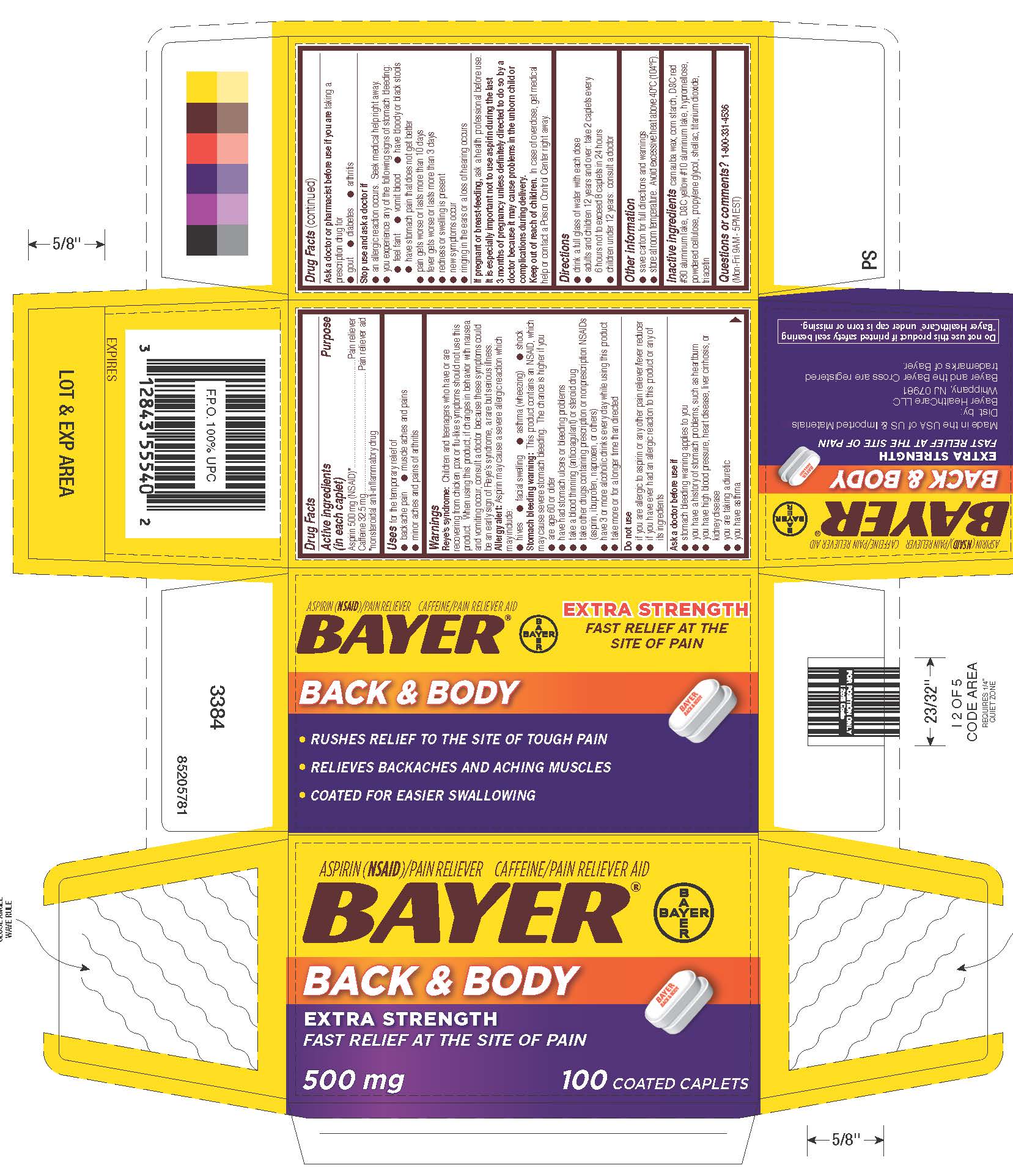
Label: BAYER BACK AND BODY EXTRA STRENGTH- aspirin,caffeine tablet
-
NDC Code(s):
0280-2150-01,
0280-2150-02,
0280-2150-10,
0280-2150-24, view more0280-2150-50
- Packager: Bayer HealthCare LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients (in each caplet)
- Purpose
- Uses
-
Warnings
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction which may include:
- hives
- facial swelling
- asthma (wheezing)
- shock
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
- Do not use
- Ask a doctor before use if
- ASK DOCTOR/PHARMACIST
-
Stop use and ask a doctor if
- an allergic reaction occurs. Seek medical help right away.
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- redness or swelling is present
- new symptoms occur
- ringing in the ears or a loss of hearing occurs
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other Information
- Inactive Ingredients
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BAYER BACK AND BODY EXTRA STRENGTH
aspirin,caffeine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0280-2150 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 35.5 mg ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 500 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) D&C RED NO. 30 (UNII: 2S42T2808B) ALUMINUM OXIDE (UNII: LMI26O6933) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) HYPROMELLOSES (UNII: 3NXW29V3WO) POWDERED CELLULOSE (UNII: SMD1X3XO9M) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) CARNAUBA WAX (UNII: R12CBM0EIZ) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color white Score no score Shape OVAL Size 19mm Flavor Imprint Code BAYER;BACK;BODY Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0280-2150-24 1 in 1 CARTON 01/12/2015 1 24 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:0280-2150-50 1 in 1 CARTON 01/12/2015 2 50 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC:0280-2150-10 1 in 1 CARTON 01/12/2015 3 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 4 NDC:0280-2150-02 200 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 01/12/2015 5 NDC:0280-2150-01 1 in 1 CARTON 01/12/2015 5 6 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 01/12/2015 Labeler - Bayer HealthCare LLC. (112117283)