Label: ROTOSXIKA VARICOCE VEINS PLASTER- aesculus chinensis, arnica patch
- NDC Code(s): 83702-016-11, 83702-016-12
- Packager: Puning Xinhua Trade Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated October 24, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
-
WARNINGS
Precautions:
1.This product is an external product, do not take it internally;
2.This product is a health care product and does not replace drugs and medical devices fortreatment;
3.Pregnant women, infants, breastfeeding, and those allergic to this product are prohibited;
4.Do not use skin allergies, skin damage, skin diseases and other symptoms;
5.If the skin appears red, itchy and other abnormal conditions during use, please suspend use. - QUESTIONS
- INACTIVE INGREDIENT
- STATEMENT OF IDENTITY
- KEEP OUT OF REACH OF CHILDREN
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ROTOSXIKA VARICOCE VEINS PLASTER
aesculus chinensis, arnica patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83702-016 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AESCULUS CHINENSIS WHOLE (UNII: PGU072XJ6U) (AESCULUS CHINENSIS WHOLE - UNII:PGU072XJ6U) AESCULUS CHINENSIS WHOLE 4 g in 100 g ARNICA CORDIFOLIA WHOLE (UNII: 82CNE9JKJI) (ARNICA CORDIFOLIA WHOLE - UNII:82CNE9JKJI) ARNICA CORDIFOLIA WHOLE 3 g in 100 g Inactive Ingredients Ingredient Name Strength POTASSIUM SORBATE (UNII: 1VPU26JZZ4) WATER (UNII: 059QF0KO0R) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83702-016-12 6 in 1 BAG 06/01/2023 1 NDC:83702-016-11 2.1 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 06/01/2023 Labeler - Puning Xinhua Trade Co., Ltd. (632673277) Registrant - Nanyang Jingzhilan Biotechnology Co., Ltd. (724044399) Establishment Name Address ID/FEI Business Operations Nanyang Jingzhilan Biotechnology Co., Ltd. 724044399 manufacture(83702-016) , label(83702-016)

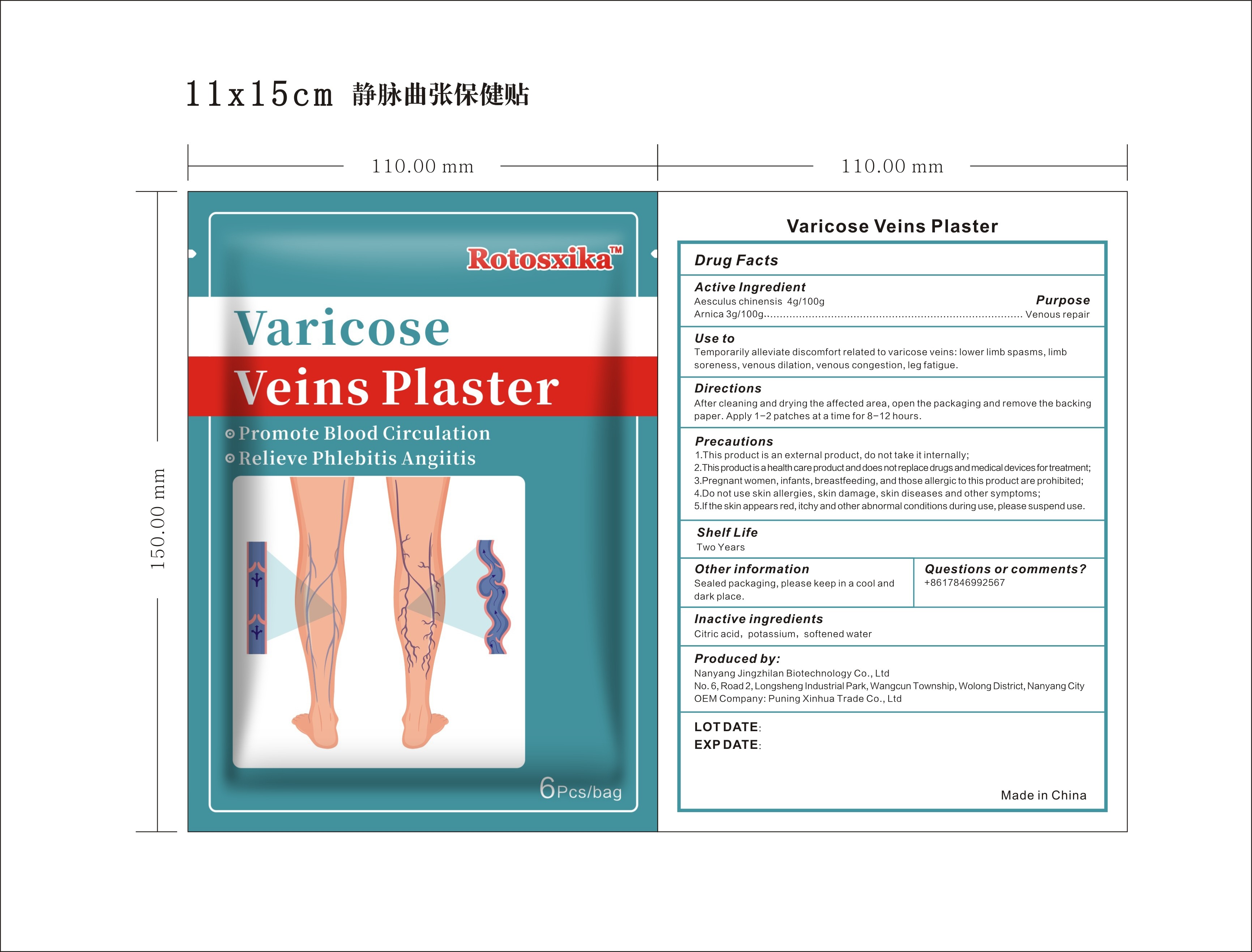
 Label
Label