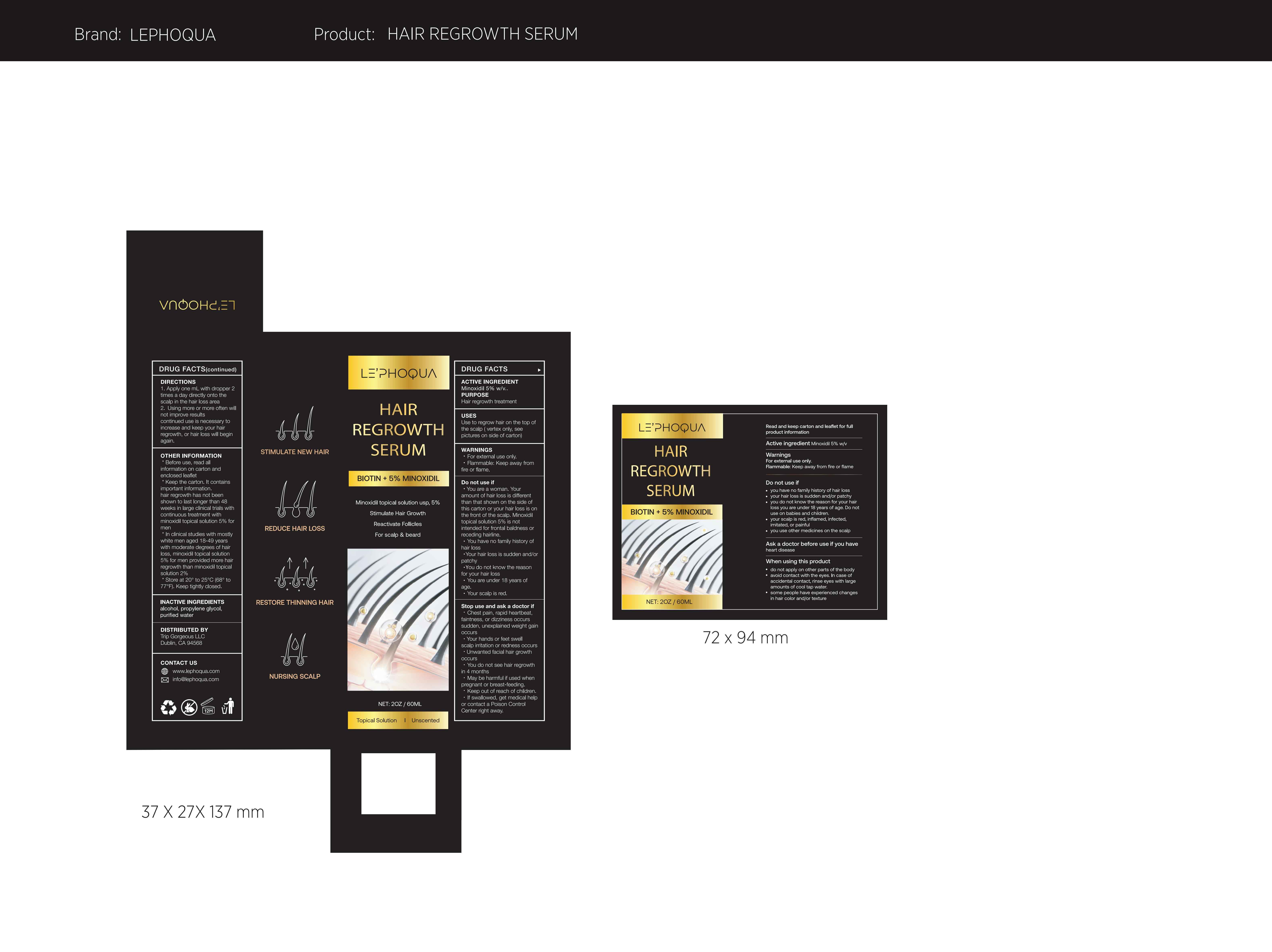
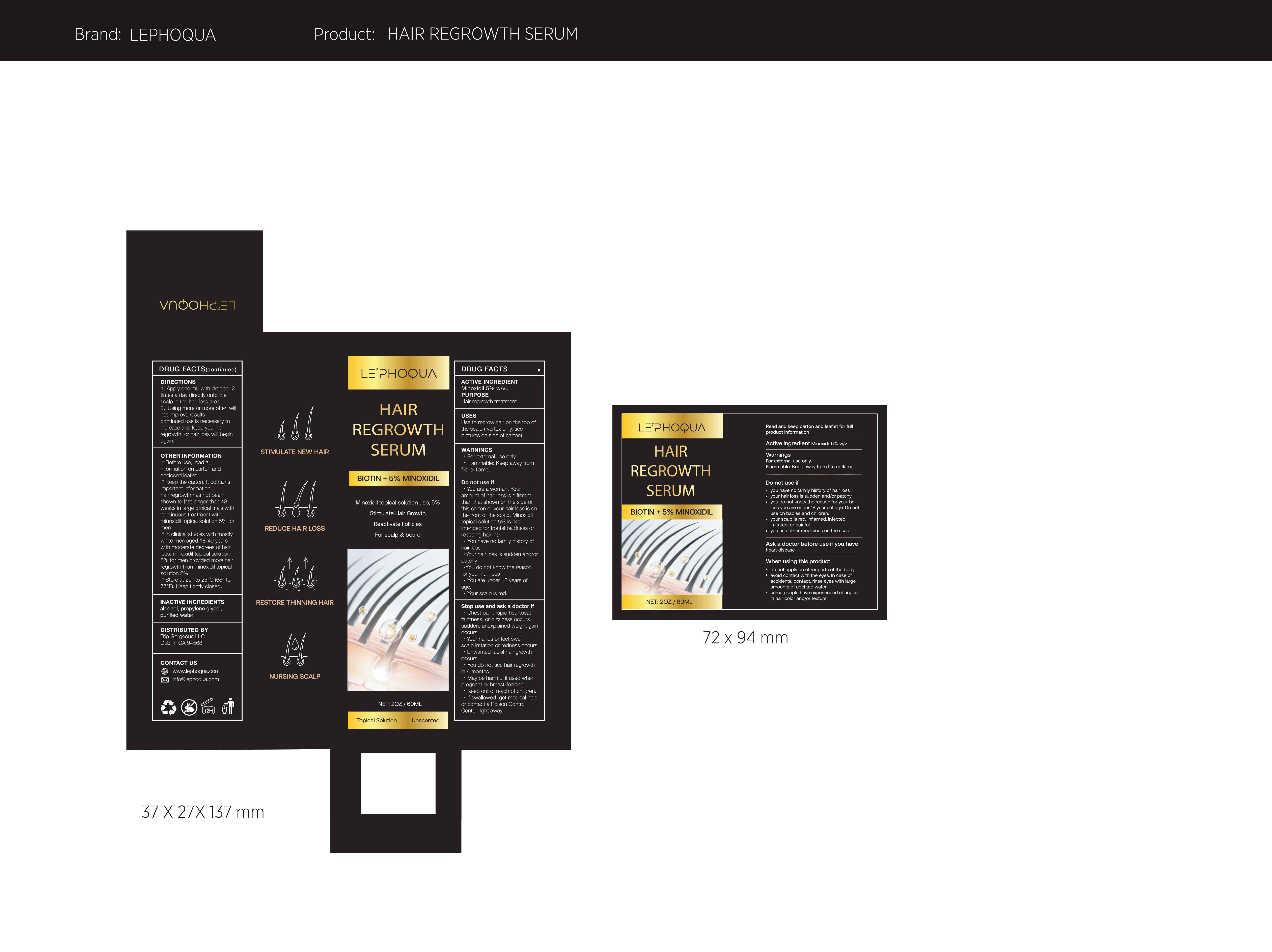
Label: LE PHOQUA HAIR REGROWTH SERUM BIOTIN 5% MINOXIDIL- hair regrowth serum liquid
- NDC Code(s): 83565-007-01
- Packager: Stellans Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 25, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
-
Do not use
·You are a woman. Your amount of hair loss is different than that shown on the side of this carton or your hair loss is on the front of the scalp. Minoxidil topical solution 5% is not intended for frontal baldness or receding hairline.
·You have no family history of hair loss
·Your hair loss is sudden and/or patchy
·You do not know the reason for your hair loss
·You are under 18 years of age.
·Your scalp is red. -
Stop Use
·Chest pain, rapid heartbeat, faintness , or dizziness occurs
sudden, unexplained weight gain occurs
·Your hands or feet swell scalp irritation or redness occurs
·Unwanted fa cial hair growth occurs
·You do not see hair regrowth in 4 months
·May be harmful if used when pregnant or breast-feeding.
·Keep out of reach of children,
·If swallowed, get medical help or contact a Poison Control Center right away. - Keep Oot Of Reach Of Children
- Directions
-
Other information
*Before use, read all information on carton and
enclosed leaflet
*Keep the carton. It contains important information.
hair regrowth has not been shown to last longer than 48 weeks in large clinical trials with continuous treatment with minoxidill topical solution 5% for men
*In clinical studies with mostly white men aged 18-49 years with moderate degrees of hair loss, minoxidil topical solution 5% for men provided more hair regrowth than minoxidil topical solution 2%
*Store at 20°to 25℃(68°to 77°F). Keep tightly closed. - Inactive ingredients
- Questions
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LE PHOQUA HAIR REGROWTH SERUM BIOTIN 5% MINOXIDIL
hair regrowth serum liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83565-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 5 g in 100 mL Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ALCOHOL (UNII: 3K9958V90M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83565-007-01 60 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/24/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075357 11/24/2023 Labeler - Stellans Inc. (111157321) Establishment Name Address ID/FEI Business Operations Stellans Inc. 111157321 label(83565-007) , manufacture(83565-007)