Label: ATACAND- candesartan cilexetil tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 54868-4413-0, 54868-4612-0, 54868-5489-0, 54868-5591-0 - Packager: Physicians Total Care, Inc.
- This is a repackaged label.
- Source NDC Code(s): 0186-0004, 0186-0008, 0186-0016, 0186-0032
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated August 12, 2013
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ATACAND safely and effectively. See full prescribing information for ATACAND.
ATACAND® (candesartan cilexetil) TABLETS
INITIAL U.S. APPROVAL: 1998WARNING: USE IN PREGNANCY: See Full Prescribing Information for complete boxed warning.
When used in pregnancy during the second and third trimesters, drugs that act directly on the renin-angiotensin system can cause injury and even death to the developing fetus. When pregnancy is detected, ATACAND should be discontinued as soon as possible. See WARNINGS AND PRECAUTIONS, Fetal/Neonatal Morbidity and Mortality (5.1).
INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
(2)
Starting Dose (2)
Dose Range (2)
Target Maintenance Dose (2)
Adult Hypertension (2.1) (2)
16 mg tablet once daily (2)
8 - 32 mg tablet total daily dose (2)
– (2)
Pediatric Hypertension (1 to < 6 years) (2.2) (2)
0.20 mg/kg oral suspension once daily (2)
0.05 - 0.4 mg/kg oral suspension once daily or consider divided dose (2)
- (2)
Pediatric Hypertension (6 to < 17 years) (2.2) (2)
< 50 kg 4 – 8 mg tablet once daily (2)
> 50 kg 8 – 16 mg tablet once daily (2)
< 50 kg 4 – 16 mg tablet once daily or consider divided dose (2)
> 50 kg 4 – 32 mg tablet once daily or consider divided dose (2)
– (2)
Adult Heart Failure (2.3) (2)
4 mg tablet once daily (2)
(2)
32 mg tablet once daily (2)
(2)
DOSAGE FORMS AND STRENGTHS
Tablets 4 mg, 8 mg, 16 mg, 32 mg (3). (3)
CONTRAINDICATIONS
Known hypersensitivity to product components (4). (4)
WARNINGS AND PRECAUTIONS
· Avoid fetal (in utero) and neonatal exposure (5.1). (5)
· Children < 1 year of age must not receive ATACAND for hypertension (5.2). (5)
· Observe for signs and symptoms of hypotension (5.3). (5)
· Use with caution in patients with impaired hepatic (5.4) or renal (5.5) function. (5)
· Hyperkalemia may occur in heart failure patients treated with ATACAND (5.6). (5)
ADVERSE REACTIONS
· Most common adverse reactions which caused adult patients to discontinue therapy for: (6)
· Hypertension were headache (0.6%) and dizziness (0.3%) (6.1). (6)
· Heart Failure were hypotension (4.1%) (5.3), abnormal renal function (6.3%) (5.5), and hyperkalemia (2.4%) (5.6). (6)
· Most common adverse reactions (incidence ≥ 2% and greater than placebo) are back pain, dizziness, upper respiratory tract infection, pharyngitis and rhinitis (6.1). (6)
To report SUSPECTED ADVERSE REACTIONS contact AstraZeneca at 1–800–236–9933 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. (6)
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
· Nursing Mothers: Either nursing or drug should be discontinued (8.3). (8)
· Pediatrics: Children < 1 year of age must not receive ATACAND for hypertension (5.2). Inhibitors of the renin-angiotensin system can cause renal abnormalities in neonatal animals (12.3). (8)
· Geriatrics: No overall difference in efficacy or safety vs. younger adult patients, but greater sensitivity of some older individuals cannot be ruled out (8.5). (8)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2012
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
Warning Use in Pregnancy
1 INDICATIONS AND USAGE
1.1 Hypertension
1.2 Heart Failure
2 DOSAGE AND ADMINISTRATION
2.1 Adult Hypertension
2.2 Pediatric Hypertension 1 to < 17 Years of age
2.3 Adult Heart Failure
3 DOSAGE FORMS and STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Fetal/Neonatal Morbidity and Mortality
5.2 Morbidity in Infants
5.3 Hypotension
5.4 Impaired Hepatic Function
5.5 Renal Function Deterioration
5.6 Hyperkalemia
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
6.2 Postmarketing Experience
6.3 Laboratory Test Findings
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Labor and Delivery
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14.1 Hypertension
14.2 Heart Failure
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
Warning Use in Pregnancy
When used in pregnancy during the second and third trimesters, drugs that act directly on the renin-angiotensin system can cause injury and even death to the developing fetus. When pregnancy is detected, ATACAND should be discontinued as soon as possible [see WARNINGS AND PRECAUTIONS, Fetal/Neonatal Morbidity and Mortality (5.1)].
