Label: UREA CREAM 40%- urea cream
- NDC Code(s): 71399-8456-1, 71399-8456-3, 71399-8456-7
- Packager: Akron Pharma Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated March 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
- CLINICAL PHARMACOLOGY
- PHARMACOKINETICS
-
INDICATIONS & USAGE
Indications and Usage
For debridement and promotion of normal healing of hyperkeratotic surface lesions, particularly where healing is retarded by local infection, necrotic tissue, fibrinous or purulent debris or eshar. Urea is useful for the treatment of hyperkeratotic conditions such as dry, rough skin, dermatitis, psoriasis, xerosis, ichthyosis, eczema, keratosis pilaris, keratosis palmaris, keratoderma, corns and calluses, as well as damaged, ingrown and devitalized nails.
- CONTRAINDICATIONS
- WARNINGS
- PRECAUTIONS
-
PREGNANCY
PREGNANACY
Pregnancy Category B. Animal reproduction studies have revealed no evidence of harm to the fetus, however, there are no adequate and well-controlled studies in pregnant women. Because animal reproductive studies are not always predictive of human response, Urea 40% should be given to a pregnant woman only if clearly needed.
- NURSING MOTHERS
- ADVERSE REACTIONS
-
DOSAGE & ADMINISTRATION
Dosage and Administration
Apply Urea 40% to affected skin twice per day, or as directed by your physician. Rub in until completely absorbed.
Apply to diseased or damaged nail(s) twice per day, or as directed by a physician.
All prescription substitutions and/or recommendations using this product shall be made subject to state and federal statutes as applicable. Please note: this is not an Orange Book product and has not been subjected to FDA therapeutic equivalency or other equivalency testing. No representation is made as to generic status or bioequivalency. Each person recommending a prescription substitution using this product shall make such recommendations based on each person's professional opinion and knowledge, upon evaluation of the active ingredients, excipients, inactive ingredients and chemical information provided herein.
-
HOW SUPPLIED
How Supplied
Urea 40% Cream 1 oz.(28.35 g): NDC 71399-8456-1; Urea 40% Cream 3 oz.(85 g): NDC NDC 71399-8456-3; Urea 40% Cream 7 oz.(198.45 g): NDC NDC 71399-8456-7
Store at 20°C to 25°C (68°F to 77°F), excursions permitted between 15°C and 30°C (between 59°F and 86°F). Protect from freezing and excessive heat. Keep bottle tightly closed.
Manufactured for:
Akron Pharma Inc.,
Fairfield, NJ-07004
www.akronpharma.comManufactured In USA
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
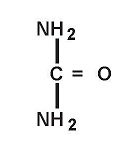
UREA CREAM 40%
urea creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:71399-8456 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength UREA (UNII: 8W8T17847W) (UREA - UNII:8W8T17847W) UREA 400 mg in 1 g Inactive Ingredients Ingredient Name Strength CETYL ALCOHOL (UNII: 936JST6JCN) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) CARBOMER HOMOPOLYMER TYPE C (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 4Q93RCW27E) MINERAL OIL (UNII: T5L8T28FGP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) TROLAMINE (UNII: 9O3K93S3TK) WHITE PETROLATUM (UNII: B6E5W8RQJ4) XANTHAN GUM (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71399-8456-1 28.35 g in 1 BOTTLE; Type 0: Not a Combination Product 11/04/2021 2 NDC:71399-8456-3 85 g in 1 BOTTLE; Type 0: Not a Combination Product 11/04/2021 3 NDC:71399-8456-7 198.45 g in 1 BOTTLE; Type 0: Not a Combination Product 11/04/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 11/04/2021 Labeler - Akron Pharma Inc. (067878881)