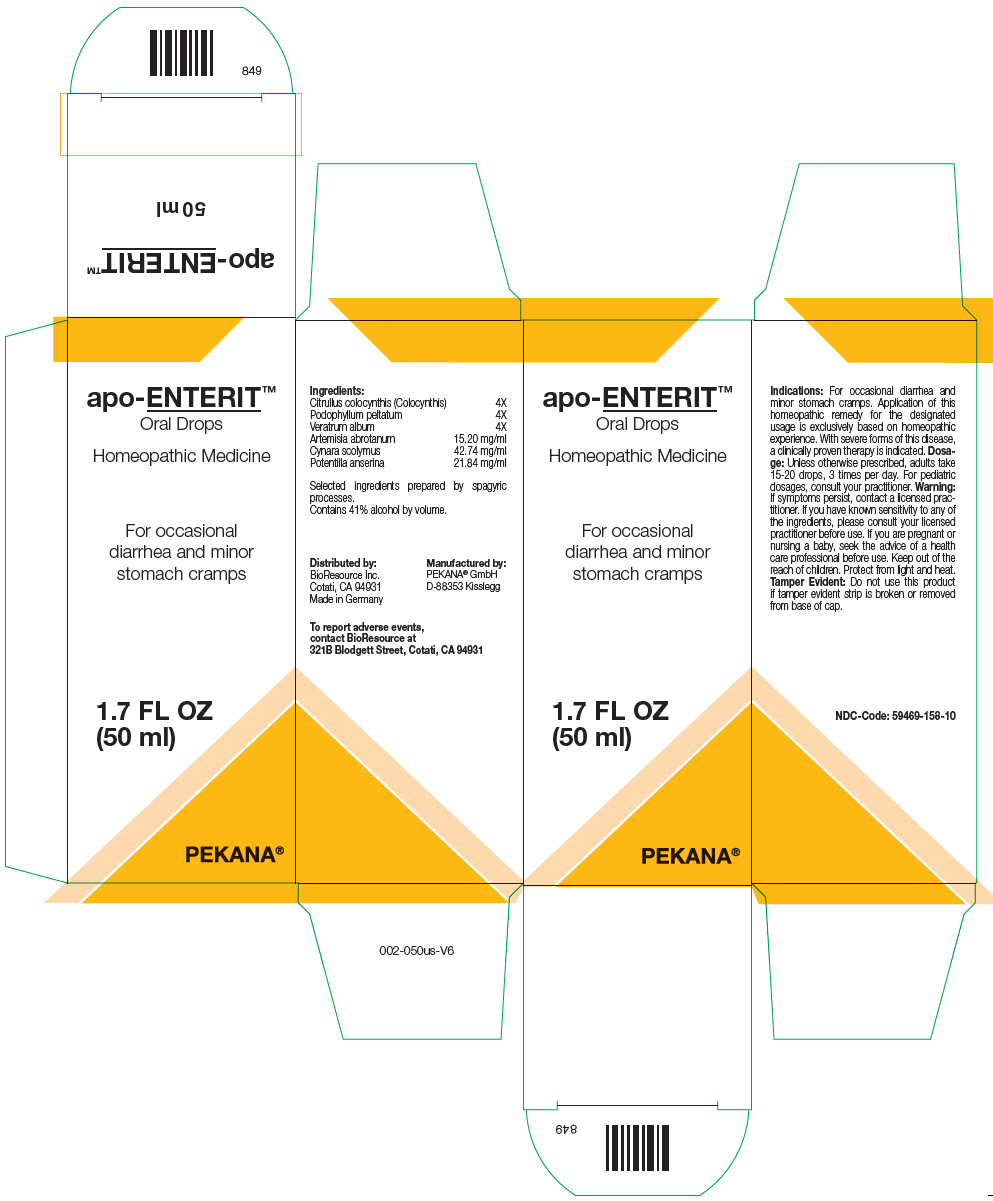
Label: APO-ENTERIT- citrullus colocynthis fruit pulp, podophyllum, veratrum album root, artemisia abrotanum flowering top, cynara scolymus whole, and potentilla anserina flowering top solution/ drops
- NDC Code(s): 59469-158-10
- Packager: PEKANA Naturheilmittel GmbH
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 2, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- Indications
- Dosage
- Warning
- QUESTIONS
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 50 ml Bottle Box
-
INGREDIENTS AND APPEARANCE
APO-ENTERIT
citrullus colocynthis fruit pulp, podophyllum, veratrum album root, artemisia abrotanum flowering top, cynara scolymus whole, and potentilla anserina flowering top solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59469-158 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Citrullus colocynthis Fruit Pulp (UNII: 23H32AOH17) (Citrullus colocynthis Fruit Pulp - UNII:23H32AOH17) Citrullus colocynthis Fruit Pulp 4 [hp_X] in 50 mL Podophyllum (UNII: 2S713A4VP3) (Podophyllum - UNII:2S713A4VP3) Podophyllum 4 [hp_X] in 50 mL Veratrum album Root (UNII: QNS6W5US1Z) (Veratrum Album Root - UNII:QNS6W5US1Z) Veratrum album Root 4 [hp_X] in 50 mL Artemisia abrotanum Flowering Top (UNII: QG07G580U0) (Artemisia abrotanum Flowering Top - UNII:QG07G580U0) Artemisia abrotanum Flowering Top 760 mg in 50 mL Cynara scolymus Whole (UNII: 9N3437ZUU0) (Cynara Scolymus Whole - UNII:9N3437ZUU0) Cynara scolymus Whole 2137 mg in 50 mL Potentilla anserina Flowering Top (UNII: H00Y243SK0) (Potentilla anserina Flowering Top - UNII:H00Y243SK0) Potentilla anserina Flowering Top 1092 mg in 50 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Alcohol (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59469-158-10 1 in 1 BOX 04/12/2008 1 50 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED HOMEOPATHIC 04/12/2008 Labeler - PEKANA Naturheilmittel GmbH (320344542) Establishment Name Address ID/FEI Business Operations PEKANA Naturheilmittel GmbH 320344542 MANUFACTURE(59469-158)