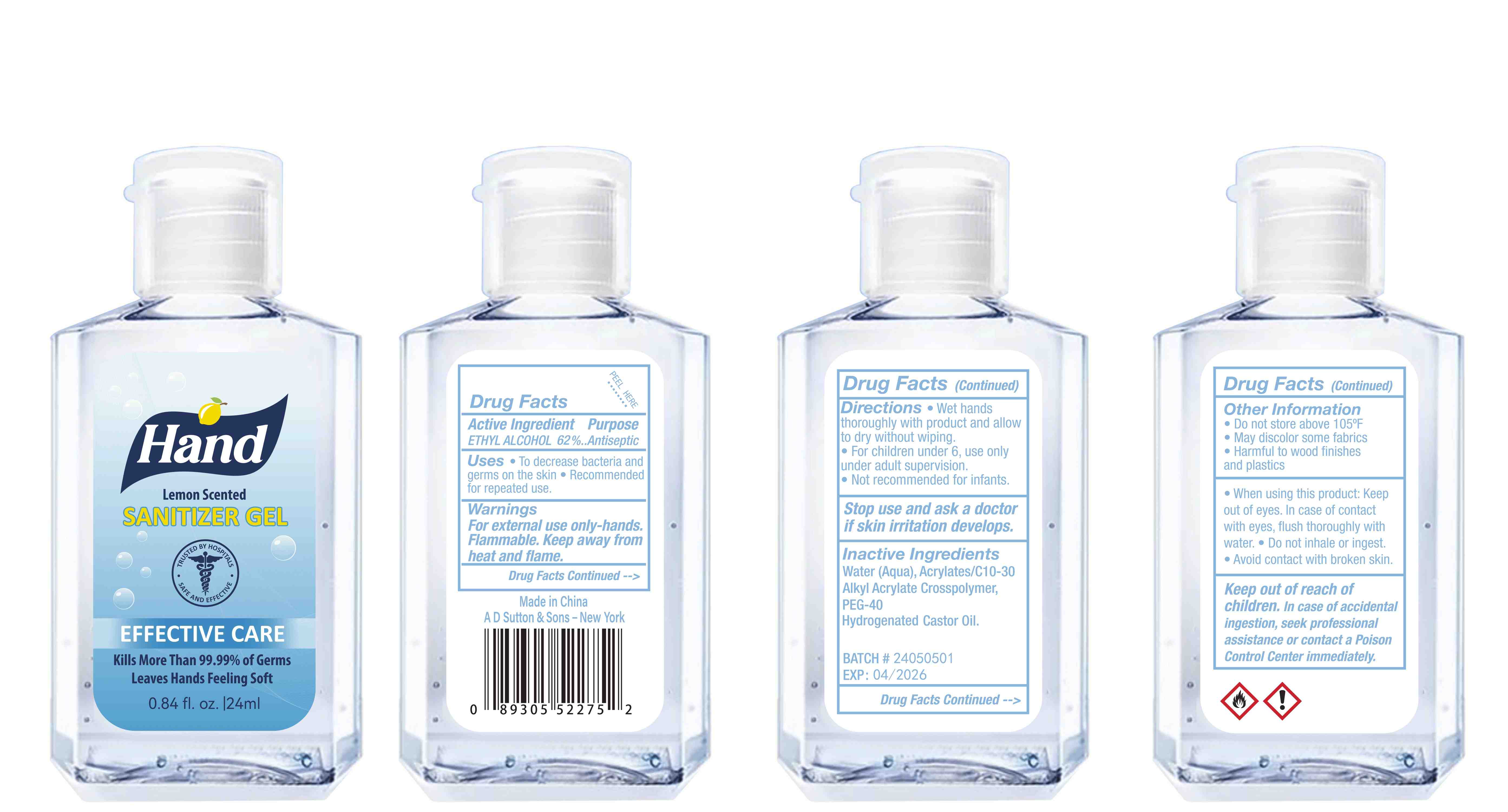
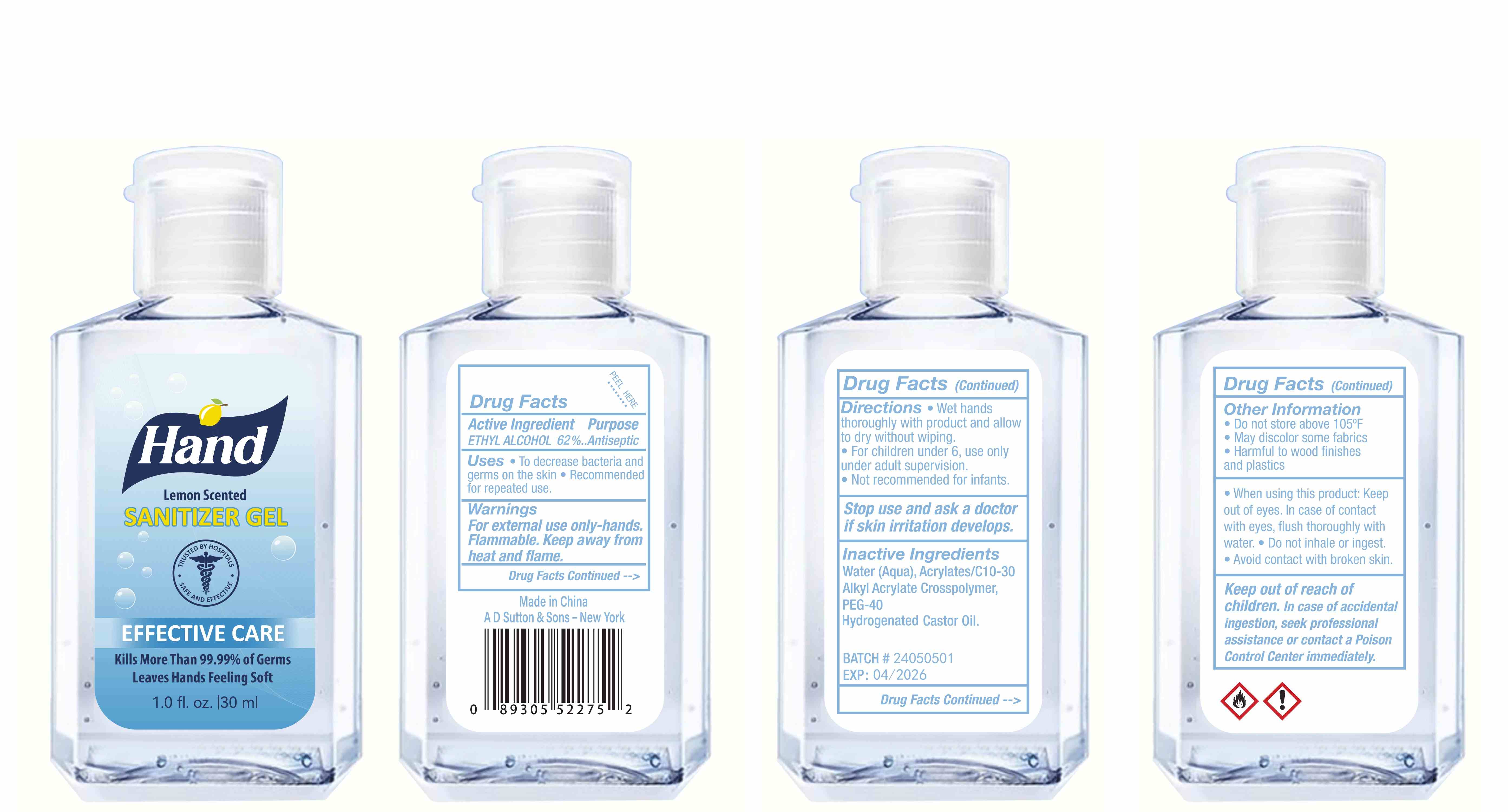
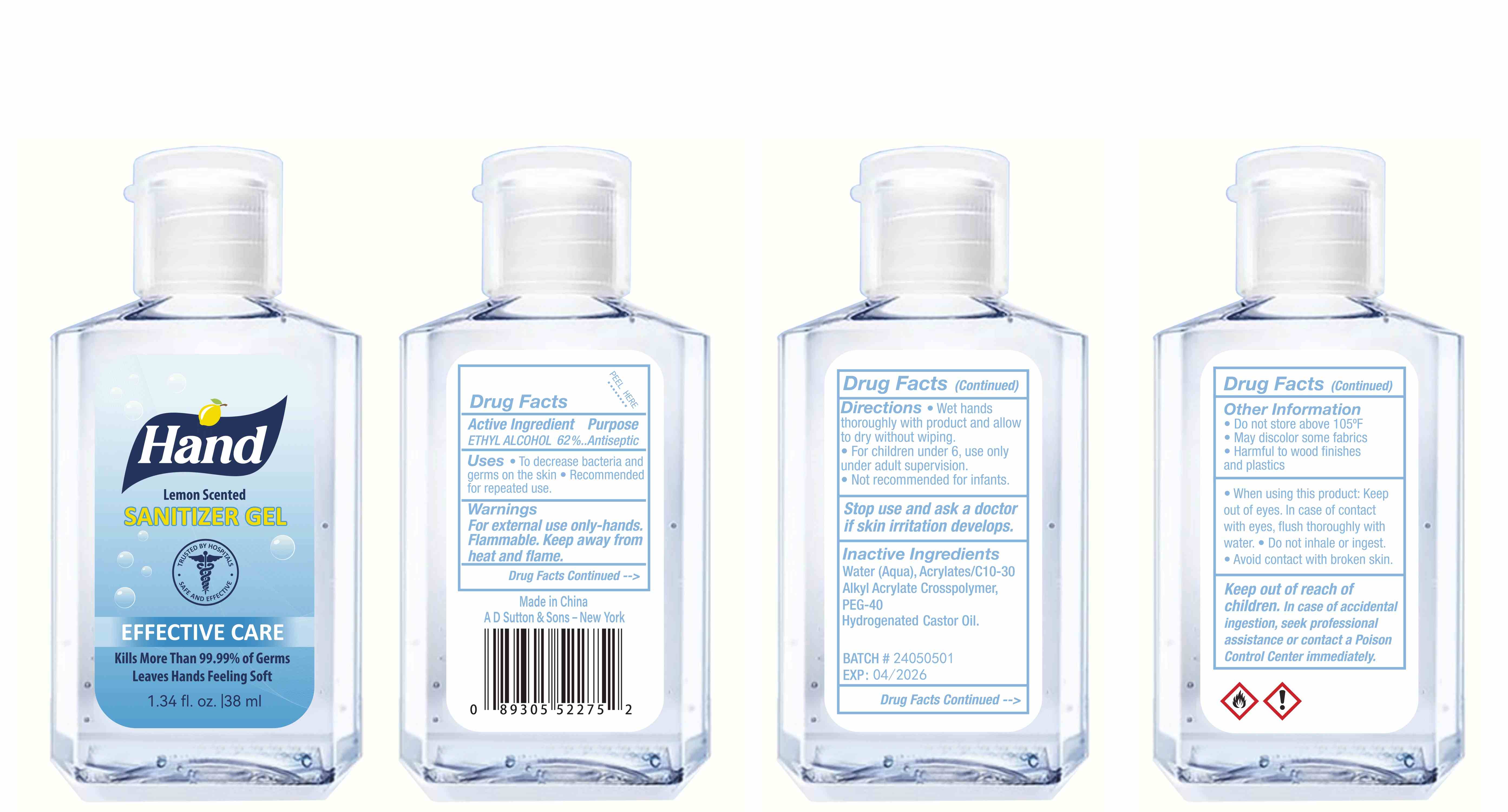
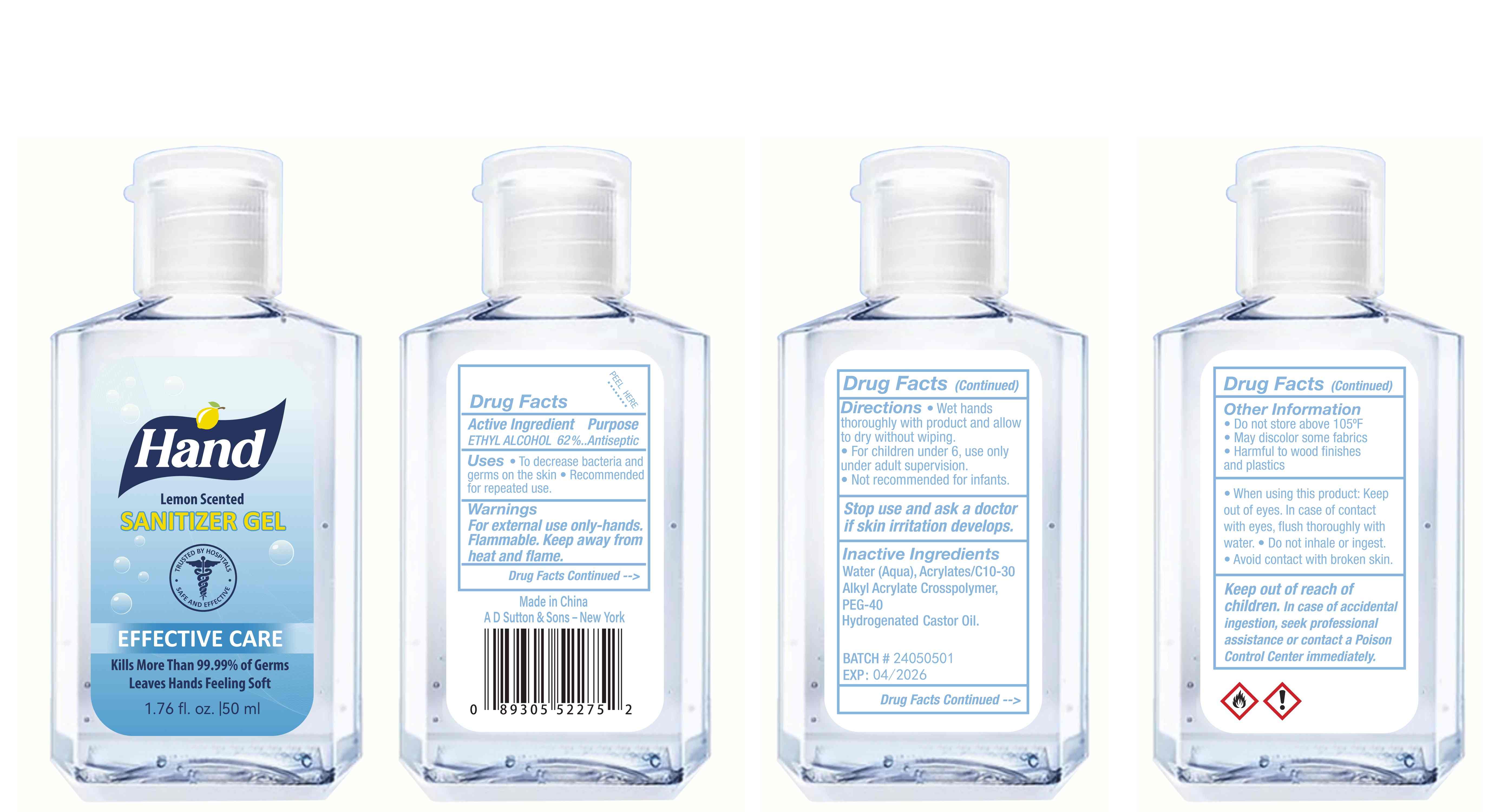
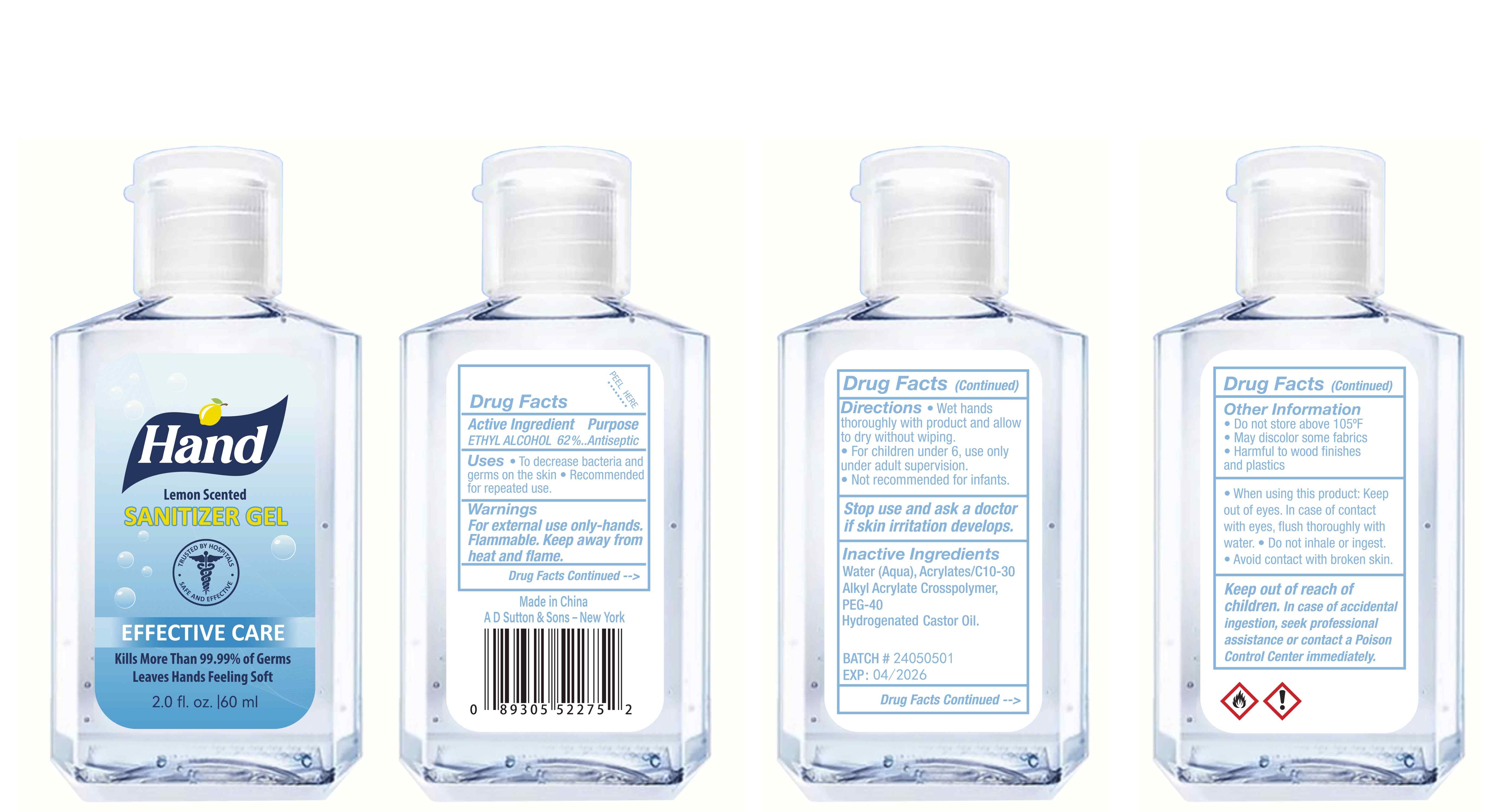
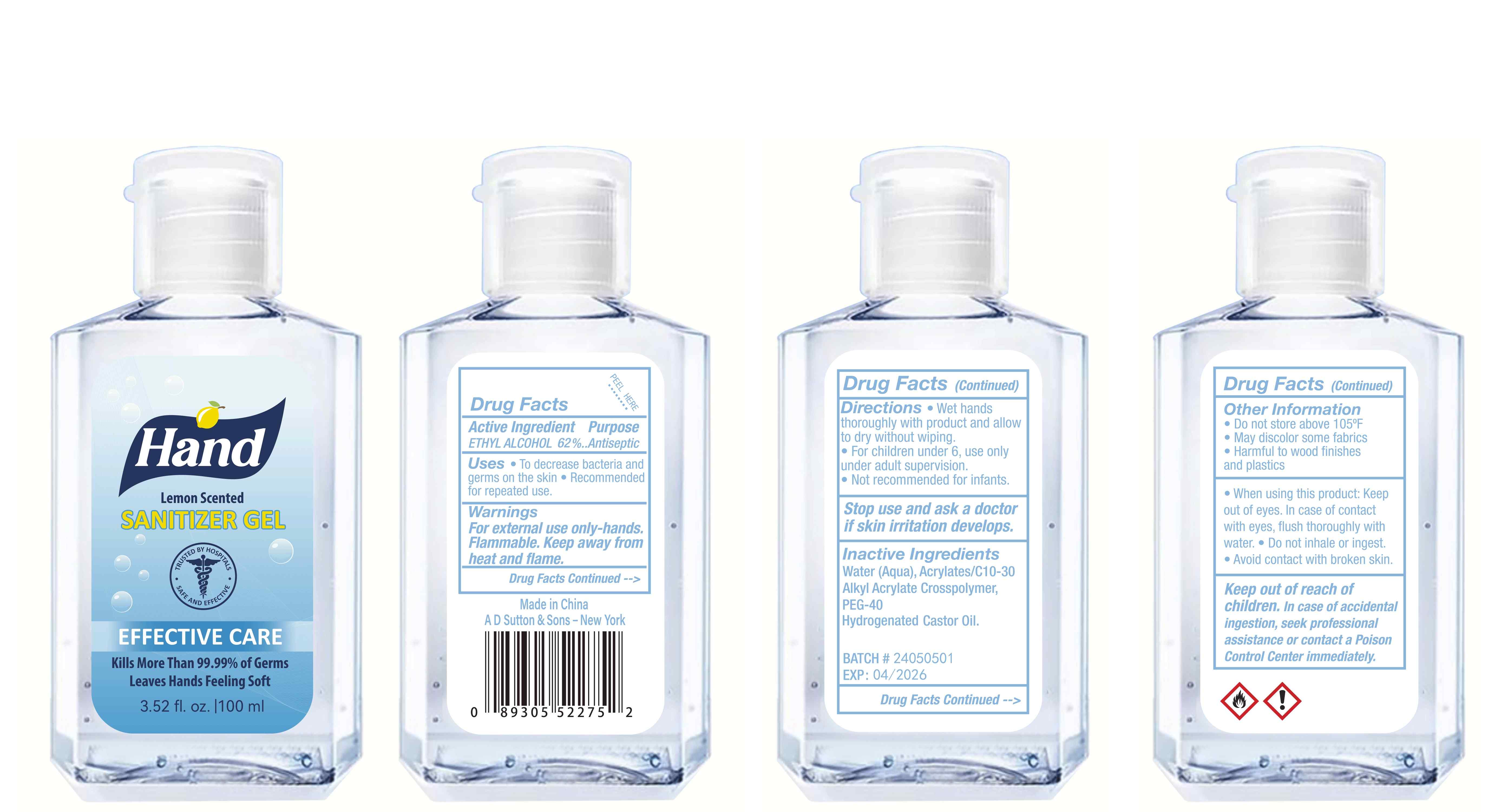
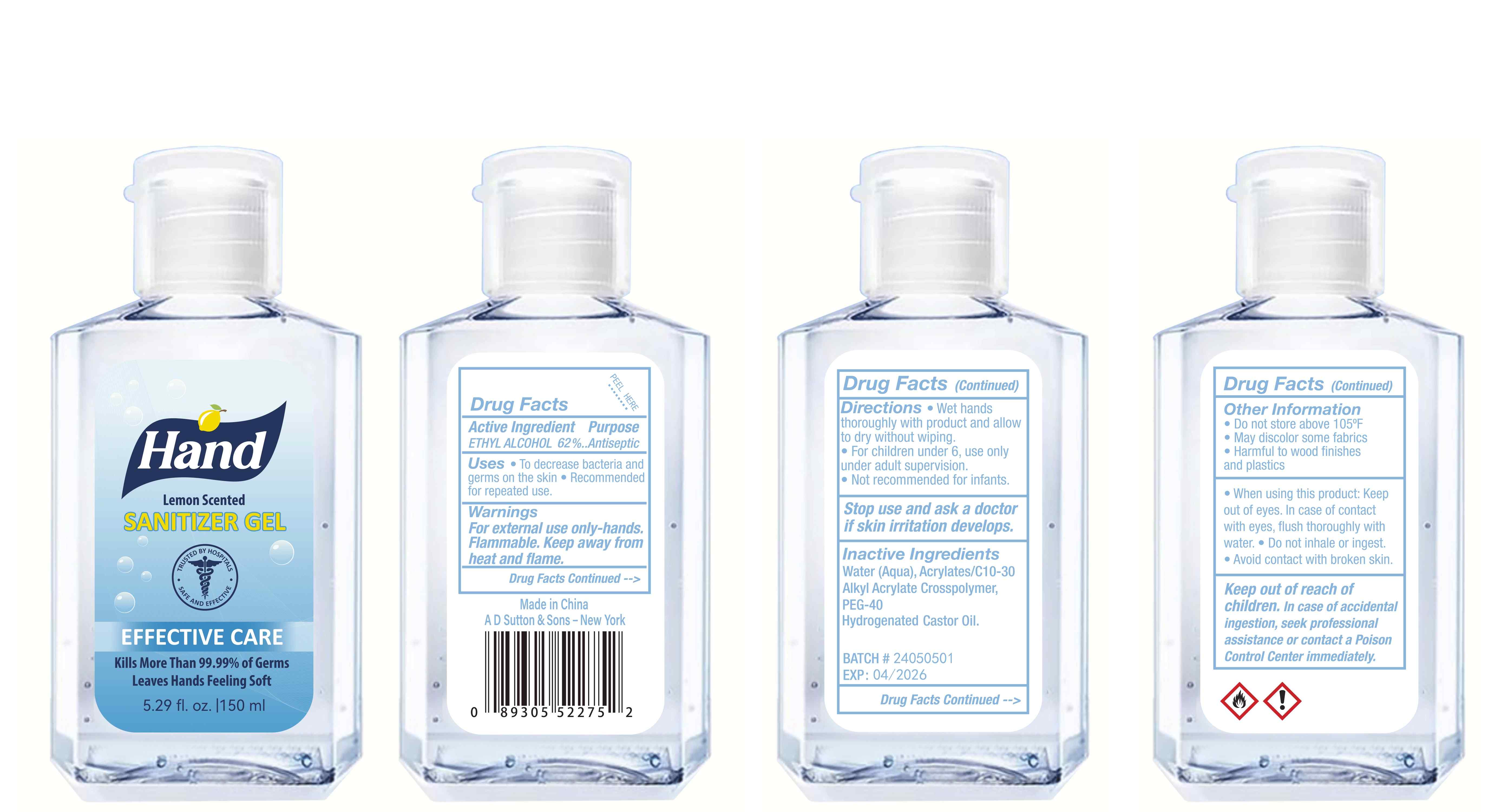