-
1 INDICATIONS AND USAGE
1.1 Hypertension
ATACAND is indicated for the treatment of hypertension in adults and children 1 to < 17 years of age. It may be used alone or in combination with other antihypertensive agents.
1.2 Heart Failure
ATACAND is indicated for the treatment of heart failure (NYHA class II-IV) in adults with left ventricular systolic dysfunction (ejection fraction ≤ 40%) to reduce cardiovascular death and to reduce heart failure hospitalizations [see CLINICAL STUDIES (14.2)]. ATACAND also has an added effect on these outcomes when used with an ACE inhibitor.
-
2 DOSAGE AND ADMINISTRATION
2.1 Adult Hypertension
Dosage must be individualized. Blood pressure response is dose related over the range of 2 to 32 mg. The usual recommended starting dose of ATACAND is 16 mg once daily when it is used as monotherapy in patients who are not volume depleted. ATACAND can be administered once or twice daily with total daily doses ranging from 8 mg to 32 mg. Larger doses do not appear to have a greater effect, and there is relatively little experience with such doses. Most of the antihypertensive effect is present within 2 weeks, and maximal blood pressure reduction is generally obtained within 4 to 6 weeks of treatment with ATACAND.
No initial dosage adjustment is necessary for elderly patients, for patients with mildly impaired renal function, or for patients with mildly impaired hepatic function [see CLINICAL PHARMACOLOGY (12.3)]. In patients with moderate hepatic impairment, consideration should be given to initiation of ATACAND at a lower dose [see CLINICAL PHARMACOLOGY (12.3)]. For patients with possible depletion of intravascular volume (eg, patients treated with diuretics, particularly those with impaired renal function), ATACAND should be initiated under close medical supervision and consideration should be given to administration of a lower dose [see WARNINGS AND PRECAUTIONS (5.3)].
ATACAND may be administered with or without food.
If blood pressure is not controlled by ATACAND alone, a diuretic may be added. ATACAND may be administered with other antihypertensive agents.
2.2 Pediatric Hypertension 1 to < 17 Years of age
ATACAND may be administered once daily or divided into two equal doses. Adjust the dosage according to blood pressure response. For patients with possible depletion of intravascular volume (e.g., patients treated with diuretics, particularly those with impaired renal function), initiate ATACAND under close medical supervision and consider administration of a lower dose [see WARNINGS AND PRECAUTIONS (5.3)].
Children 1 to < 6 years of age:
The dose range is 0.05 to 0.4 mg/kg per day. The recommended starting dose is 0.20 mg/kg (oral suspension).
Children 6 to < 17 years of age:
For those less than 50 kg, the dose range is 2 to 16 mg per day. The recommended starting dose is 4 to 8 mg.
For those greater than 50 kg, the dose range is 4 to 32 mg per day. The recommended starting dose is 8 to 16 mg.
Doses above 0.4 mg/kg (1 to < 6 year olds) or 32 mg (6 to < 17 year olds) have not been studied in pediatric patients [see CLINICAL STUDIES (14.1)].
An antihypertensive effect is usually present within 2 weeks, with full effect generally obtained within 4 weeks of treatment with ATACAND.
Children < 1 year of age must not receive ATACAND for hypertension.
All pediatric patients with a glomerular filtration rate less than 30 ml/min/1.73m2 should not receive ATACAND since ATACAND has not been studied in this population [see WARNINGS AND PRECAUTIONS (5.2)].
For children who cannot swallow tablets, an oral suspension may be substituted [see Preparation of Oral Suspension].
Preparation of Oral Suspension:
ATACAND oral suspension can be prepared in concentrations within the range of 0.1 to 2.0 mg/mL. Typically, a concentration of 1 mg/mL will be suitable for the prescribed dose. Any strength of ATACAND tablets can be used in the preparation of the suspension.
Follow the steps below for preparation of the suspension. The number of tablets and volume of vehicle specified below will yield 160 mL of a 1 mg/mL suspension.
· Prepare the vehicle by adding equal volumes of *Ora-Plus® (80 mL) and *Ora-Sweet SF® (80 mL) or, alternatively, use *,†Ora-Blend SF® (160 mL).
· Add a small amount of vehicle to the required number of ATACAND tablets (five 32 mg tablets) and grind into a smooth paste using a mortar and pestle.
· Add the paste to a preparation vessel of suitable size.
