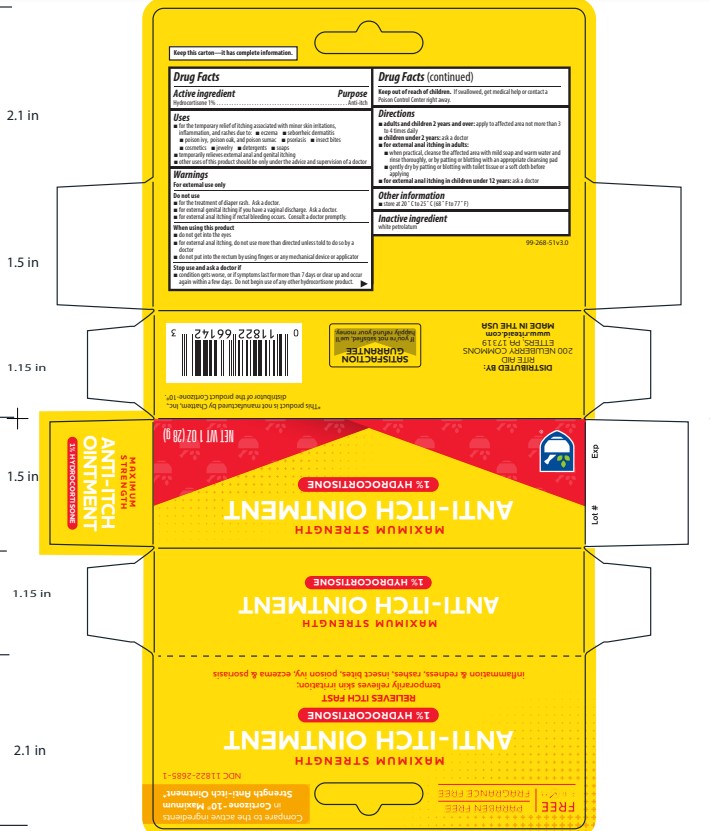
Label: RITE AID HYDROCORTISONE- hydrocortisone 1% ointment
- NDC Code(s): 11822-2685-1, 11822-2685-2
- Packager: Rite Aid
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 12, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
-
Uses
For the temporary relief of itching associated with minor skin irritations, inflammation, and rashes due to:
- eczema
- seborrheic dermatitis
- poison ivy, poison oak, and poison sumac
- psoriasis
- insect bites
- cosmetics
- jewelry
- detergents
- soaps
temporarily relieves external anal and genital itching
other uses of this product should be only under the advise and supervision of a doctor
-
Warnings
For external use only
Do not use
- for the treatment of diaper rash. Ask a doctor.
- for external genital itching if you have a vaginal discharge. Ask a doctor.
- for external anal itching if rectal bleeding occurs. Consult a doctor promptly.
When using this product
- do not get in the eyes
- for external anal itching, do not use more than directed unless told to do so by a doctor
- do not put into the rectum by using fingers or any mechanical device or applicator
-
Directions
adults and children 2 years and over: apply to affected area not more than 3 to 4 times daily
children under 2 years: ask a doctor
for external anal itching in adults:
- when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly, or by patting or blotting with an appropriate cleansing pad
- gently dry by patting or blotting with toilet tissue or a soft cloth before applying
for external anal itching in children under 12 years: ask a doctor
- Other information
- Inactive ingredient
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RITE AID HYDROCORTISONE
hydrocortisone 1% ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11822-2685 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE (UNII: WI4X0X7BPJ) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE 1 g in 100 g Inactive Ingredients Ingredient Name Strength WHITE PETROLATUM (UNII: B6E5W8RQJ4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11822-2685-1 1 in 1 CARTON 10/01/2023 1 28 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:11822-2685-2 1 in 1 CARTON 10/01/2023 2 59 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 10/01/2023 Labeler - Rite Aid (014578892) Establishment Name Address ID/FEI Business Operations Weeks & Leo Co., Inc. 005290028 manufacture(11822-2685)