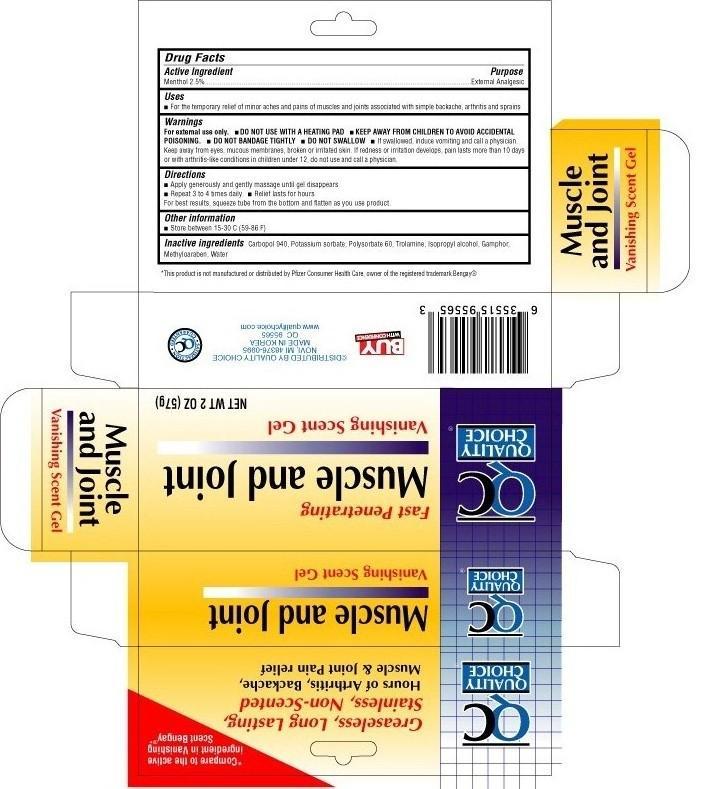
Label: QUALITY CHOICE MUSCLE AND JOINT- menthol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 63868-013-02 - Packager: Chain Drug Marketing Association
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 16, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Uses
-
Warnings
For external use only.
- DO NOT USE WITH A HEATING PAD
- DO NOT BANDAGE TIGHTLY
- DO NOT SWALLOW
- If swallowed, induce vomiting and call a physician. Keep away from eyes, mucous membranes, broken or irritated skin. If redness or irritation develops, pain lasts more than 10 days or with arthritis-like conditions in children under 12, do not use and call a physician.
- Directions
- Other information
- Inactive ingredients
- Package label
- Purpose
-
INGREDIENTS AND APPEARANCE
QUALITY CHOICE MUSCLE AND JOINT
menthol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63868-013 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Menthol (UNII: L7T10EIP3A) (Menthol - UNII:L7T10EIP3A) Menthol 2.5 g in 100 g Inactive Ingredients Ingredient Name Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) CARBOMER HOMOPOLYMER TYPE C (UNII: 4Q93RCW27E) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) isopropyl alcohol (UNII: ND2M416302) methylparaben (UNII: A2I8C7HI9T) polysorbate 60 (UNII: CAL22UVI4M) water (UNII: 059QF0KO0R) trolamine (UNII: 9O3K93S3TK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63868-013-02 1 in 1 BOX 1 57 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 09/16/2011 Labeler - Chain Drug Marketing Association (011920774)