· Rinse the mortar and pestle clean using the vehicle and add this to the vessel. Repeat, if necessary.
· Prepare the final volume by adding the remaining vehicle.
· Mix thoroughly.
· Dispense into suitably sized amber PET bottles.
· Label with an expiry date of 100 days and include the following instructions:
Store at room temperature (below 30°C/86°F). Use within 30 days after first opening. Do not use after the expiry date stated on the bottle.
Do not freeze.
Shake well before each use.
*Ora-Plus®, Ora-Sweet SF®, and Ora-Blend SF® are registered trademarks of Paddock Laboratories, Inc.
†Supplied as a 50/50% pre-mix of Ora-Plus® and Ora-Sweet SF®.
-
3 DOSAGE FORMS and STRENGTHS
4 mg are white to off-white, circular/biconvex-shaped, non-film-coated scored tablets, coded ACF on one side and 004 on the other.
8 mg are light pink, circular/biconvex-shaped, non-film-coated scored tablets, coded ACG on one side and 008 on the other.
16 mg are pink, circular/biconvex-shaped, non-film-coated scored tablets, coded ACH on one side and 016 on the other.
32 mg are pink, circular/biconvex-shaped, non-film-coated scored tablets, coded ACL on one side and 032 on the other.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Fetal/Neonatal Morbidity and Mortality
Drugs that act directly on the renin-angiotensin system can cause fetal and neonatal morbidity and death when administered to pregnant women. Several dozen cases have been reported in the world literature in patients who were taking angiotensin-converting enzyme inhibitors. Post-marketing experience has identified reports of fetal and neonatal toxicity in babies born to women treated with ATACAND during pregnancy. When pregnancy is detected, ATACAND should be discontinued as soon as possible.
The use of drugs that act directly on the renin-angiotensin system during the second and third trimesters of pregnancy has been associated with fetal and neonatal injury, including hypotension, neonatal skull hypoplasia, anuria, reversible or irreversible renal failure, and death. Oligohydramnios has also been reported, presumably resulting from decreased fetal renal function; oligohydramnios in this setting has been associated with fetal limb contractures, craniofacial deformation, and hypoplastic lung development. Prematurity, intrauterine growth retardation, and patent ductus arteriosus have also been reported, although it is not clear whether these occurrences were due to exposure to the drug.
These adverse effects do not appear to have resulted from intrauterine drug exposure that has been limited to the first trimester. Mothers whose embryos and fetuses are exposed to an angiotensin II receptor antagonist only during the first trimester should be so informed. Nonetheless, when patients become pregnant, physicians should have the patient discontinue the use of ATACAND as soon as possible.
Rarely (probably less often than once in every thousand pregnancies), no alternative to a drug acting on the renin-angiotensin system will be found. In these rare cases, the mothers should be apprised of the potential hazards to their fetuses, and serial ultrasound examinations should be performed to assess the intra-amniotic environment.
If oligohydramnios is observed, ATACAND should be discontinued unless it is considered life saving for the mother. Contraction stress testing (CST), a nonstress test (NST), or biophysical profiling (BPP) may be appropriate, depending upon the week of pregnancy. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury.
Infants with histories of in utero exposure to an angiotensin II receptor antagonist should be closely observed for hypotension, oliguria, and hyperkalemia. If oliguria occurs, attention should be directed toward support of blood pressure and renal perfusion. Exchange transfusion or dialysis may be required as means of reversing hypotension and/or substituting for disordered renal function.
Oral doses ≥10 mg of candesartan cilexetil/kg/day administered to pregnant rats during late gestation and continued through lactation were associated with reduced survival and an increased incidence of hydronephrosis in the offspring. The 10-mg/kg/day dose in rats is approximately 2.8 times the maximum recommended daily human dose (MRHD) of 32 mg on a mg/m2 basis (comparison assumes human body weight of 50 kg). Candesartan cilexetil given to pregnant rabbits at an oral dose of 3 mg/kg/day (approximately 1.7 times the MRHD on a mg/m2 basis) caused maternal toxicity (decreased body weight and death) but, in surviving dams, had no adverse effects on fetal survival, fetal weight, or external, visceral, or skeletal development. No maternal toxicity or adverse effects on fetal development were observed when oral doses up to 1000 mg of candesartan cilexetil/kg/day (approximately 138 times the MRHD on a mg/m2 basis) were administered to pregnant mice.
5.2 Morbidity in Infants
Children < 1 year of age must not receive ATACAND for hypertension. The consequences of administering drugs that act directly on the renin-angiotensin system (RAS) can have effects on the development of immature kidneys.
