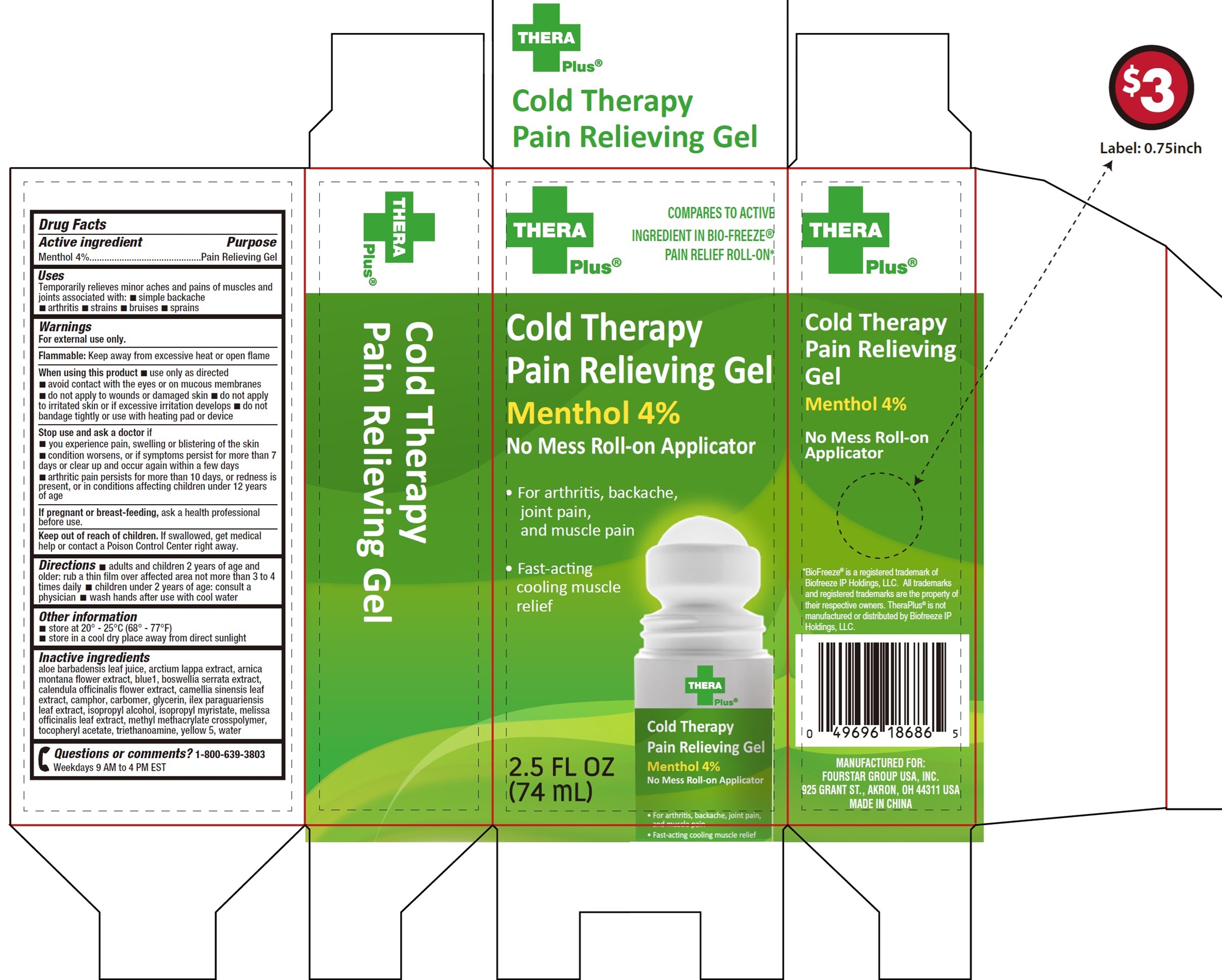
Label: THERA PLUS COLD THERAPY PAIN RELIEVING MENTHOL- menthol gel
- NDC Code(s): 80684-101-00
- Packager: Fourstar Group USA, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Uses
-
Warnings
For external use only.
Flammable:Keep away from excessive heat or open flame
When using this product
- use only as directed
- avoid contact with the eyes or on mucous membranes
- do not apply to wounds or damaged skin
- do not apply to irritated skin or if excessive irritation develops
- do not bandage tightly or use with heating pad or device
Stop use and ask a doctor if
- you experience pain, swelling or blistering of the skin
- condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days
- arthritic pain persists for more than 10 days, or redness is present, or in conditions affecting children under 12 years of age
- Directions
- Other information
-
Inactive ingredients
aloe barbadensis leaf juice, arctium lappa extract, arnica montana flower extract, blue1, boswellia serrata extract, calendula officinalis flower extract, camellia sinensis leaf extract, camphor, carbomer, glycerin, ilex paraguariensis leaf extract, isopropyl alcohol, isopropyl myristate, melissa officinalis leaf extract, methyl methacrylate crosspolymer, tocopheryl acetate, triethanoamine, yellow 5, water
- Questions or comments?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
THERA PLUS COLD THERAPY PAIN RELIEVING MENTHOL
menthol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80684-101 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 40 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) INDIAN FRANKINCENSE (UNII: 4PW41QCO2M) CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) GREEN TEA LEAF (UNII: W2ZU1RY8B0) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) GLYCERIN (UNII: PDC6A3C0OX) ILEX PARAGUARIENSIS LEAF (UNII: 1Q953B4O4F) ISOPROPYL ALCOHOL (UNII: ND2M416302) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) MELISSA OFFICINALIS LEAF (UNII: 50D2ZE9219) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) TROLAMINE (UNII: 9O3K93S3TK) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80684-101-00 74 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 02/01/2024 Labeler - Fourstar Group USA, Inc. (140099503)