Citrus Aurantium Bergamia (Bergamot) Peel Oil, Allantoin, Sodium Polyacrylate, Disodium EDTA, Glyceryl Acrylate/Acrylic Acid Copolymer, Lavandula Angustifolia (Lavender) Oil, Tromethamine, Propylene Glycol, Coceth-7, PEG-40 Hydrogenated Castor Oil, PPG-1-PEG-9 Lauryl Glycol Ether, Panthenol, Ethylhexylglycerin, CI 19140, CI 42090 - PURPOSE
-
WARNINGS
1.If you experience any symptoms such as redness, swelling, or itchiness during usage or from exposure to direct sunlight after usage, suspend use and consult a physician.
2. Do not use on wounds or other areas of skin irritation.
3. Storage and handling
a. Keep out of reach of children.
b. Keep away from direct sunlight. - KEEP OUT OF REACH OF CHILDREN
- Uses
-
Directions
1) After washing your face, arrange your skin texture with toner.
2) Open the product by removing the syringe cap
3) Insert the cap into the bottom of syringe and press gently. Make sure you only squeeze out a small amount of ampoule since it is highly concentrated.
4) Apply on entire face twice a day.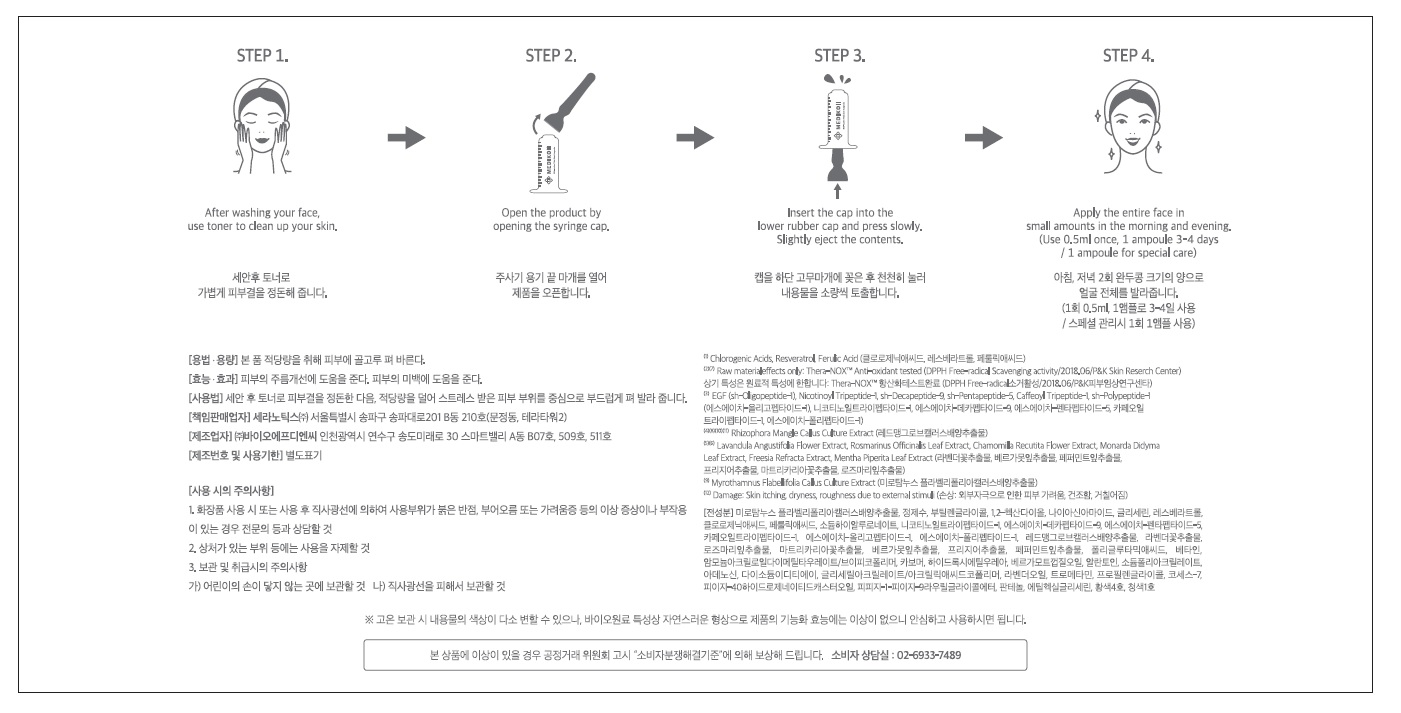
- QUESTIONS
- PACKAGE LABEL (Front)
- PACKAGE LABEL (Drug Facts Label)
-
INGREDIENTS AND APPEARANCE
MEDIKOII BIO ACTIVE VITAL SHOT AMPOULE
niacinamide, adenosine gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72695-0011 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Niacinamide (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) Niacinamide 2 g in 100 mL ADENOSINE (UNII: K72T3FS567) (ADENOSINE - UNII:K72T3FS567) ADENOSINE 0.04 g in 100 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Butylene Glycol (UNII: 3XUS85K0RA) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72695-0011-2 7 in 1 CARTON 05/01/2020 1 NDC:72695-0011-1 3 mL in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/01/2020 Labeler - THERANOTICS CO.,LTD. (694963085) Registrant - THERANOTICS CO.,LTD. (694963085) Establishment Name Address ID/FEI Business Operations BIO-FD&C. Co., Ltd. 688203268 manufacture(72695-0011)