5.3 Hypotension
In adult or children patients with an activated renin-angiotensin system, such as volume- and/or salt-depleted patients (eg, those being treated with diuretics), symptomatic hypotension may occur. These conditions should be corrected prior to administration of ATACAND, or the treatment should start under close medical supervision [see DOSAGE AND ADMINISTRATION (2.1)].
If hypotension occurs, the patients should be placed in the supine position and, if necessary, given an intravenous infusion of normal saline. A transient hypotensive response is not a contraindication to further treatment which usually can be continued without difficulty once the blood pressure has stabilized.
Caution should be observed when initiating therapy in patients with heart failure. Patients with heart failure given ATACAND commonly have some reduction in blood pressure. In patients with symptomatic hypotension this may require temporarily reducing the dose of ATACAND, or diuretic, or both, and volume repletion. In the CHARM program, hypotension was reported in 18.8% of patients on ATACAND versus 9.8% of patients on placebo. The incidence of hypotension leading to drug discontinuation in ATACAND-treated patients was 4.1% compared with 2.0% in placebo-treated patients.
Monitoring of blood pressure is recommended during dose escalation and periodically thereafter.
Major Surgery/Anesthesia
Hypotension may occur during major surgery and anesthesia in patients treated with angiotensin II receptor antagonists, including ATACAND, due to blockade of the renin-angiotensin system. Very rarely, hypotension may be severe such that it may warrant the use of intravenous fluids and/or vasopressors.
5.4 Impaired Hepatic Function
Based on pharmacokinetic data which demonstrate significant increases in candesartan AUC and Cmax in patients with moderate hepatic impairment, a lower initiating dose should be considered for patients with moderate hepatic impairment [see CLINICAL PHARMACOLOGY (12.3)].
5.5 Renal Function Deterioration
As a consequence of inhibiting the renin-angiotensin-aldosterone system, changes in renal function may be anticipated in some individuals treated with ATACAND. In patients whose renal function may depend upon the activity of the renin-angiotensin-aldosterone system (eg, patients with severe heart failure), treatment with angiotensin-converting enzyme inhibitors and angiotensin receptor antagonists has been associated with oliguria and/or progressive azotemia and (rarely) with acute renal failure and/or death. Similar results may be anticipated in patients treated with ATACAND [see CLINICAL PHARMACOLOGY (12.3)].
In studies of ACE inhibitors in patients with unilateral or bilateral renal artery stenosis, increases in serum creatinine or blood urea nitrogen (BUN) have been reported. There has been no long-term use of ATACAND in patients with unilateral or bilateral renal artery stenosis, but similar results may be expected.
In heart failure patients treated with ATACAND, increases in serum creatinine may occur. Dosage reduction or discontinuation of the diuretic or ATACAND, and volume repletion may be required. In the CHARM program, the incidence of abnormal renal function (e.g., creatinine increase) was 12.5% in patients treated with ATACAND versus 6.3% in patients treated with placebo. The incidence of abnormal renal function (eg, creatinine increase) leading to drug discontinuation in ATACAND-treated patients was 6.3% compared with 2.9% in placebo-treated patients. Evaluation of patients with heart failure should always include assessment of renal function and volume status. Monitoring of serum creatinine is recommended during dose escalation and periodically thereafter.
Pediatrics - ATACAND has not been studied in children with estimated glomerular filtration rate < 30 mL/min/1.73m2.
5.6 Hyperkalemia
In heart failure patients treated with ATACAND, hyperkalemia may occur, especially when taken concomitantly with ACE inhibitors and potassium-sparing diuretics such as spironolactone. In the CHARM program, the incidence of hyperkalemia was 6.3% in patients treated with ATACAND versus 2.1% in patients treated with placebo. The incidence of hyperkalemia leading to drug discontinuation in ATACAND-treated patients was 2.4% compared with 0.6% in placebo-treated patients. During treatment with ATACAND in patients with heart failure, monitoring of serum potassium is recommended during dose escalation and periodically thereafter.
-
6 ADVERSE REACTIONS
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
Adult Hypertension
ATACAND has been evaluated for safety in more than 3600 patients/subjects, including more than 3200 patients treated for hypertension. About 600 of these patients were studied for at least 6 months and about 200 for at least 1 year. In general, treatment with ATACAND was well tolerated. The overall incidence of adverse events reported with ATACAND was similar to placebo.
