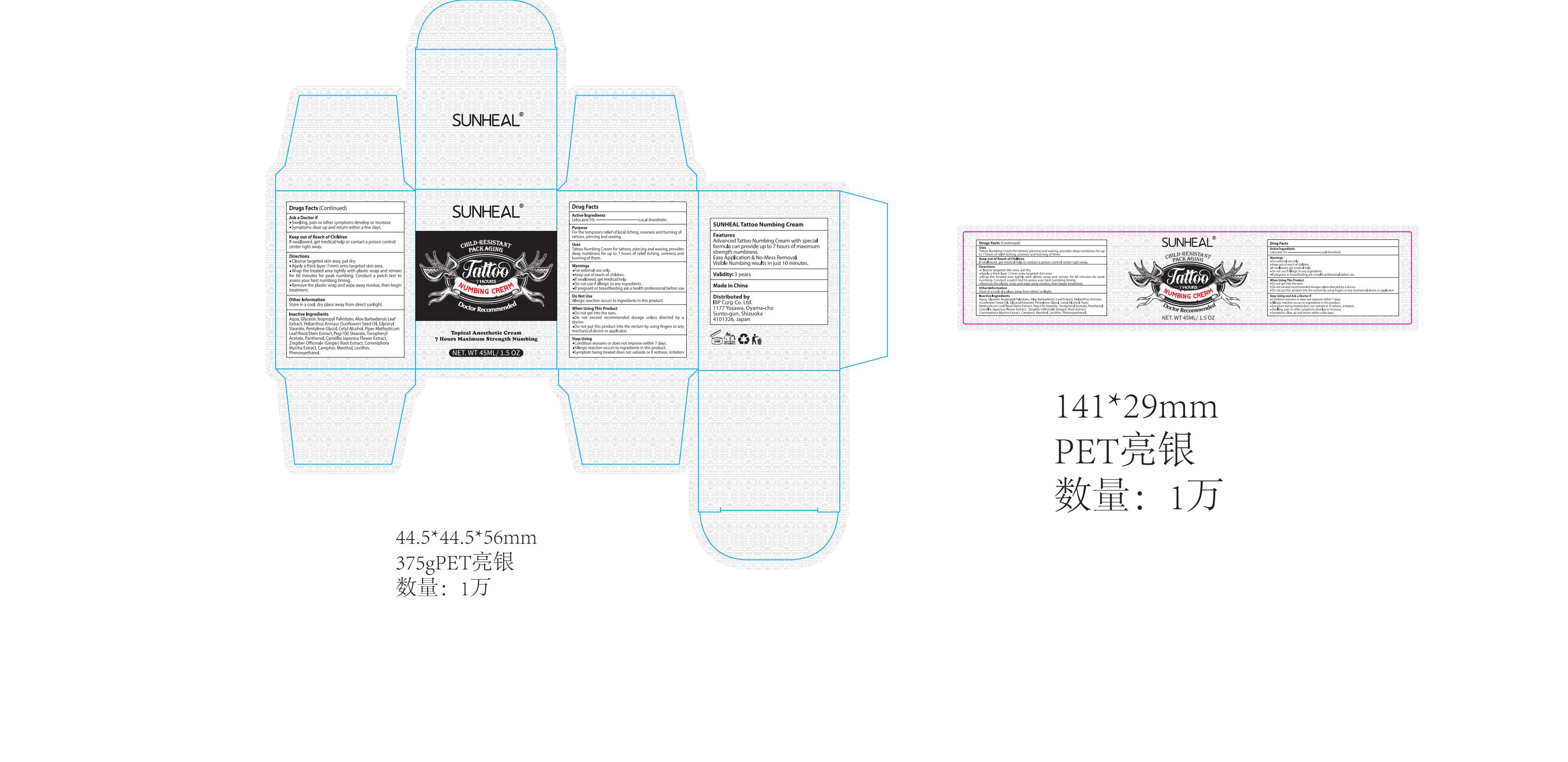
Label: SUNHEAL TATTOO NUMBING CREAM cream
- NDC Code(s): 83871-001-01
- Packager: IBP CORP CO. LTD.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 19, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
- When Using
- Stop Use
- Ask Doctor
- Keep Oot Of Reach Of Children
-
Directions
1.Cleanse targeted skin area, pat dry.
2.Apply a thick layer (1mm) onto targeted skin area.
3.Wrap the treated area tightly with plastic wrap and remain for 60 minutes for peak numbing. Conduct a patch test to assess your best numbing timing.
4.Remove the plastic wrap and wipe away residue, then begin treatment. - Other information
-
Inactive ingredients
Aqua, Glycerin, Isopropyl Palmitate, Aloe Barbadensis Leaf Extract,
Helianthus Annuus (Sunflower) Seed Oil, Glyceryl Stearate,
Pentylene Glycol, Cetyl Alcohol, Piper Methysticum Leaf/Root/Stem Extract, Peg-100 Stearate, Tocopheryl Acetate, Panthenol, Camellia Japonica Flower Extract,
Zingiber Officinale (Ginger) Root Extract, Commiphora Myrrha Extract, Camphor, Menthol, Lecithin, Phenoxyethanol. - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SUNHEAL TATTOO NUMBING CREAM
sunheal tattoo numbing cream creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83871-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 5 g in 100 g Inactive Ingredients Ingredient Name Strength ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) GINGER (UNII: C5529G5JPQ) CETYL ALCOHOL (UNII: 936JST6JCN) MACROPIPER METHYSTICUM ROOT (UNII: BOW48C81XP) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) SUNFLOWER OIL (UNII: 3W1JG795YI) PENTYLENE GLYCOL (UNII: 50C1307PZG) PANTHENOL (UNII: WV9CM0O67Z) MENTHOL (UNII: L7T10EIP3A) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ALOE VERA LEAF (UNII: ZY81Z83H0X) POLYOXYL 100 STEARATE (UNII: YD01N1999R) COMMIPHORA MYRRHA WHOLE (UNII: UU81N77RI7) CAMPHOR (NATURAL) (UNII: N20HL7Q941) CAMELLIA JAPONICA FLOWER (UNII: KUB8101TNF) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83871-001-01 45 g in 1 BOTTLE; Type 0: Not a Combination Product 12/18/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 12/18/2023 Labeler - IBP CORP CO. LTD. (692424700) Establishment Name Address ID/FEI Business Operations IBP CORP CO. LTD. 692424700 label(83871-001) , manufacture(83871-001)