Label: NAPROXEN SODIUM AND DIPHENHYDRAMINE HCL tablet, film coated
- NDC Code(s): 62207-814-38, 62207-814-39, 62207-814-47, 62207-814-95
- Packager: Granules India Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
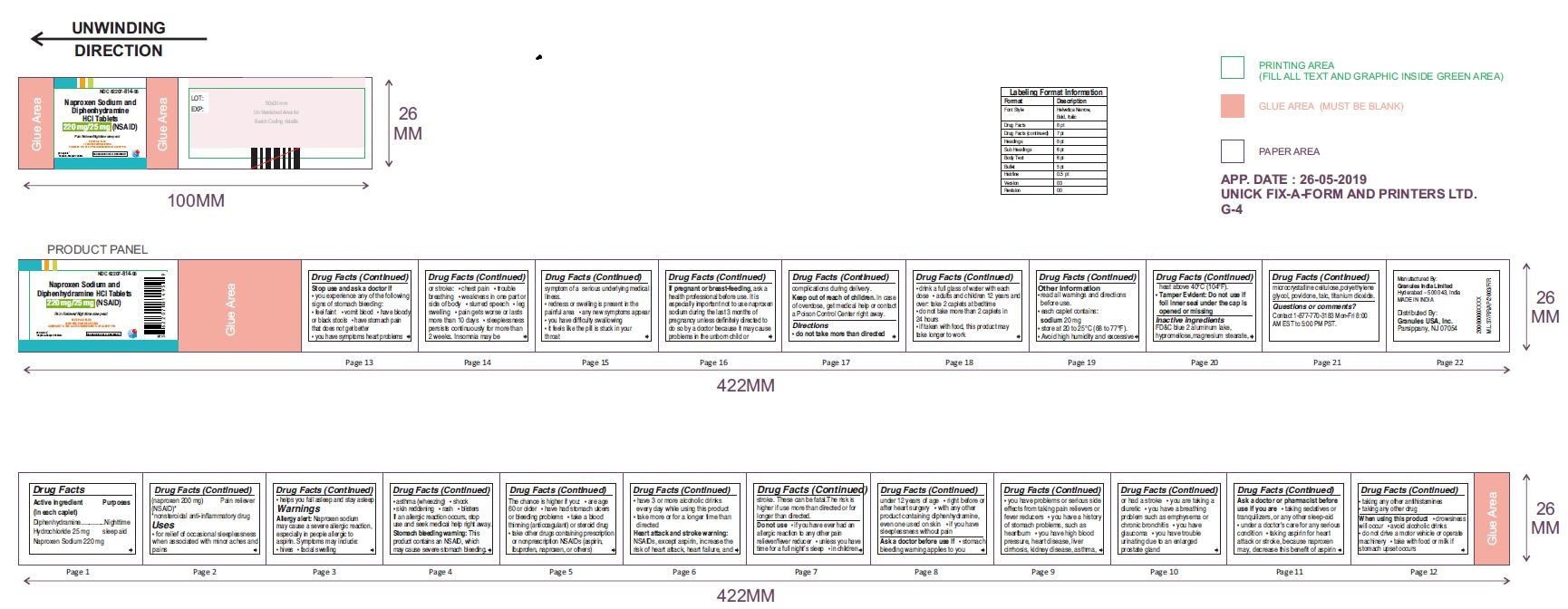
- ACTIVE INGREDIENT(S)
- PURPOSE
- USE(S)
-
WARNINGS
Allergy alert:
Naproxen sodium may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
• hives
• facial swelling
• asthma(wheezing)
• shock
• skin reddening
• rash
• blisters
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning:
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
• are age 60 or older
• have had stomach ulcers or bleeding problems
• take a blood thinning (anticoagulant) or steroid drug
• take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
• have 3 or more alcoholic drinks every day while using this product
• take more or for a longer time than directed
Heart attack and stroke warning:
NSAIDs, except aspirin, increase the risk of heart attack, heart failure, and stroke. These can be fatal. The risk is higher if use more than directed or for longer than directed. -
DO NOT USE
• if you have ever had an allergic reaction to any other pain reliever/fever reducer
• unless you have time for a full night’s sleep
• in children under 12 years of age
• right before or after heart surgery
• with any other product containing diphenhydramine, even one used on skin
• if you have sleeplessness without pain -
ASK A DOCTOR BEFORE USE IF
• stomach bleeding warning applies to you
• you have problems or serious side effects from taking pain relievers or fever reducers
• you have a history of stomach problems, such as heartburn
• you have high blood pressure, heart disease, liver cirrhosis, kidney disease, asthma, Drug Facts Drug Facts or had a stroke
• you are taking a diuretic
• you have a breathing problem such as emphysema or chronic bronchitis
• you have glaucoma
• you have trouble urinating due to an enlarged prostate gland - ASK A DOCTOR OR PHARMACIST BEFORE USE IF
- WHEN USING THIS PRODUCT
-
STOP USE AND ASK DOCTOR IF
• you experience any of the following signs of stomach bleeding:
• feel faint
• vomit blood
• have bloody or black stools
• have stomach pain that does not get better
• you have symptoms heart problems or stroke:
• chest pain
• trouble breathing
• weakness in one part or side of body
• slurred speech
• leg swelling
• pain gets worse or lasts more than 10 days
• sleeplessness persists continuously for more than 2 weeks. Insomnia may be symptom of a serious underlying medical illness.
• redness or swelling is present in the painful area
• any new symptoms appear
• you have difficulty swallowing
• it feels like the pill is stuck in your throat - IF PREGNANT OR BREASTFEEDING
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS ?
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NAPROXEN SODIUM AND DIPHENHYDRAMINE HCL
naproxen sodium and diphenhydramine hcl tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62207-814 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NAPROXEN SODIUM (UNII: 9TN87S3A3C) (NAPROXEN - UNII:57Y76R9ATQ) NAPROXEN SODIUM 220 mg DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 2--ALUMINUM LAKE (UNII: 4AQJ3LG584) HYPROMELLOSE 2910 (3 MPA.S) (UNII: 0VUT3PMY82) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) Product Characteristics Color blue Score no score Shape CAPSULE Size 15mm Flavor Imprint Code G;17 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62207-814-95 1 in 1 CARTON 02/20/2021 1 20 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:62207-814-47 500 in 1 BOTTLE; Type 0: Not a Combination Product 02/20/2021 3 NDC:62207-814-38 37500 in 1 POUCH; Type 0: Not a Combination Product 02/20/2021 4 NDC:62207-814-39 7500 in 1 POUCH; Type 0: Not a Combination Product 02/20/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA213663 02/20/2021 Labeler - Granules India Ltd (915000087)