The rate of withdrawals due to adverse events in all trials in patients (7510 total) was 3.3% (ie, 108 of 3260) of patients treated with ATACAND as monotherapy and 3.5% (ie, 39 of 1106) of patients treated with placebo. In placebo-controlled trials, discontinuation of therapy due to clinical adverse events occurred in 2.4% (ie, 57 of 2350) of patients treated with ATACAND and 3.4% (ie, 35 of 1027) of patients treated with placebo.
The most common reasons for discontinuation of therapy with ATACAND were headache (0.6%) and dizziness (0.3%).
The adverse events that occurred in placebo-controlled clinical trials in at least 1% of patients treated with ATACAND and at a higher incidence in candesartan cilexetil (n = 2350) than placebo (n = 1027) patients included back pain (3% vs. 2%), dizziness (4% vs. 3%), upper respiratory tract infection (6% vs. 4%), pharyngitis (2% vs. 1%), and rhinitis (2% vs. 1%).
The following adverse events occurred in placebo-controlled clinical trials at a more than 1% rate but at about the same or greater incidence in patients receiving placebo compared to ATACAND: fatigue, peripheral edema, chest pain, headache, bronchitis, coughing, sinusitis, nausea, abdominal pain, diarrhea, vomiting, arthralgia, albuminuria.
Other potentially important adverse events that have been reported, whether or not attributed to treatment, with an incidence of 0.5% or greater from the 3260 patients worldwide treated in clinical trials with ATACAND are listed below. It cannot be determined whether these events were causally related to ATACAND. Body as a Whole: asthenia, fever; Central and Peripheral Nervous System: paresthesia, vertigo; Gastrointestinal System Disorder: dyspepsia, gastroenteritis; Heart Rate and Rhythm Disorders: tachycardia, palpitation; Metabolic and Nutritional Disorders: creatine phosphokinase increased, hyperglycemia, hypertriglyceridemia, hyperuricemia; Musculoskeletal System Disorders: myalgia; Platelet/Bleeding-Clotting Disorders: epistaxis; Psychiatric Disorders: anxiety, depression, somnolence; Respiratory System Disorders: dyspnea; Skin and Appendages Disorders: rash, sweating increased; Urinary System Disorders: hematuria.
Other reported events seen less frequently included angina pectoris, myocardial infarction, and angioedema.
Adverse events occurred at about the same rates in men and women, older and younger patients, and black and non-black patients.
Pediatric Hypertension
Among children in clinical studies, 1 in 93 children age 1 to < 6 and 3 in 240 age 6 to < 17 experienced worsening renal disease. The association between candesartan and exacerbation of the underlying condition could not be excluded.
Heart Failure
The adverse event profile of ATACAND in adult heart failure patients was consistent with the pharmacology of the drug and the health status of the patients. In the CHARM program, comparing ATACAND in total daily doses up to 32 mg once daily (n=3803) with placebo (n=3796), 21.0% of patients discontinued ATACAND for adverse events vs. 16.1% of placebo patients.
6.2 Postmarketing Experience
The following adverse reactions were identified during post-approval use of ATACAND. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following have been very rarely reported in post-marketing experience:
Digestive: Abnormal hepatic function and hepatitis.
Hematologic: Neutropenia, leukopenia, and agranulocytosis.
Metabolic and Nutritional Disorders: hyperkalemia, hyponatremia.
Renal: renal impairment, renal failure.
Skin and Appendages Disorders: Pruritus and urticaria.
Rare reports of rhabdomyolysis have been reported in patients receiving angiotensin II receptor blockers.
6.3 Laboratory Test Findings
Hypertension
In controlled clinical trials, clinically important changes in standard laboratory parameters were rarely associated with the administration of ATACAND.
Creatinine, Blood Urea Nitrogen
Minor increases in blood urea nitrogen (BUN) and serum creatinine were observed infrequently.
Hyperuricemia
Hyperuricemia was rarely found (19 or 0.6% of 3260 patients treated with ATACAND and 5 or 0.5% of 1106 patients treated with placebo).
Hemoglobin and Hematocrit
Small decreases in hemoglobin and hematocrit (mean decreases of approximately 0.2 grams/dL and 0.5 volume percent, respectively) were observed in patients treated with ATACAND alone but were rarely of clinical importance. Anemia, leukopenia, and thrombocytopenia were associated with withdrawal of one patient each from clinical trials.
Potassium
A small increase (mean increase of 0.1 mEq/L) was observed in patients treated with ATACAND alone but was rarely of clinical importance. One patient from a congestive heart failure trial was withdrawn for hyperkalemia (serum potassium = 7.5 mEq/L). This patient was also receiving spironolactone [see WARNINGS AND PRECAUTIONS (5.6)].
