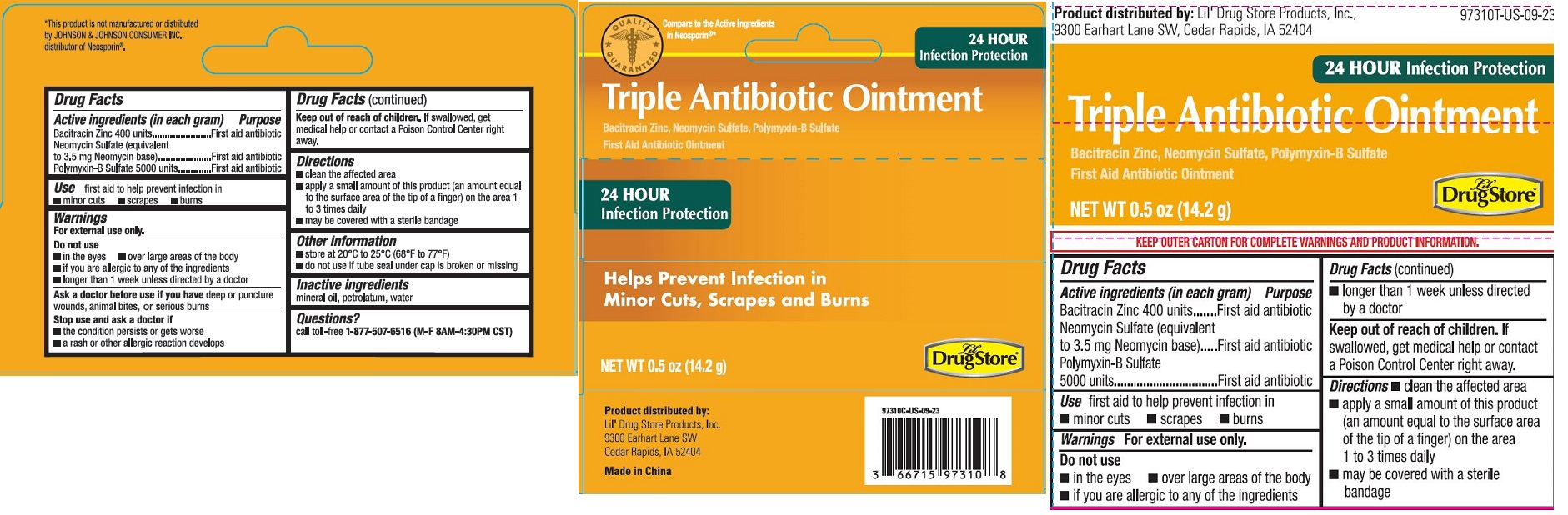
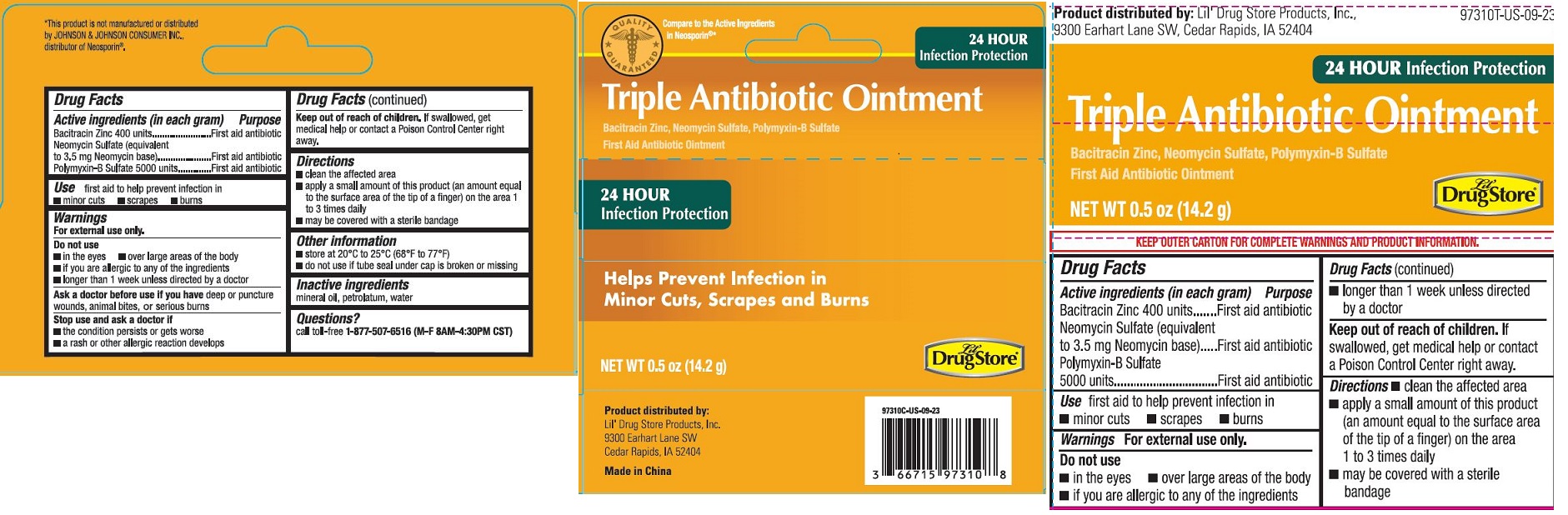
Label: TRIPLE ANTIBIOTIC OINTMENT, LIL DRUG STORE- triple antibiotic ointment
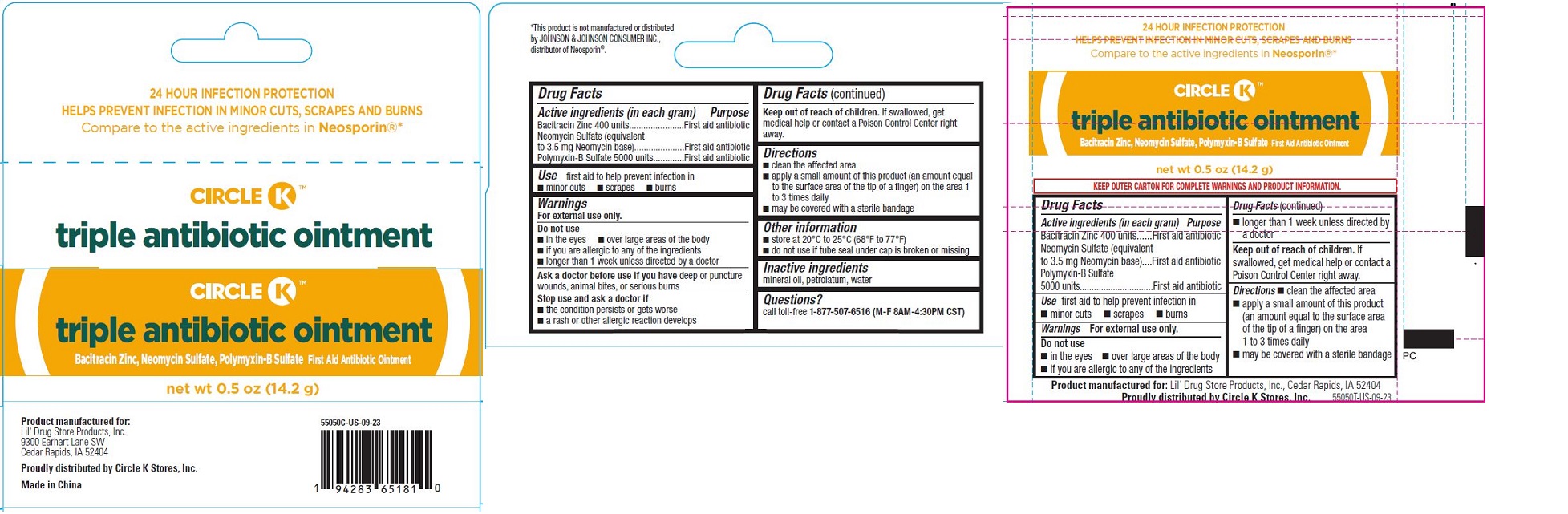
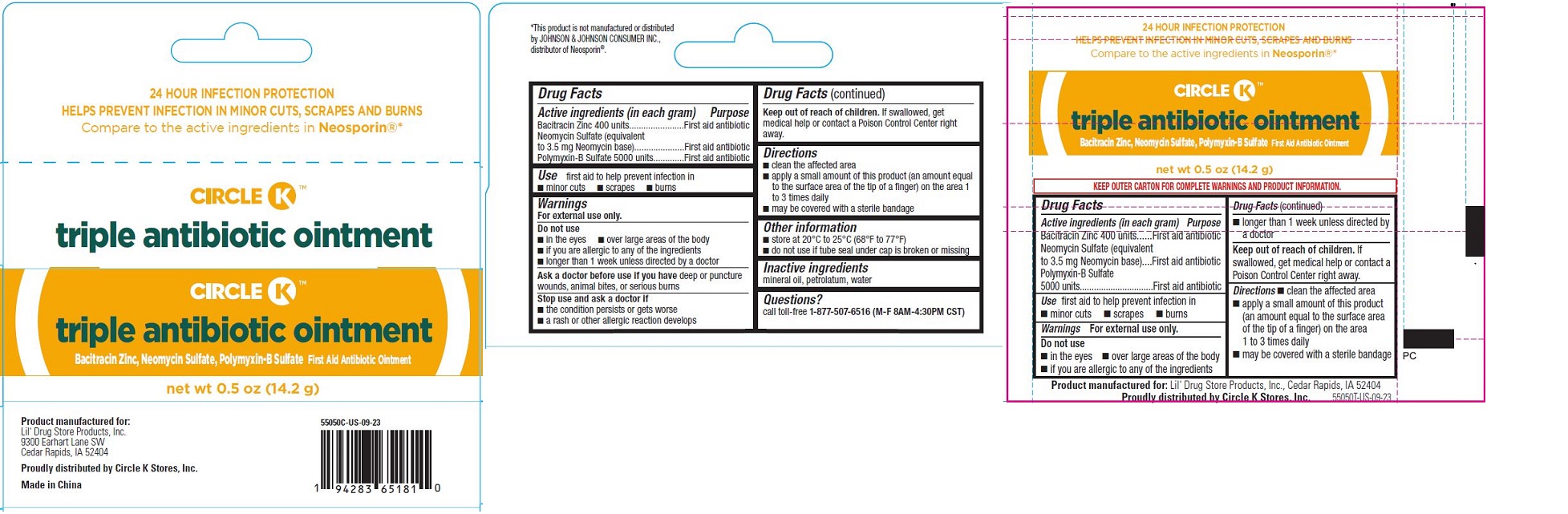
TRIPLE ANTIBIOTIC OINTMENT, CIRCLE K- triple antibiotic ointment
- NDC Code(s): 66715-5405-0, 66715-9631-0
- Packager: Lil' Drug Store products, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 3, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients (in each gram)
- Purpose
- Use
-
Warnings
Warnings
For external use only.Do not use
Do not use
- in the eyes
- over large areas of the body
- if you are allergic to any of the ingredients
- longer than 1 week unless directed by a doctor
Ask a doctor before use
Ask a doctor before use if you have deep or puncture wounds, animal bites, or serious burns
