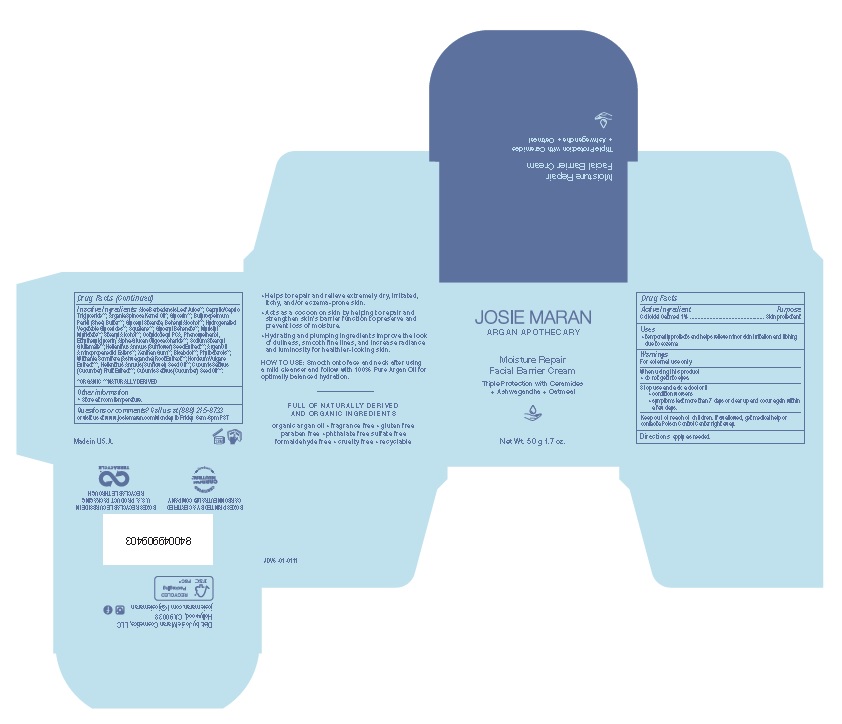
Label: JOSIE MARAN MOISTURE REPAIR FACIAL BARRIER- colloidal oatmeal 1% cream
- NDC Code(s): 54111-163-50
- Packager: Bentley Laboratories, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 19, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
-
INACTIVE INGREDIENT
Inactive Ingredients: Aloe Barbadensis Leaf Juice**, Caprylic/Capric
Triglyceride**, Argania Spinosa Kernel Oil*, Glycerin, Butyrospermum
Parkii (Shea) Butter**, Glyceryl Stearate, Behenyl Alcohol**, Hydrogenated
Vegetable Glycerides**, Squalane**, Glyceryl Behenate**, Myristyl
Myristate**, Stearyl Alcohol**, Octyldodecyl PCA, Phenoxyethanol,
Ethylhexylglycerin, Alpha-Glucan Oligosaccharide**, Sodium Stearoyl
Glutamate**, Helianthus Annuus (Sunflower) Seed Extract**, Argan Oil
Aminopropanediol Esters**, Xanthan Gum**, Bisabolol, Phytosterols**,
Withania Somnifera (Ashwagandha) Root Extract**, Hordeum vulgare
Extract**, Helianthus Annuus (Sunflower) Seed Oil**, Cucumis Sativus
(Cucumber) Fruit Extract**, Cucumis Sativus (Cucumber) Seed Oil**
*Organic ** Naturally Derived - OTHER SAFETY INFORMATION
- QUESTIONS
- Product Package
-
INGREDIENTS AND APPEARANCE
JOSIE MARAN MOISTURE REPAIR FACIAL BARRIER
colloidal oatmeal 1% creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54111-163 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OATMEAL (UNII: 8PI54V663Y) (OATMEAL - UNII:8PI54V663Y) OATMEAL 10.0 mg in 1 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ARGAN OIL (UNII: 4V59G5UW9X) GLYCERIN (UNII: PDC6A3C0OX) SHEA BUTTER (UNII: K49155WL9Y) GLYCERYL STEARATE CITRATE (UNII: WH8T92A065) DOCOSANOL (UNII: 9G1OE216XY) C14-18 MONO-AND DI-GLYCERIDES (UNII: 82TZ9D9546) SQUALANE (UNII: GW89575KF9) GLYCERYL MONOBEHENATE (UNII: A626UU0W2A) MYRISTYL MYRISTATE (UNII: 4042ZC00DY) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) OCTYLDODECYL PCA (UNII: E25TY46YTD) PHENOXYETHANOL (UNII: HIE492ZZ3T) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) .ALPHA.-GLUCAN OLIGOSACCHARIDE (UNII: S95658MI3W) SODIUM STEAROYL GLUTAMATE (UNII: 65A9F4P024) SUNFLOWER SEED (UNII: R9N3379M4Z) ARGAN OIL AMINOPROPANEDIOL ESTERS (UNII: 2HA4BS9RJJ) XANTHAN GUM (UNII: TTV12P4NEE) LEVOMENOL (UNII: 24WE03BX2T) WITHANIA SOMNIFERA ROOT (UNII: V038D626IF) HORDEUM VULGARE WHOLE (UNII: 8JBE478M5Q) SUNFLOWER OIL (UNII: 3W1JG795YI) CUCUMBER (UNII: YY7C30VXJT) CUCUMBER SEED OIL (UNII: AKP926H71P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54111-163-50 1 in 1 CARTON 03/09/2022 1 50 g in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 03/09/2022 Labeler - Bentley Laboratories, LLC (068351753)