Liver Function Tests
Elevations of liver enzymes and/or serum bilirubin were observed infrequently. Five patients assigned to ATACAND in clinical trials were withdrawn because of abnormal liver chemistries. All had elevated transaminases. Two had mildly elevated total bilirubin, but one of these patients was diagnosed with Hepatitis A.
Heart Failure
In the CHARM program, small increases in serum creatinine (mean increase 0.2 mg/dL in candesartan-treated patients and 0.1 mg/dL in placebo-treated patients) and serum potassium (mean increase 0.15 mEq/L in ATACAND-treated patients and 0.02 mEq/L in placebo-treated patients), and small decreases in hemoglobin (mean decrease 0.5 gm/dL in ATACAND-treated patients and 0.3 gm/dL in placebo-treated patients) and hematocrit (mean decrease 1.6% in ATACAND-treated patients and 0.9% in placebo-treated patients) were observed.
-
7 DRUG INTERACTIONS
No significant drug interactions have been reported in studies of candesartan cilexetil given with other drugs such as glyburide, nifedipine, digoxin, warfarin, hydrochlorothiazide, and oral contraceptives in healthy volunteers, or given with enalapril to patients with heart failure (NYHA class II and III). Because candesartan is not significantly metabolized by the cytochrome P450 system and at therapeutic concentrations has no effects on P450 enzymes, interactions with drugs that inhibit or are metabolized by those enzymes would not be expected.
Non-Steroidal Anti-Inflammatory Agents including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors). In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, co-administration of NSAIDs, including selective COX-2 inhibitors, with angiotensin II receptor antagonists, including candesartan, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving candesartan and NSAID therapy.
The antihypertensive effect of angiotensin II receptor antagonists, including candesartan may be attenuated by NSAIDs including selective COX-2 inhibitors.
Lithium
Reversible increases in serum lithium concentrations and toxicity have been reported during concomitant administration of lithium with ACE inhibitors, and with some angiotensin II receptor antagonists. An increase in serum lithium concentration has been reported during concomitant administration of lithium with ATACAND, so careful monitoring of serum lithium levels is recommended during concomitant use.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Categories C (first trimester) and D (second and third trimesters) [see WARNINGS AND PRECAUTIONS (5.1)].
8.2 Labor and Delivery
The effect of ATACAND on labor and delivery in humans is unknown [see WARNINGS AND PRECAUTIONS (5.1)].
8.3 Nursing Mothers
It is not known whether candesartan is excreted in human milk, but candesartan has been shown to be present in rat milk. Because of the potential for adverse effects on the nursing infant, a decision should be made whether to discontinue nursing or discontinue ATACAND, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
The antihypertensive effects of ATACAND were evaluated in hypertensive children 1 to < 17 years of age in randomized, double-blind clinical studies [see CLINICAL STUDIES (14.1)]. The pharmacokinetics of ATACAND have been evaluated in pediatric patients 1 to < 17 years of age [see Pharmacokinetics (12.3)].
Children < 1 year of age must not receive ATACAND for hypertension [see WARNINGS AND PRECAUTIONS (5.2)].
8.5 Geriatric Use
Hypertension
Of the total number of subjects in clinical studies of ATACAND, 21% (683/3260) were 65 and over, while 3% (87/3260) were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger adult subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. In a placebo-controlled trial of about 200 elderly hypertensive patients (ages 65 to 87 years), administration of candesartan cilexetil was well tolerated and lowered blood pressure by about 12/6 mm Hg more than placebo.
Heart Failure
Of the 7599 patients with heart failure in the CHARM program, 4343 (57%) were age 65 years or older and 1736 (23%) were 75 years or older. In patients ≥ 75 years of age, the incidence of drug discontinuations due to adverse events was higher for those treated with ATACAND or placebo compared with patients <75 years of age. In these patients, the most common adverse events leading to drug discontinuation at an incidence of at least 3%, and more frequent with ATACAND than placebo, were abnormal renal function (7.9% vs. 4.0%), hypotension (5.2% vs. 3.2%) and hyperkalemia (4.2% vs. 0.9%). In addition to monitoring of serum creatinine, potassium, and blood pressure during dose escalation and periodically thereafter, greater sensitivity of some older individuals with heart failure must be considered.
-
10 OVERDOSAGE
No lethality was observed in acute toxicity studies in mice, rats, and dogs given single oral doses of up to 2000 mg/kg of candesartan cilexetil. In mice given single oral doses of the primary metabolite, candesartan, the minimum lethal dose was greater than 1000 mg/kg but less than 2000 mg/kg.
