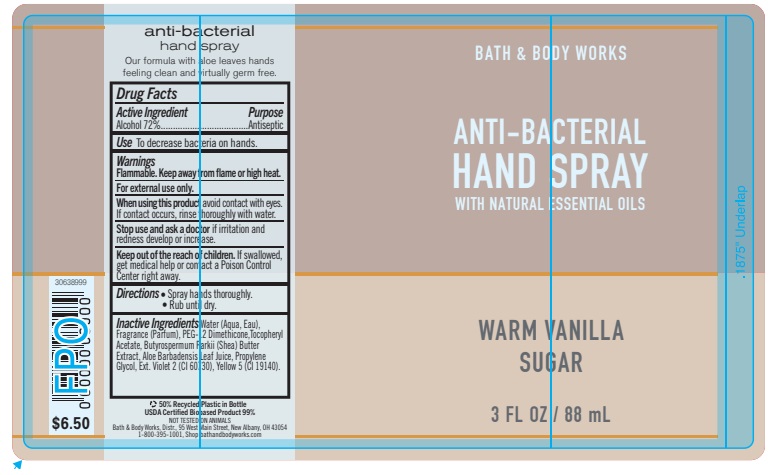
Label: ANTIBACTERIALHANDSPRAY WARM VANILLA SUGAR- alcohol spray
- NDC Code(s): 62670-6601-0
- Packager: Bath & Body Works, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 16, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
- WARNINGS
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- Directions
- INACTIVE INGREDIENT
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ANTIBACTERIALHANDSPRAY WARM VANILLA SUGAR
alcohol sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62670-6601 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 72 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62670-6601-0 88 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 11/16/2023 11/16/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 11/16/2023 11/16/2026 Labeler - Bath & Body Works, Inc. (878952845) Establishment Name Address ID/FEI Business Operations MCP TN 117443103 manufacture(62670-6601) Establishment Name Address ID/FEI Business Operations KDC US Holdings, Inc. (Groveport) 118156334 manufacture(62670-6601)