- Directions
- Other information
- Inactive ingredients
- Questions?
-
Triple Antibiotic Ointment, Lil' Drug Store
® - PDP/Package
[caduceus]
Compare to the Active Ingredients
in Neosporin ®
24 HOUR
Infection Protection
Triple Antibiotic Ointment
Bacitracin Zinc, Neomycin Sulfate, Polymyxin-B Sulfate
First Aid Antibiotic Ointment
Helps Prevent Infection in
Minor Cuts, Scrapes and Burns
NET WT 0.5 oz (14.2 g)
[Lil' Drug Store logo]
- Triple Antibiotic Ointment, Circle K™ - PDP/Package
-
INGREDIENTS AND APPEARANCE
TRIPLE ANTIBIOTIC OINTMENT, LIL DRUG STORE
triple antibiotic ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66715-9631 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN SULFATE 3.5 mg in 1 g BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) PETROLATUM (UNII: 4T6H12BN9U) WATER (UNII: 059QF0KO0R) Product Characteristics Color white ((translucent to opaque)) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66715-9631-0 1 in 1 CARTON 12/28/2023 1 14.2 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 12/28/2023 TRIPLE ANTIBIOTIC OINTMENT, CIRCLE K
triple antibiotic ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66715-5405 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN SULFATE 3.5 mg in 1 g BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) PETROLATUM (UNII: 4T6H12BN9U) WATER (UNII: 059QF0KO0R) Product Characteristics Color white ((translucent to opaque)) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66715-5405-0 1 in 1 CARTON 12/28/2023 1 14.2 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 12/28/2023 Labeler - Lil' Drug Store products, Inc. (093103646)