The most likely manifestation of overdosage with ATACAND would be hypotension, dizziness, and tachycardia; bradycardia could occur from parasympathetic (vagal) stimulation. If symptomatic hypotension should occur, supportive treatment should be instituted.
Candesartan cannot be removed by hemodialysis.
Treatment: To obtain up-to-date information about the treatment of overdose, consult your Regional Poison Control Center. Telephone numbers of certified poison control centers are listed in the Physicians’ Desk Reference (PDR). In managing overdose, consider the possibilities of multiple-drug overdoses, drug-drug interactions, and altered pharmacokinetics in your patient.
-
11 DESCRIPTION
ATACAND (candesartan cilexetil), a prodrug, is hydrolyzed to candesartan during absorption from the gastrointestinal tract. Candesartan is a selective AT1 subtype angiotensin II receptor antagonist.
Candesartan cilexetil, a nonpeptide, is chemically described as (±)-1-Hydroxyethyl 2-ethoxy-1-[p-(o-1H-tetrazol-5-ylphenyl)benzyl]-7-benzimidazolecarboxylate, cyclohexyl carbonate (ester).
Its empirical formula is C33H34N6O6, and its structural formula is:
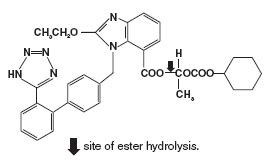
Candesartan cilexetil is a white to off-white powder with a molecular weight of 610.67. It is practically insoluble in water and sparingly soluble in methanol. Candesartan cilexetil is a racemic mixture containing one chiral center at the cyclohexyloxycarbonyloxy ethyl ester group. Following oral administration, candesartan cilexetil undergoes hydrolysis at the ester link to form the active drug, candesartan, which is achiral.
ATACAND is available for oral use as tablets containing either 4 mg, 8 mg, 16 mg, or 32 mg of candesartan cilexetil and the following inactive ingredients: hydroxypropyl cellulose, polyethylene glycol, lactose, corn starch, carboxymethylcellulose calcium, and magnesium stearate. Ferric oxide (reddish brown) is added to the 8-mg, 16-mg, and 32-mg tablets as a colorant.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Angiotensin II is formed from angiotensin I in a reaction catalyzed by angiotensin-converting enzyme (ACE, kininase II). Angiotensin II is the principal pressor agent of the renin-angiotensin system, with effects that include vasoconstriction, stimulation of synthesis and release of aldosterone, cardiac stimulation, and renal reabsorption of sodium. Candesartan blocks the vasoconstrictor and aldosterone-secreting effects of angiotensin II by selectively blocking the binding of angiotensin II to the AT1 receptor in many tissues, such as vascular smooth muscle and the adrenal gland. Its action is, therefore, independent of the pathways for angiotensin II synthesis.
There is also an AT2 receptor found in many tissues, but AT2 is not known to be associated with cardiovascular homeostasis. Candesartan has much greater affinity (>10,000-fold) for the AT1 receptor than for the AT2 receptor.
Blockade of the renin-angiotensin system with ACE inhibitors, which inhibit the biosynthesis of angiotensin II from angiotensin I, is widely used in the treatment of hypertension. ACE inhibitors also inhibit the degradation of bradykinin, a reaction also catalyzed by ACE. Because candesartan does not inhibit ACE (kininase II), it does not affect the response to bradykinin. Whether this difference has clinical relevance is not yet known. Candesartan does not bind to or block other hormone receptors or ion channels known to be important in cardiovascular regulation.
Blockade of the angiotensin II receptor inhibits the negative regulatory feedback of angiotensin II on renin secretion, but the resulting increased plasma renin activity and angiotensin II circulating levels do not overcome the effect of candesartan on blood pressure.
12.2 Pharmacodynamics
Candesartan inhibits the pressor effects of angiotensin II infusion in a dose-dependent manner. After 1 week of once daily dosing with 8 mg of candesartan cilexetil, the pressor effect was inhibited by approximately 90% at peak with approximately 50% inhibition persisting for 24 hours.
Plasma concentrations of angiotensin I and angiotensin II, and plasma renin activity (PRA), increased in a dose-dependent manner after single and repeated administration of candesartan cilexetil to healthy subjects, hypertensive, and heart failure patients. ACE activity was not altered in healthy subjects after repeated candesartan cilexetil administration. The once-daily administration of up to 16 mg of candesartan cilexetil to healthy subjects did not influence plasma aldosterone concentrations, but a decrease in the plasma concentration of aldosterone was observed when 32 mg of candesartan cilexetil was administered to hypertensive patients. In spite of the effect of candesartan cilexetil on aldosterone secretion, very little effect on serum potassium was observed.