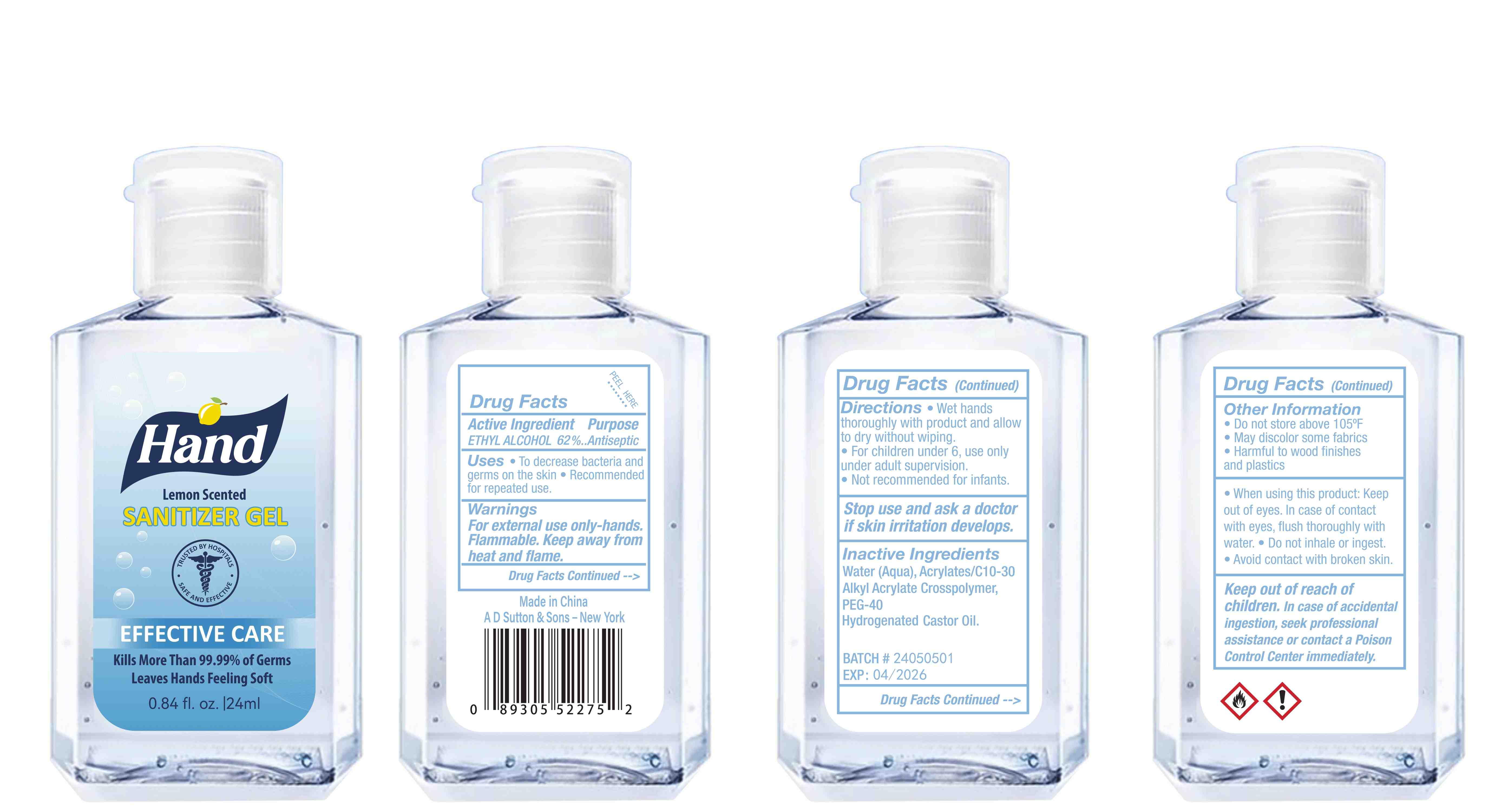
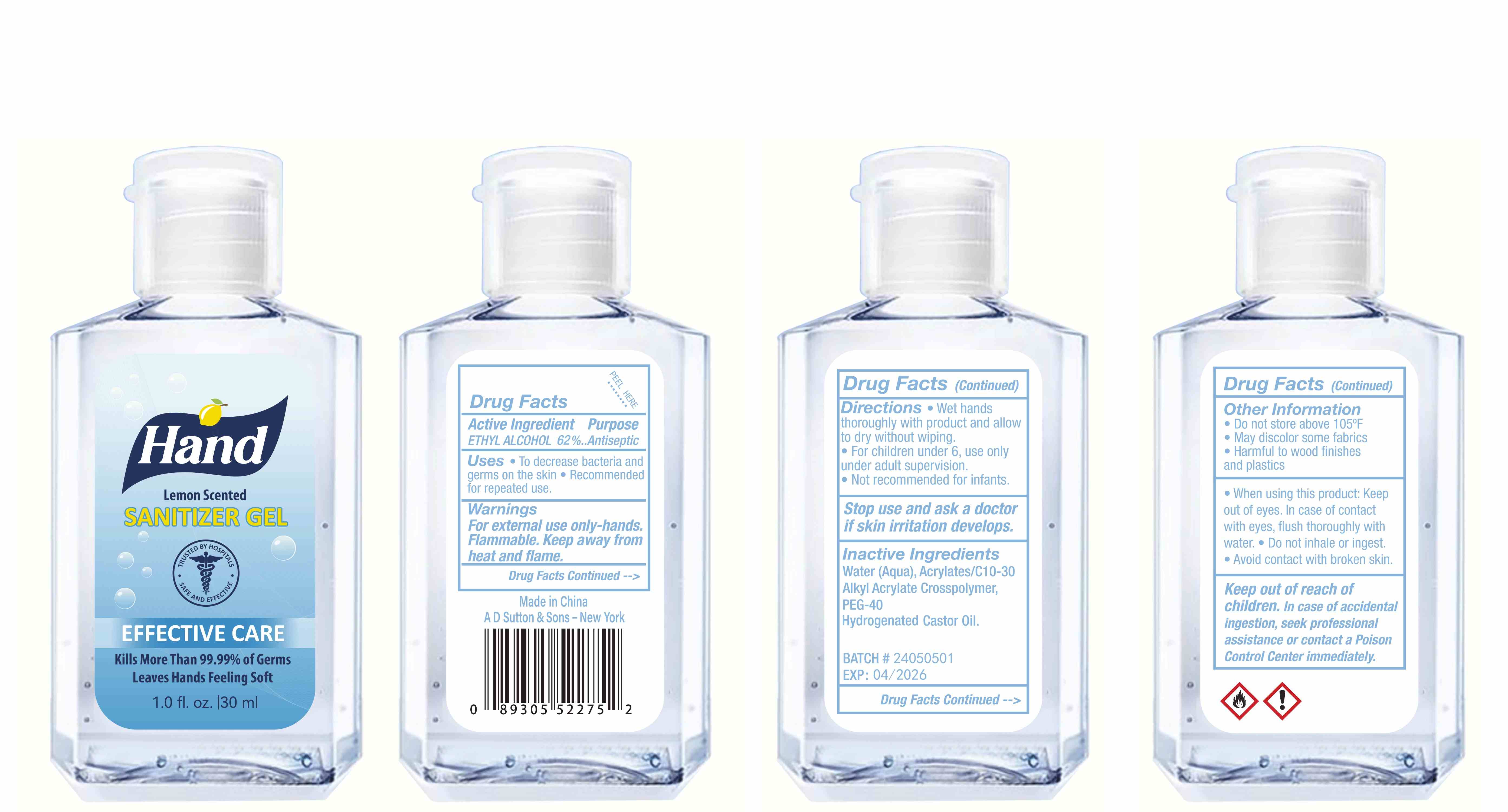
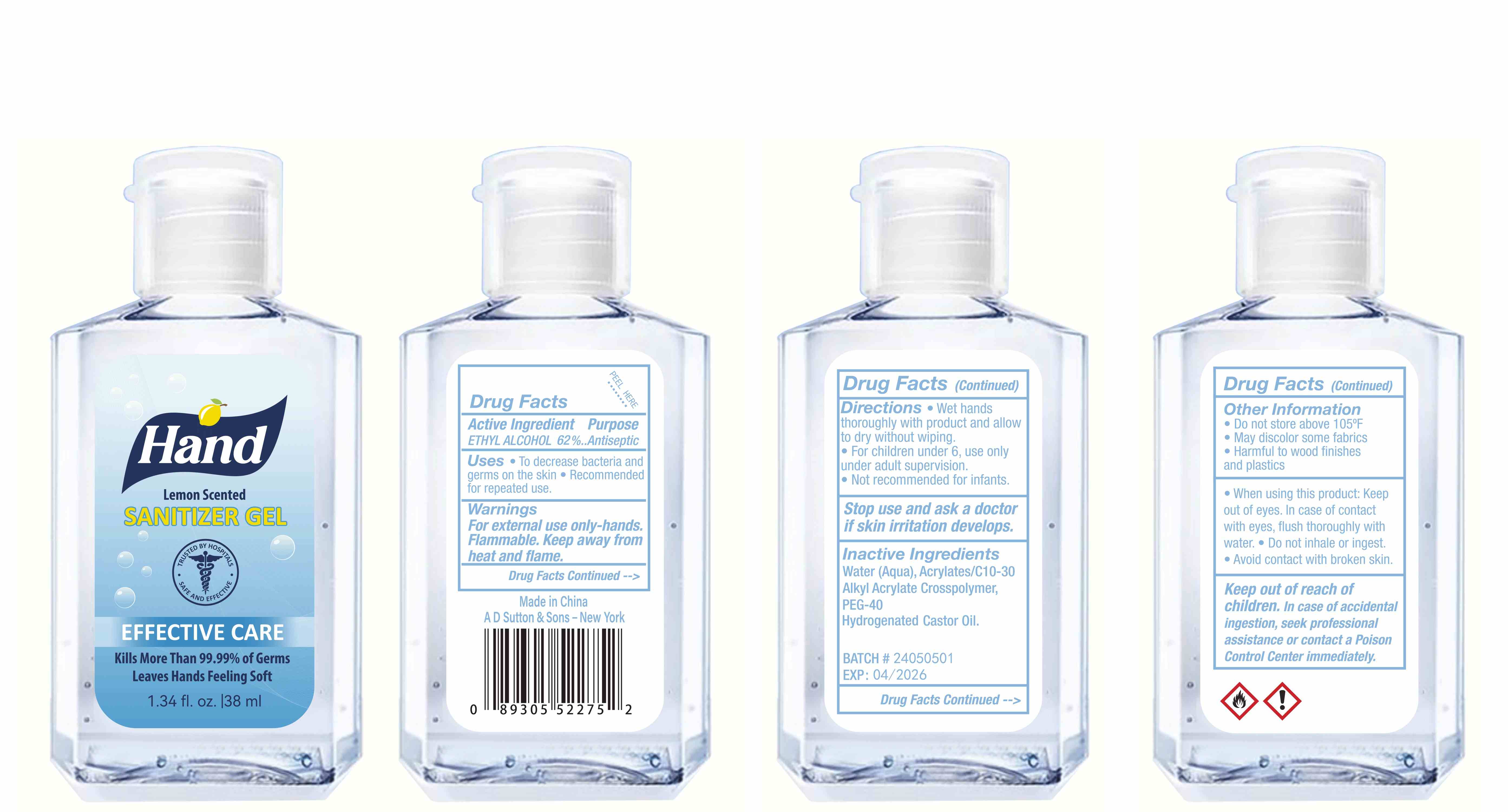
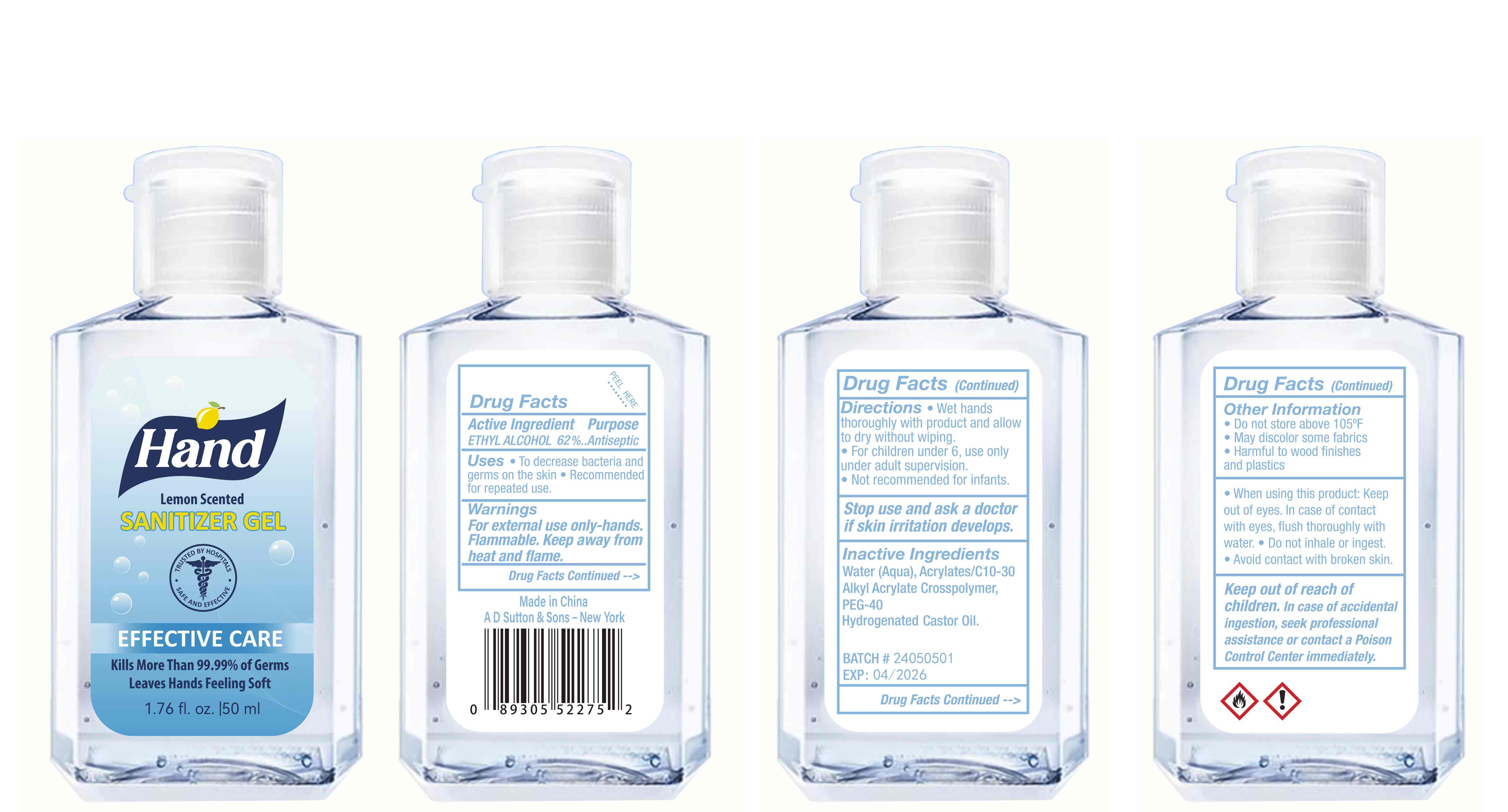
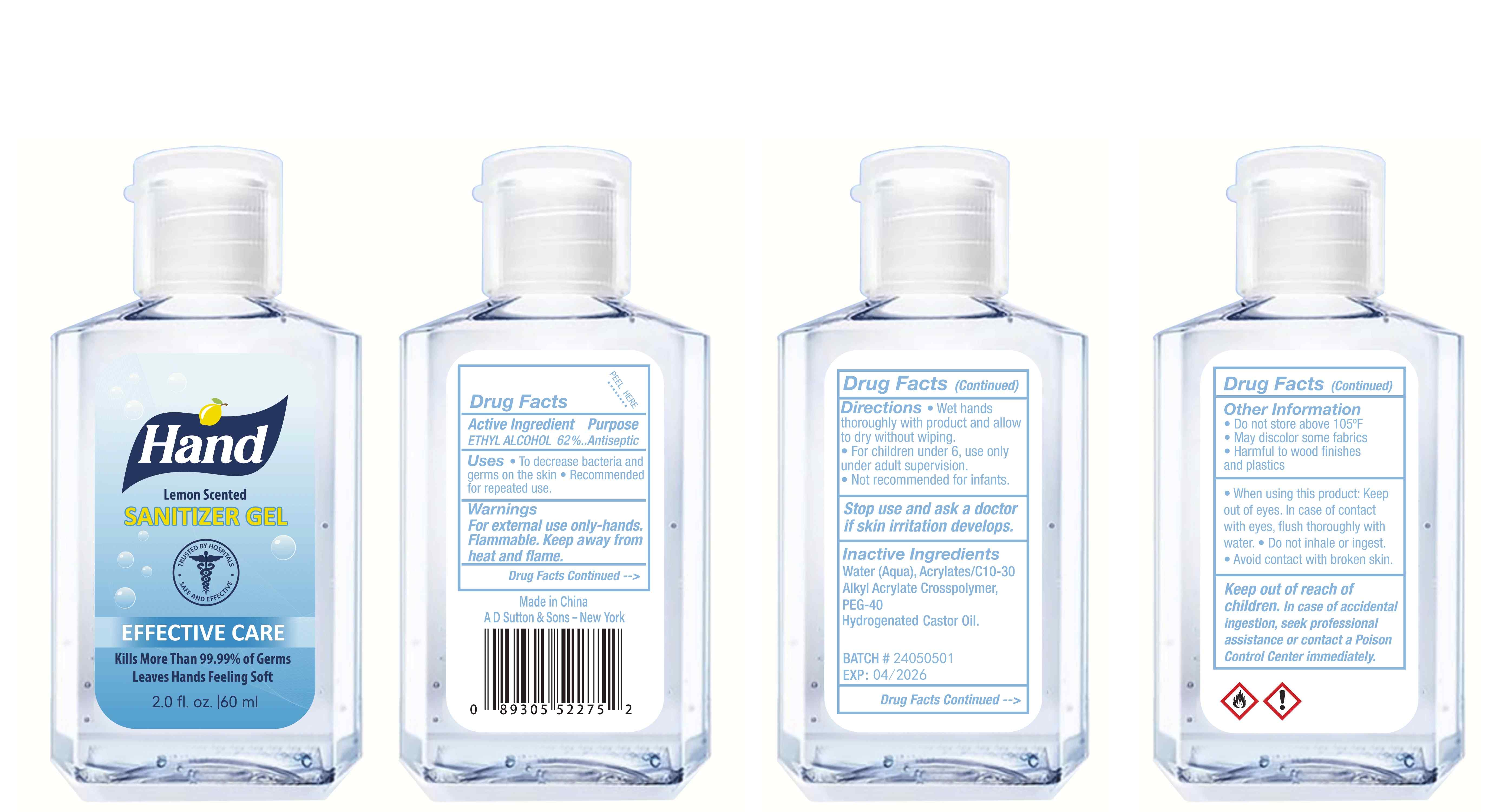
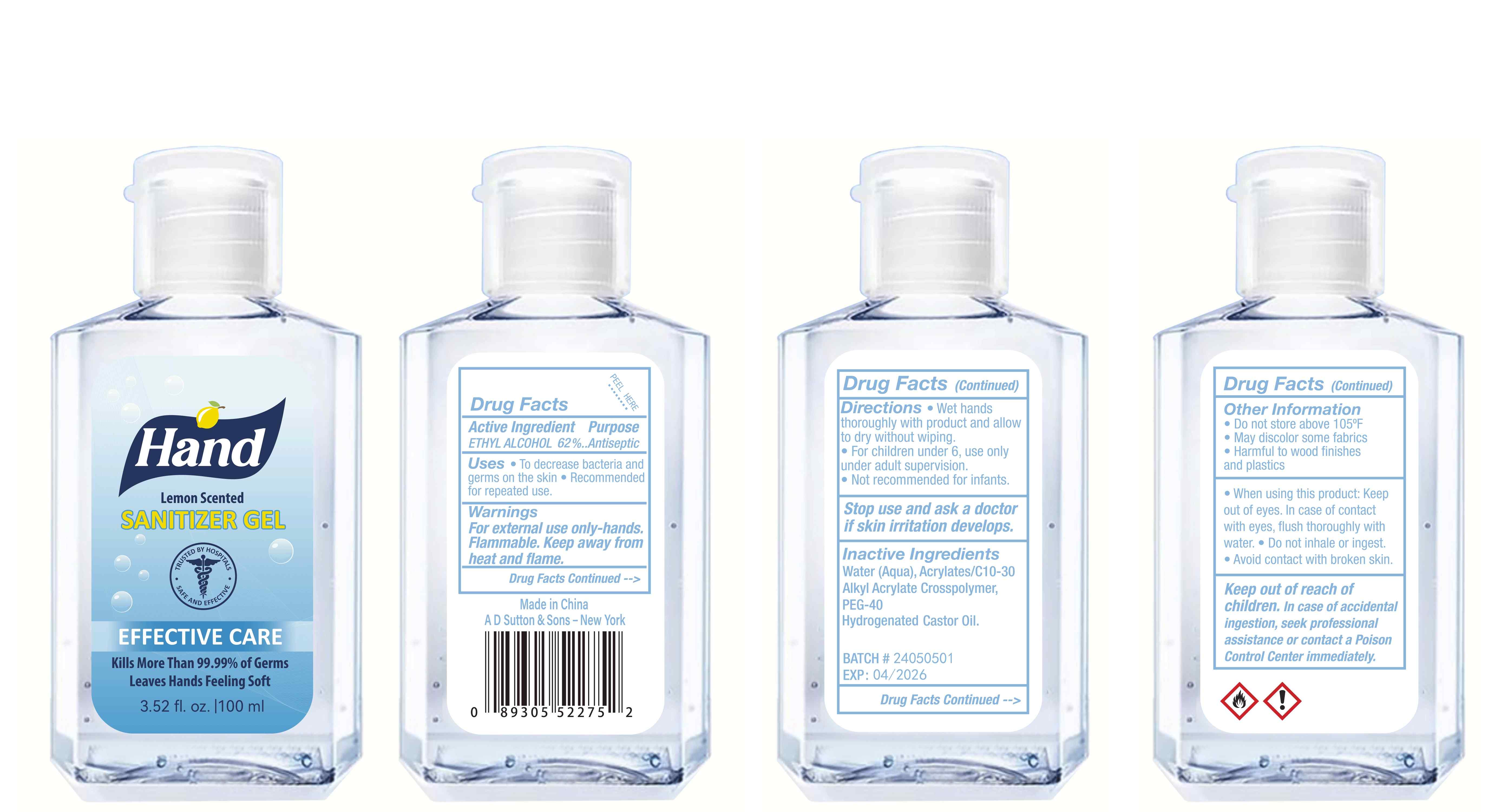
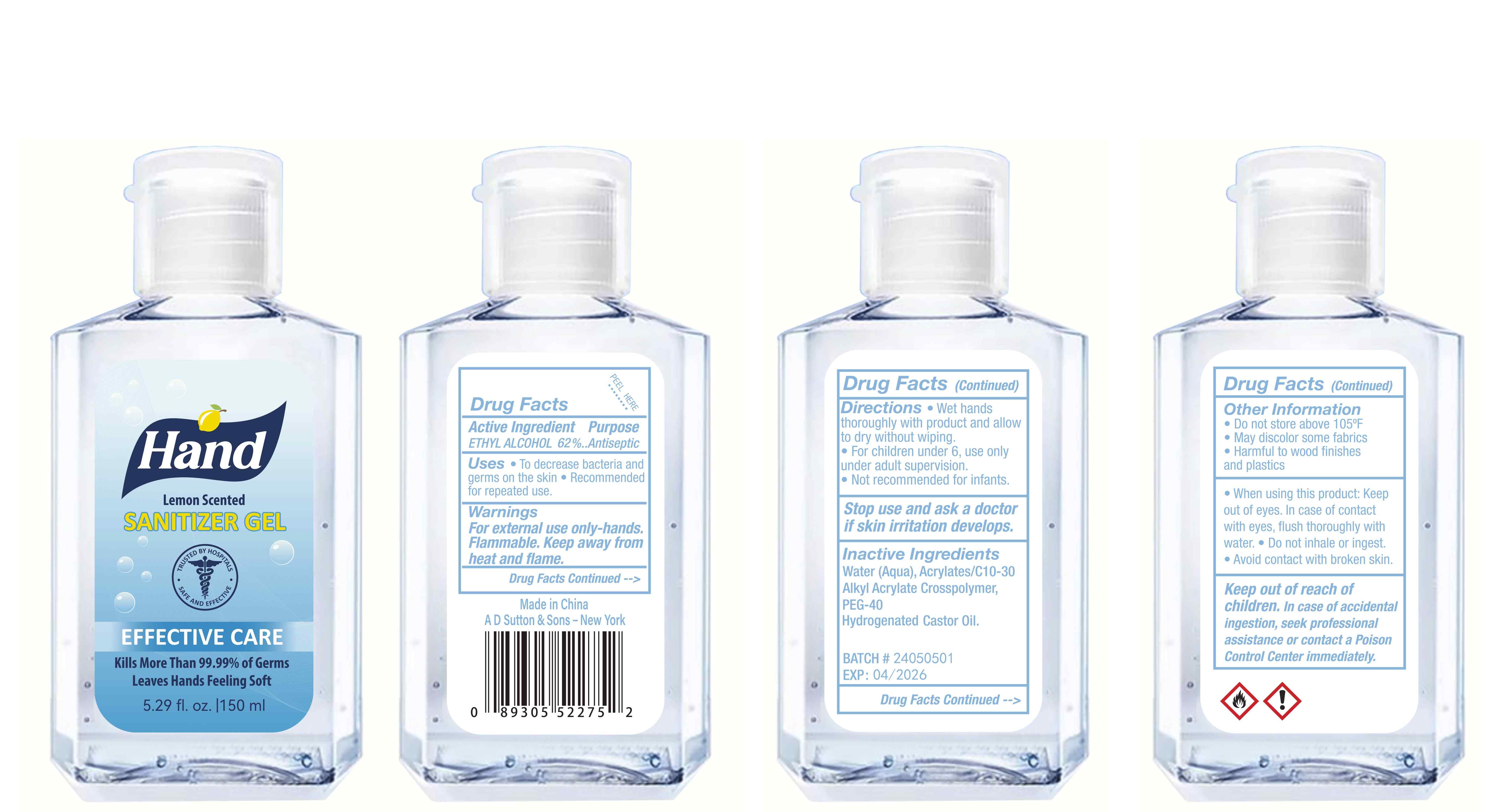
Label: HAND SANITIZER gel
