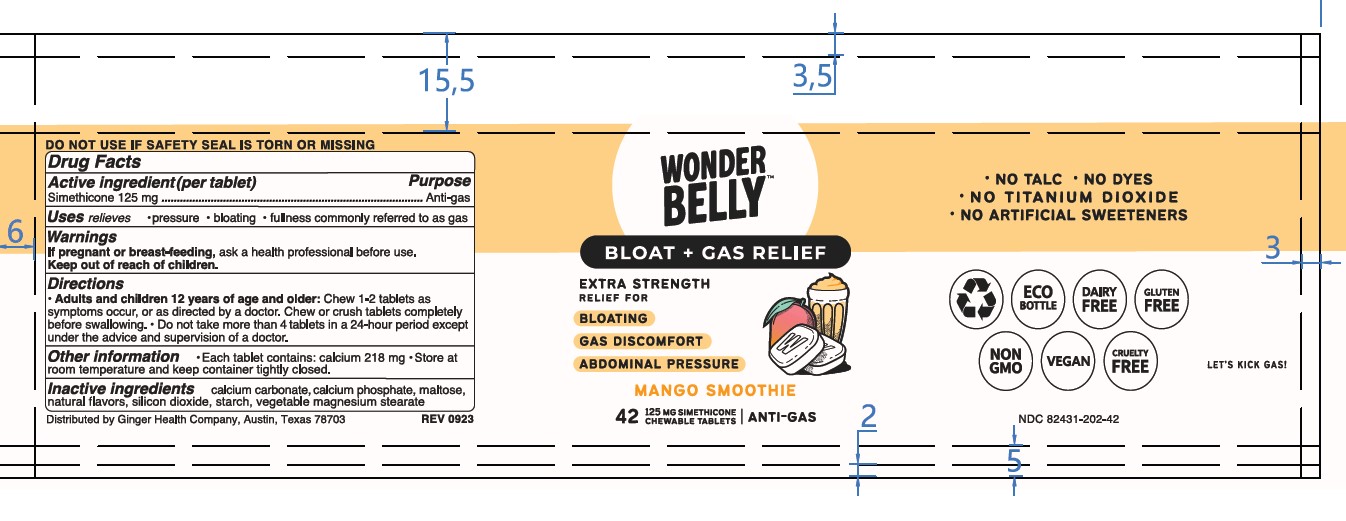
Label: WONDERBELLY EXTRA STRENGTH ANTI-GAS- simethicone tablet, chewable
- NDC Code(s): 82431-202-42
- Packager: Ginger Health Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 12, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- PREGNANCY OR BREAST FEEDING
- Directions
- Other Information
- Inactive ingredients
- Principal Display Panel-MS 42ct UC Rev 1123 Principal Display Panel-MS 42ct Bottle Rev 0923
-
INGREDIENTS AND APPEARANCE
WONDERBELLY EXTRA STRENGTH ANTI-GAS
simethicone tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82431-202 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 125 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTOSE MONOHYDRATE (UNII: DM477EE40D) TRIBASIC CALCIUM PHOSPHATE (UNII: 91D9GV0Z28) CALCIUM CARBONATE (UNII: H0G9379FGK) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color white Score no score Shape RECTANGLE (Rounded Corners) Size 17mm Flavor MANGO (Smoothie) Imprint Code W;202 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82431-202-42 42 in 1 BOTTLE; Type 0: Not a Combination Product 03/01/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M002 03/01/2024 Labeler - Ginger Health Company (118431122)