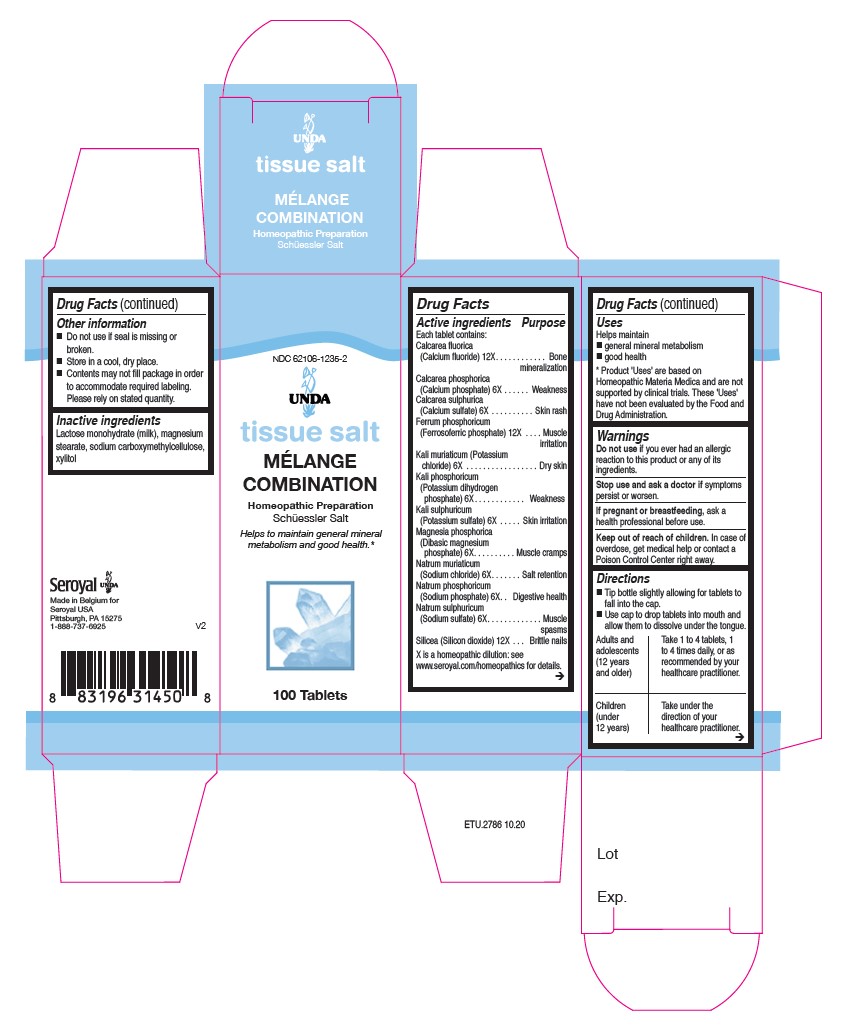
Label: MELANGE COMBINATION- calcarea sulphurica, calcium phosphoricum, kali muriaticum, kalium phosphoricum, kali sulphuricum, magnesia phosphorica, natrum muriaticum, natrum phosphoricum, natrium sulfuricum, calcarea fluorica, ferrum phosphoricum, silicea tablet
- NDC Code(s): 62106-1235-2
- Packager: Seroyal USA
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated July 14, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active Ingredients
Each Tablet Contains:
Calcarea fluorica (Calcium fluoride) 12X
Calcarea phosphorica (Calcium phosphate) 6X
Calcarea sulphurica (Calcium sulfate) 6X
Ferrum phosphoricum (Ferrosoferric phosphate) 12X
Kali muriaticum (Potassium chloride) 6X
Kali phosphoricum (Dibasic potassium phosphate) 6X
Kali sulphuricum (Potassium sulfate) 6X
Magnesia phosphorica (Dibasic magnesium phosphate) 6X
Natrum muriaticum (Sodium chloride) 6X
Natrum phosphoricum (Sodium phosphate) 6X
Natrum sulphuricum (Sodium sulfate) 6X
Silicea (Silicon dioxide) 12X - PURPOSE
-
WARNINGS
Warnings
Do not use if you ever had an allergic reaction to this product or any of its ingredients.