-
NDC Code(s):
77975-015-01,
77975-015-02,
77975-015-03,
77975-015-04, view more77975-015-05, 77975-015-06, 77975-015-07, 77975-015-08, 77975-015-09, 77975-015-10
- Packager: Guangzhou Tingcai Cosmetic Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 6, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient(s)
- Purpose
- Use
- Warnings
- Do not use
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
HAND SANITIZER
hand sanitizer gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:77975-015 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ACRYLATES/C10-30 ALKYL ACRYLATE CROSSPOLYMER (60000 MPA.S) (UNII: 8Z5ZAL5H3V) PEG-40 CASTOR OIL (UNII: 4ERD2076EF) LEMON PEEL (UNII: 72O054U628) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:77975-015-06 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 2 NDC:77975-015-09 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 3 NDC:77975-015-05 60 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 4 NDC:77975-015-04 50 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 5 NDC:77975-015-10 300 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 6 NDC:77975-015-03 38 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 7 NDC:77975-015-07 150 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 8 NDC:77975-015-01 24 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 9 NDC:77975-015-08 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 10 NDC:77975-015-02 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/06/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 03/06/2024 Labeler - Guangzhou Tingcai Cosmetic Co., Ltd. (529562889) Establishment Name Address ID/FEI Business Operations Guangzhou Tingcai Cosmetic Co., Ltd. 529562889 manufacture(77975-015)