Label: SENNOSIDES tablet, film coated
- NDC Code(s): 51316-112-02, 51316-112-03, 51316-112-05
- Packager: CVS Pharmacy, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- PURPOSE
- Uses
-
WARNINGS
Ask a doctor before use if you have
stomach pain
nausea
vomiting
noticed a sudden change in bowel habits that continues over a period of 2 weeks -
Directions
take preferably at bedtime or as directed by a doctor
Age starting dosage maximum dosage adults and children
12 years of age and over2 tablets once a day 4 tablets twice a day children 6 to under 12 years 1 tablet once a day 2 tablets twice a day children 2 to under 6 years 1/2 tablet once a day 1 tablet twice a day children under 2 years ask a doctor ask a doctor - Other information
- INACTIVE INGREDIENT
- QUESTIONS
-
SPL UNCLASSIFIED SECTION
Distributed by:
CVS Pharmacy, Inc.
One CVS Drive
Woonsocket, RI 02895
© 2022 CVS/pharmacy
CVS.com® 1-800-SHOP CVSTAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
KEEP THIS CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION
* This product is not manufactured or distributed by Avrio Health L.P., owner of the registered trademark Senokot®.
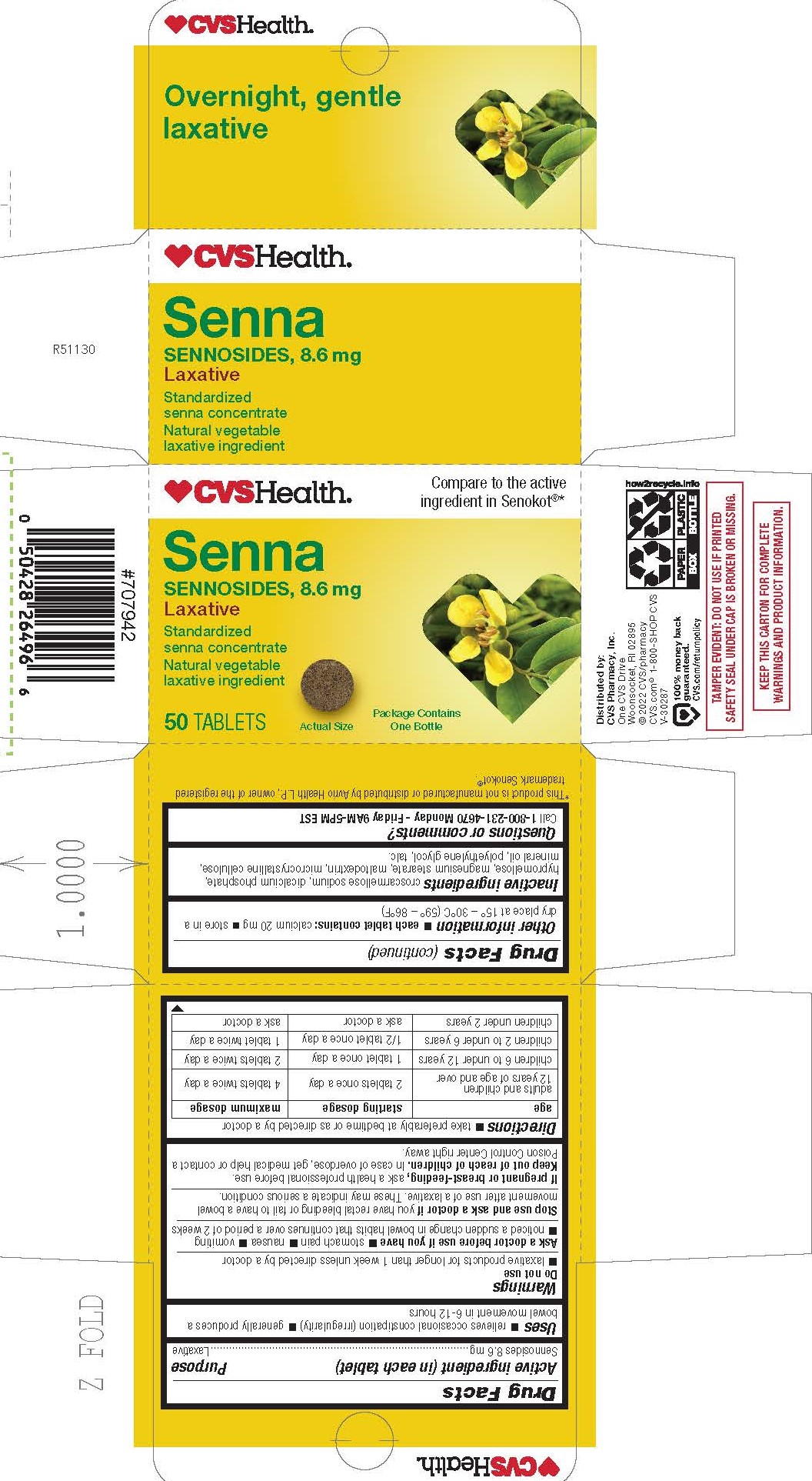
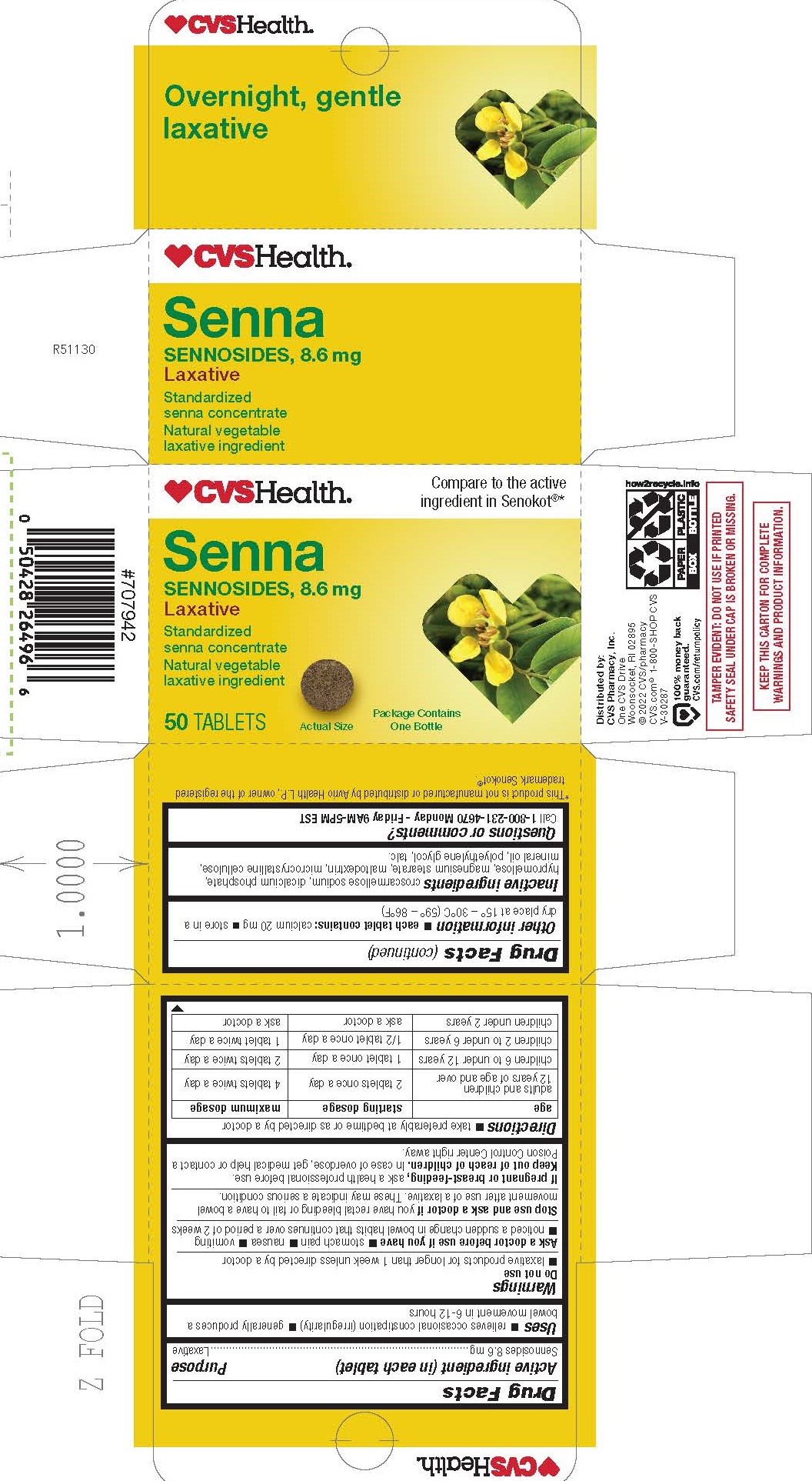
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SENNOSIDES
sennosides tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51316-112 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SENNOSIDES (UNII: 3FYP5M0IJX) (SENNOSIDES - UNII:3FYP5M0IJX) SENNOSIDES 8.6 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSES (UNII: 3NXW29V3WO) LIGHT MINERAL OIL (UNII: N6K5787QVP) CALCIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: O7TSZ97GEP) MALTODEXTRIN (UNII: 7CVR7L4A2D) TALC (UNII: 7SEV7J4R1U) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color brown Score no score Shape ROUND Size 9mm Flavor Imprint Code W2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51316-112-02 250 in 1 BOTTLE; Type 0: Not a Combination Product 03/23/2023 2 NDC:51316-112-03 1 in 1 CARTON 07/26/2023 2 50 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:51316-112-05 500 in 1 BOTTLE; Type 0: Not a Combination Product 12/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M007 03/23/2023 Labeler - CVS Pharmacy, Inc (062312574)