Stop use and ask a doctor if symptoms persist or worsen.
If pregnant or breastfeeding, ask a health professional before use.
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away.
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- ASK DOCTOR
- INACTIVE INGREDIENT
-
DOSAGE & ADMINISTRATION
Directions
Tip bottle slightly allowing for tablets to fall into the cap.
Use cap to drop tablets into mouth and allow them to dissolve under the tongue.
Adults and adolescents (12 years and older): Take 1 to 4 tablets, one to four times daily or as recommended by your healthcare practitioner.Children (under 12 years): Take under the direction of your healthcare practitioner.
-
INDICATIONS & USAGE
Uses
Helps to maintain general mineral metabolism and good health.Directions
Tip bottle slightly allowing for tablets to fall into the cap.
Use cap to drop tablets into mouth and allow them to dissolve under the tongue.
Adults and adolescents (12 years and older): Take 1 to 4 tablets, one to four times daily, or as recommended
by your healthcare practitioner.Children (under 12 years): Take under the direction of your healthcare practitioner.
- OTHER SAFETY INFORMATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MELANGE COMBINATION
calcarea sulphurica, calcium phosphoricum, kali muriaticum, kalium phosphoricum, kali sulphuricum, magnesia phosphorica, natrum muriaticum, natrum phosphoricum, natrium sulfuricum, calcarea fluorica, ferrum phosphoricum, silicea tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62106-1235 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: L11K75P92J) (CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS - UNII:L11K75P92J) CALCIUM PHOSPHATE, DIBASIC, ANHYDROUS 6 [hp_X] POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM CHLORIDE 6 [hp_X] POTASSIUM PHOSPHATE, DIBASIC (UNII: CI71S98N1Z) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM PHOSPHATE, DIBASIC 6 [hp_X] CALCIUM SULFATE ANHYDROUS (UNII: E934B3V59H) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM SULFATE ANHYDROUS 6 [hp_X] SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE (UNII: 70WT22SF4B) (PHOSPHATE ION - UNII:NK08V8K8HR) SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE 6 [hp_X] SILICON DIOXIDE (UNII: ETJ7Z6XBU4) (SILICON DIOXIDE - UNII:ETJ7Z6XBU4) SILICON DIOXIDE 12 [hp_X] POTASSIUM SULFATE (UNII: 1K573LC5TV) (SULFATE ION - UNII:7IS9N8KPMG) POTASSIUM SULFATE 6 [hp_X] MAGNESIUM PHOSPHATE, DIBASIC TRIHYDRATE (UNII: HF539G9L3Q) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM PHOSPHATE, DIBASIC TRIHYDRATE 6 [hp_X] SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM CHLORIDE 6 [hp_X] SODIUM SULFATE (UNII: 0YPR65R21J) (SODIUM SULFATE ANHYDROUS - UNII:36KCS0R750) SODIUM SULFATE 6 [hp_X] CALCIUM FLUORIDE (UNII: O3B55K4YKI) (FLUORIDE ION - UNII:Q80VPU408O) CALCIUM FLUORIDE 12 [hp_X] FERROSOFERRIC PHOSPHATE (UNII: 91GQH8I5F7) (FERROSOFERRIC PHOSPHATE - UNII:91GQH8I5F7) FERROSOFERRIC PHOSPHATE 12 [hp_X] Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) XYLITOL (UNII: VCQ006KQ1E) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color white Score no score Shape ROUND Size 7mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62106-1235-2 1 in 1 BOX 12/16/2014 1 100 in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 12/12/2014 Labeler - Seroyal USA (018361118) Establishment Name Address ID/FEI Business Operations SAN’UP 401010287 manufacture(62106-1235)