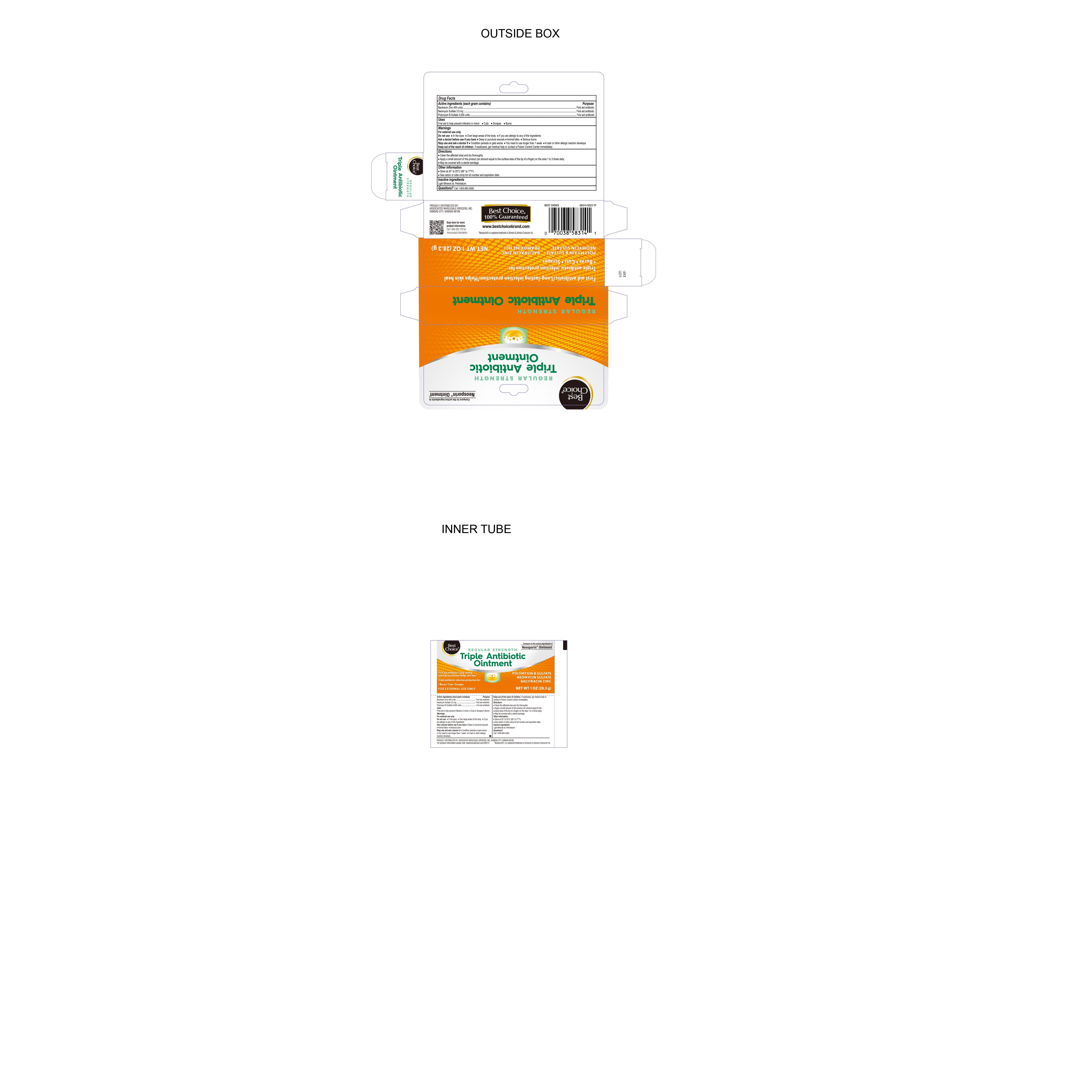
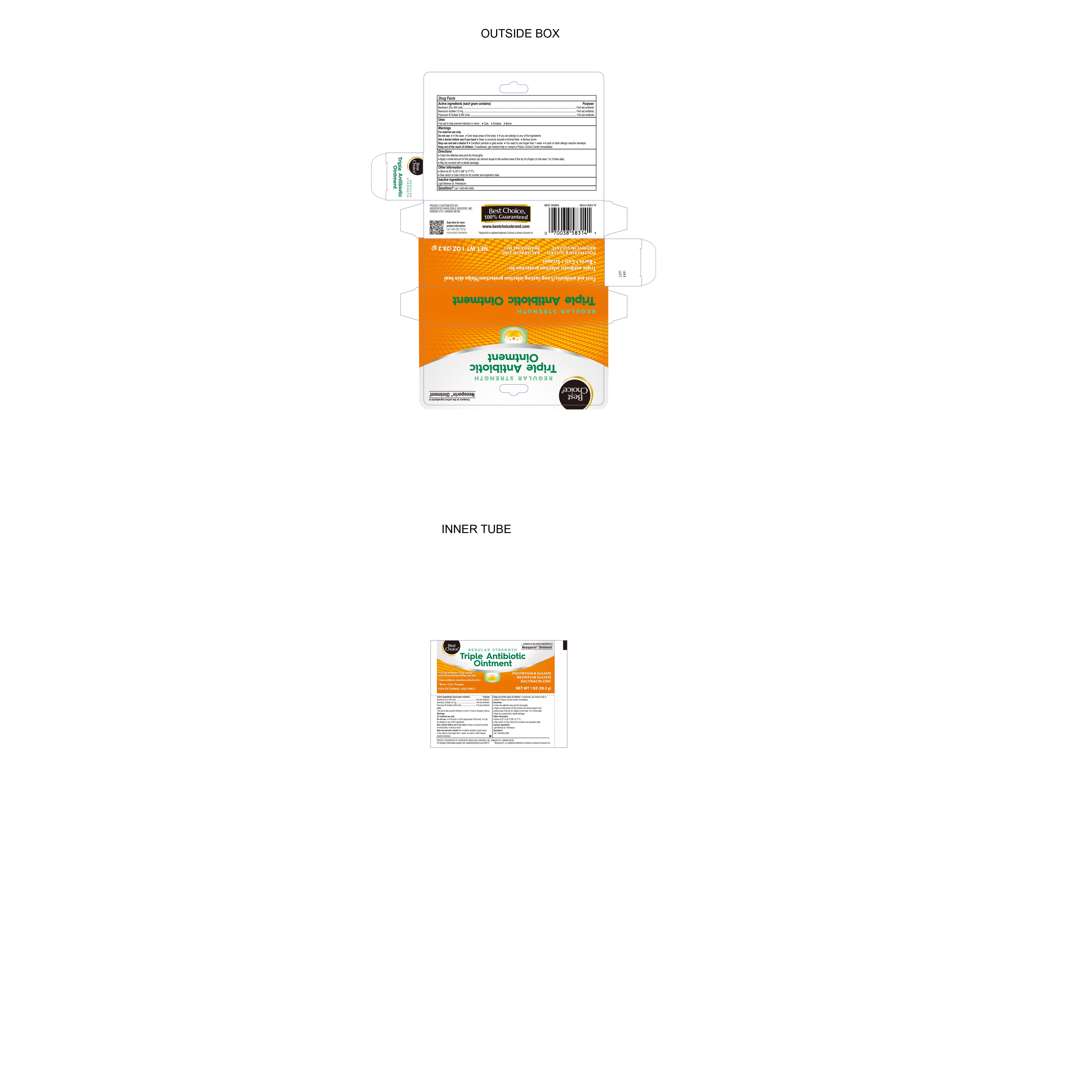
Label: TRIPLE ANTIBIOTIC- bacitracin zinc, neomycin sulfate , polymyxin b sulfate ointment
- NDC Code(s): 69396-131-01
- Packager: TRIFECTA PHARMACEUTICALS USA LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 30, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (each gram contains)
- Purpose
- Keep out of reach of Children
- Uses
- Warnings
- Directions
- Stop Use and Ask Doctor
- Ask Doctor Before Use if you have
- Do Not Use
- INACTIVE INGREDIENT
- Other information
- QUESTIONS
- Distributed By
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TRIPLE ANTIBIOTIC
bacitracin zinc, neomycin sulfate , polymyxin b sulfate ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69396-131 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [USP'U] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [USP'U] in 1 g Inactive Ingredients Ingredient Name Strength LIGHT MINERAL OIL (UNII: N6K5787QVP) PETROLATUM (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69396-131-01 1 in 1 BOX 12/08/2023 1 28.3 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 12/08/2023 Labeler - TRIFECTA PHARMACEUTICALS USA LLC (079424163)