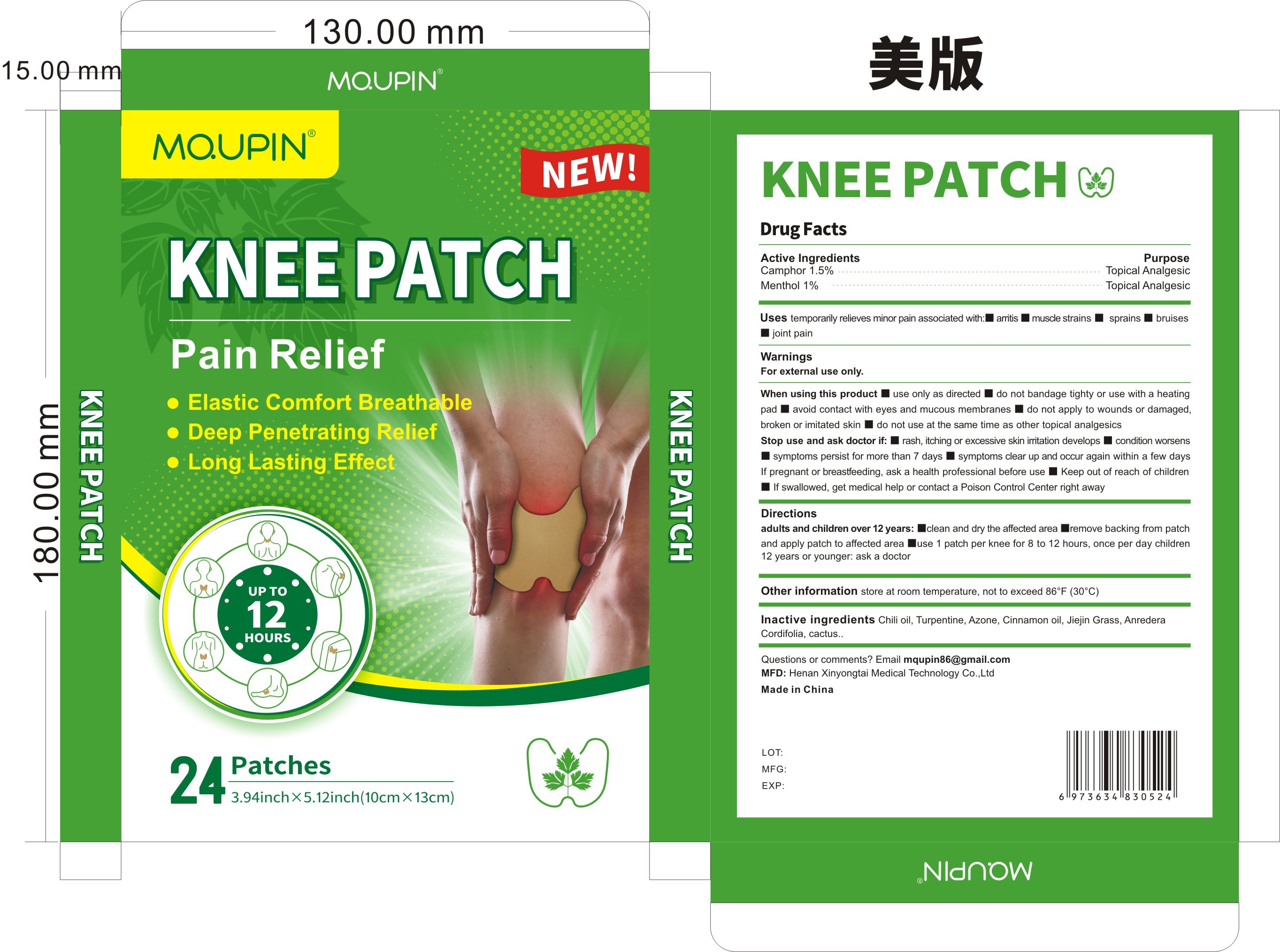
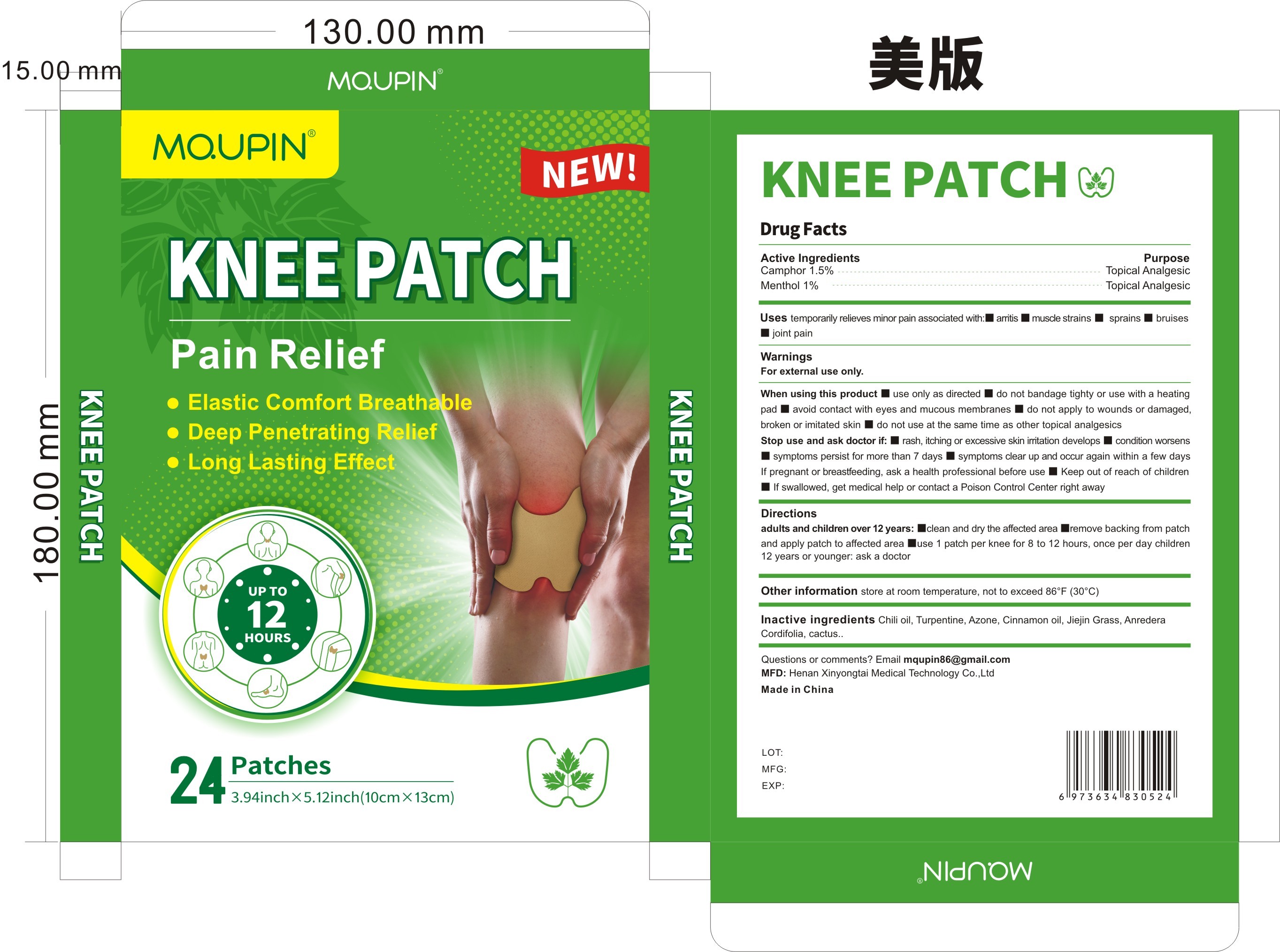
Label: MQUPIN KNEE PATCH PAIN RELIFE- knee patch pain relife patch
- NDC Code(s): 83781-004-01
- Packager: Zhengzhou Miaoqi Pharmaceutical Technology Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 8, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
- When Using
-
Stop Use
■ rash, itching or excessive skin irritation develops
■ condition worsens
■ symptoms persist for more than 7 days
■ symptoms clear up and occur again within a few days If pregnant or breastfeeding, ask a health professional before use
■ Keep out of reach of children
■ If swallowed, get medical help or contact a Poison Control Center right away
- Ask Doctor
- Keep Out Of Reach Of Children
- Directions
- Other information
- Inactive ingredients
- Questions
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MQUPIN KNEE PATCH PAIN RELIFE
knee patch pain relife patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83781-004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BORNEOL (UNII: M89NIB437X) (BORNEOL - UNII:M89NIB437X) BORNEOL 1.5 g in 100 MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 1 g in 100 Inactive Ingredients Ingredient Name Strength CINNAMON OIL (UNII: E5GY4I6YCZ) ANREDERA CORDIFOLIA LEAF (UNII: MGE8MNJ5L6) TURPENTINE (UNII: XJ6RUH0O4G) CAPSICUM (UNII: 00UK7646FG) LAUROCAPRAM (UNII: 1F3X9DRV9X) HOODIA GORDONII SAP (UNII: K7QNF2DJR8) GRASS CARP (UNII: 9A3N315I11) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83781-004-01 24 in 1 BOX; Type 0: Not a Combination Product 11/05/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 11/05/2023 Labeler - Zhengzhou Miaoqi Pharmaceutical Technology Co., Ltd (701762807) Establishment Name Address ID/FEI Business Operations Zhengzhou Miaoqi Pharmaceutical Technology Co., Ltd 701762807 manufacture(83781-004) , label(83781-004)