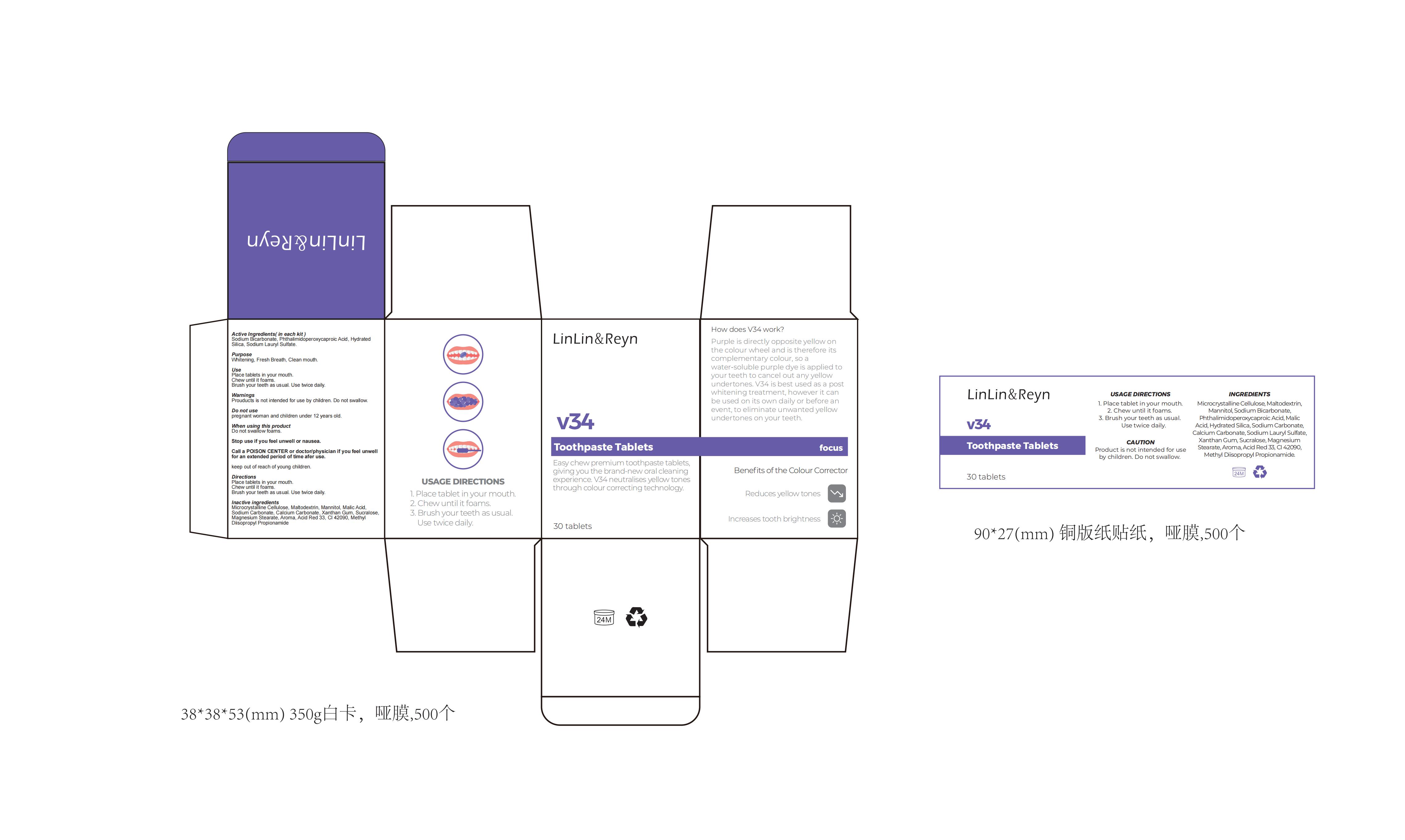
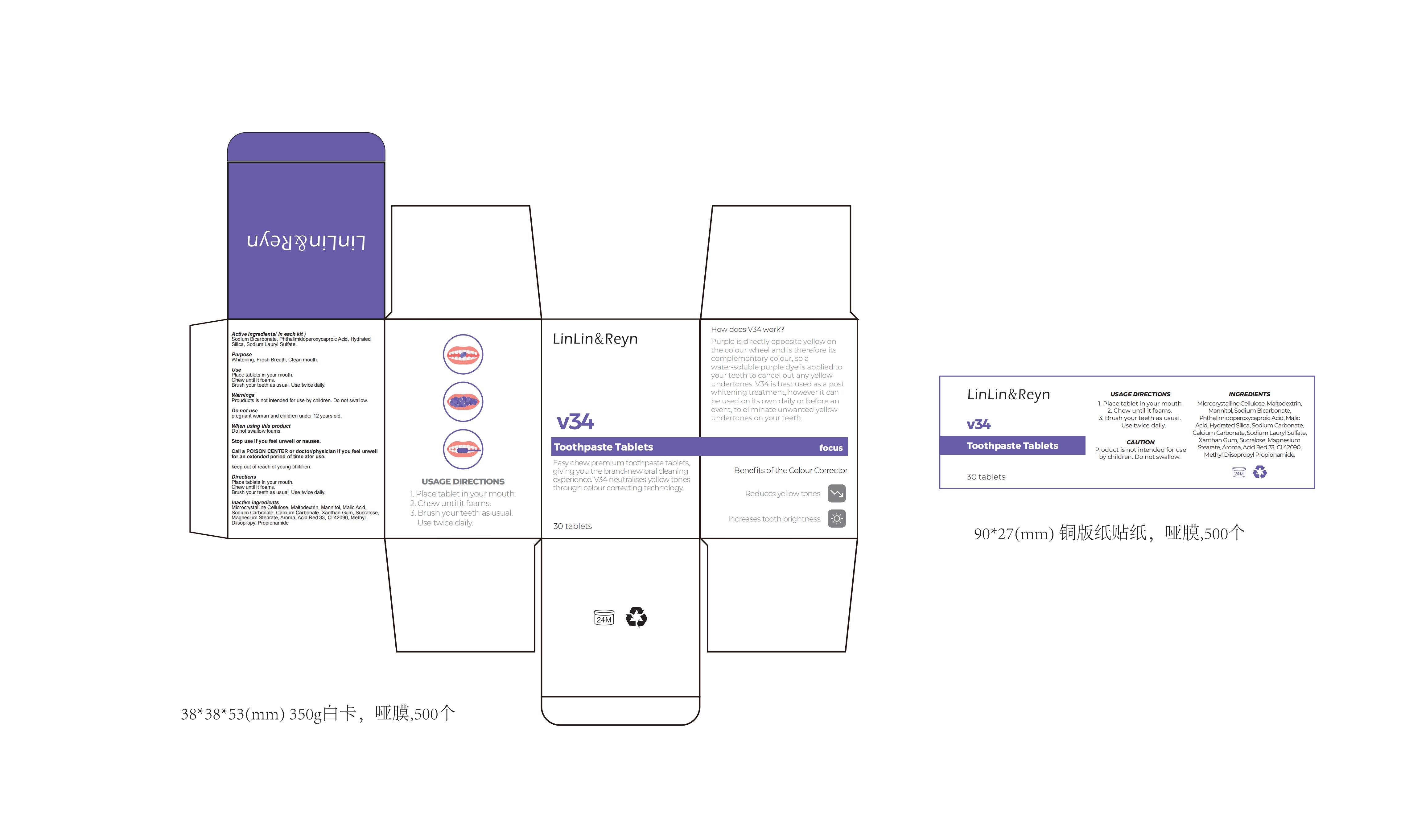
Label: V34 TOOTHPASTE. TABLETS.- v34 toothpaste tablets tablet
- NDC Code(s): 83812-005-01
- Packager: LLRN PERSONAL CARE(SHENZHEN)CO., LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated November 24, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
- When Using
- Stop Use
- Ask Doctor
- Keep Oot Of Reach Of Children
- Directions
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
V34 TOOTHPASTE. TABLETS.
v34 toothpaste tablets tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83812-005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHTHALIMIDOPEROXYCAPROIC ACID (UNII: 5OEJ6FAL6C) (PHTHALIMIDOPEROXYCAPROIC ACID - UNII:5OEJ6FAL6C) PHTHALIMIDOPEROXYCAPROIC ACID 6 g in 100 SODIUM BICARBONATE (UNII: 8MDF5V39QO) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM BICARBONATE 7 g in 100 HYDRATED SILICA (UNII: Y6O7T4G8P9) (HYDRATED SILICA - UNII:Y6O7T4G8P9) HYDRATED SILICA 5 g in 100 Inactive Ingredients Ingredient Name Strength XANTHAN GUM (UNII: TTV12P4NEE) SUCRALOSE (UNII: 96K6UQ3ZD4) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) METHYL DIISOPROPYL PROPIONAMIDE (UNII: 6QOP5A9489) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CALCIUM CARBONATE (UNII: H0G9379FGK) MALTODEXTRIN (UNII: 7CVR7L4A2D) MANNITOL (UNII: 3OWL53L36A) D&C RED NO. 33 (UNII: 9DBA0SBB0L) AROMADENDRIN (UNII: 7YA4640575) SODIUM CARBONATE (UNII: 45P3261C7T) MALIC ACID (UNII: 817L1N4CKP) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color white Score no score Shape ROUND Size 5mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83812-005-01 30 in 1 BOTTLE; Type 0: Not a Combination Product 11/24/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 11/24/2023 Labeler - LLRN PERSONAL CARE(SHENZHEN)CO., LTD (419890311) Establishment Name Address ID/FEI Business Operations LLRN PERSONAL CARE(SHENZHEN)CO., LTD 419890311 label(83812-005) , manufacture(83812-005)