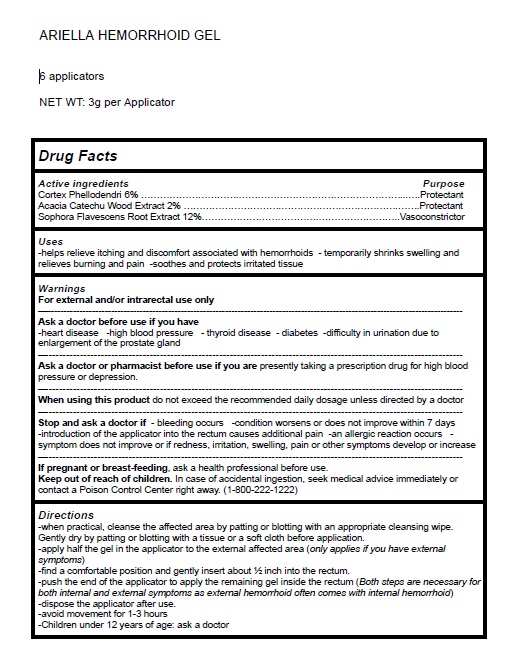
Label: ARIELLA HEMORRHOID GEL- phellodendron chinense whole,senegalia catechu wood,sophora flavescens root gel
- NDC Code(s): 83364-002-01
- Packager: YITONGBADA (SHENZHEN) INTERNATIONAL TRADE CO., LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated May 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient Purpose
- PURPOSE
- Uses
-
Warnings
For external use only.
Ask a doctorbefore use if you have heart disease -high blood pressure - thyroid disease - diabetes -difficulty in urination due to enlargement of the prostate gland
Ask a doctor or pharmacist before use if you are presently taking a prescription drug for high blood pressure or depression.
When using this product do not exceed the recommended daily dosage unless directed by a doctor
Stop and ask a doctor if - bleeding occurs -condition worsens or does not improve within 7 days introduction of the applicator into the rectum causes additional pain -an allergic reaction occurs - symptom does not improve or if redness, irritation, swelling, pain or other symptoms develop or increase
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of accidental ingestion, seek medical advice immediately or contact a Poison Control Center right away. (1-800-222-1222)
- KEEP OUT OF REACH OF CHILDREN
-
Directions
- when practical, cleanse the affected area by patting or blotting with an appropriate cleansing wipe.
- Gently dry by patting or blotting with a tissue or a soft cloth before application.
- apply half the gel in the applicator to the external affected area (only applies if you have external symptoms)
- find a comfortable position and gently insert about ½ inch into the rectum.
- push the end of the applicator to apply the remaining gel inside the rectum (Both steps are necessary for both internal and external symptoms as external hemorrhoid often comes with internal hemorrhoid)
- dispose the applicator after use.
- avoid movement for 1-3 hours
- Children under 12 years of age: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
- Product label
-
INGREDIENTS AND APPEARANCE
ARIELLA HEMORRHOID GEL
phellodendron chinense whole,senegalia catechu wood,sophora flavescens root gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83364-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHELLODENDRON CHINENSE WHOLE (UNII: QKA3ZK8IIE) (PHELLODENDRON CHINENSE WHOLE - UNII:QKA3ZK8IIE) PHELLODENDRON CHINENSE WHOLE 6 g in 100 g SENEGALIA CATECHU WOOD (UNII: CHL342Y4LA) (SENEGALIA CATECHU WOOD - UNII:CHL342Y4LA) SENEGALIA CATECHU WOOD 2 g in 100 g SOPHORA FLAVESCENS ROOT (UNII: IYR6K8KQ5K) (SOPHORA FLAVESCENS ROOT - UNII:IYR6K8KQ5K) SOPHORA FLAVESCENS ROOT 12 g in 100 g Inactive Ingredients Ingredient Name Strength ZINC OXIDE (UNII: SOI2LOH54Z) CALAMUS DRACO WHOLE (UNII: 1C8HA8O22C) MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) GLYCYRRHIZA GLABRA (UNII: 2788Z9758H) WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) TROLAMINE (UNII: 9O3K93S3TK) ETHYLPARABEN (UNII: 14255EXE39) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83364-002-01 6 in 1 BOX 05/17/2023 1 3 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/17/2023 Labeler - YITONGBADA (SHENZHEN) INTERNATIONAL TRADE CO., LTD (725220463)