Hypertension
Adults
In multiple-dose studies with hypertensive patients, there were no clinically significant changes in metabolic function, including serum levels of total cholesterol, triglycerides, glucose, or uric acid. In a 12-week study of 161 patients with non-insulin-dependent (type 2) diabetes mellitus and hypertension, there was no change in the level of HbA1c.
Heart Failure
In heart failure patients, candesartan ≥ 8 mg resulted in decreases in systemic vascular resistance and pulmonary capillary wedge pressure.
12.3 Pharmacokinetics
Distribution
The volume of distribution of candesartan is 0.13 L/kg. Candesartan is highly bound to plasma proteins (>99%) and does not penetrate red blood cells. The protein binding is constant at candesartan plasma concentrations well above the range achieved with recommended doses. In rats, it has been demonstrated that candesartan crosses the blood-brain barrier poorly, if at all. It has also been demonstrated in rats that candesartan passes across the placental barrier and is distributed in the fetus.
Metabolism and Excretion
Total plasma clearance of candesartan is 0.37 mL/min/kg, with a renal clearance of 0.19 mL/min/kg. When candesartan is administered orally, about 26% of the dose is excreted unchanged in urine. Following an oral dose of 14C-labeled candesartan cilexetil, approximately 33% of radioactivity is recovered in urine and approximately 67% in feces. Following an intravenous dose of 14C-labeled candesartan, approximately 59% of radioactivity is recovered in urine and approximately 36% in feces. Biliary excretion contributes to the elimination of candesartan.
Adults
Candesartan cilexetil is rapidly and completely bioactivated by ester hydrolysis during absorption from the gastrointestinal tract to candesartan, a selective AT1 subtype angiotensin II receptor antagonist. Candesartan is mainly excreted unchanged in urine and feces (via bile). It undergoes minor hepatic metabolism by O-deethylation to an inactive metabolite. The elimination half-life of candesartan is approximately 9 hours. After single and repeated administration, the pharmacokinetics of candesartan are linear for oral doses up to 32 mg of candesartan cilexetil. Candesartan and its inactive metabolite do not accumulate in serum upon repeated once-daily dosing.
Following administration of candesartan cilexetil, the absolute bioavailability of candesartan was estimated to be 15%. After tablet ingestion, the peak serum concentration (Cmax) is reached after 3 to 4 hours. Food with a high fat content does not affect the bioavailability of candesartan after candesartan cilexetil administration.
Pediatrics
In children 1 to 17 years of age, plasma levels are greater than 10–fold higher at peak (approximately 4 hours) than 24 hours after a single dose.
Children 1 to < 6 years of age, given 0.2 mg/kg had exposure similar to adults given 8 mg.
Children > 6 years of age had exposure similar to adults given the same dose.
The pharmacokinetics (Cmax and AUC) were not modified by age, sex or body weight.
Candesartan cilexetil pharmacokinetics have not been investigated in pediatric patients less than 1 year of age.
From the dose-ranging studies of candesartan cilexetil, there was a dose related increase in plasma candesartan concentrations.
The renin-angiotensin system (RAS) plays a critical role in kidney development. RAS blockade has been shown to lead to abnormal kidney development in very young mice. Children < 1 year of age must not receive ATACAND. Administering drugs that act directly on the renin-angiotensin system (RAS) can alter normal renal development.
Geriatric and Sex
The pharmacokinetics of candesartan have been studied in the elderly (≥ 65 years) and in both sexes. The plasma concentration of candesartan was higher in the elderly (Cmax was approximately 50% higher, and AUC was approximately 80% higher) compared to younger subjects administered the same dose. The pharmacokinetics of candesartan were linear in the elderly, and candesartan and its inactive metabolite did not accumulate in the serum of these subjects upon repeated, once-daily administration. No initial dosage adjustment is necessary [see DOSAGE AND ADMINISTRATION (2)]. There is no difference in the pharmacokinetics of candesartan between male and female subjects.
Renal Insufficiency
In hypertensive patients with renal insufficiency, serum concentrations of candesartan were elevated. After repeated dosing, the AUC and Cmax were approximately doubled in patients with severe renal impairment (creatinine clearance <30 mL/min/1.73m2) compared to patients with normal kidney function. The pharmacokinetics of candesartan in hypertensive patients undergoing hemodialysis are similar to those in hypertensive patients with severe renal impairment. Candesartan cannot be removed by hemodialysis. No initial dosage adjustment is necessary in patients with renal insufficiency [see DOSAGE AND ADMINISTRATION (2.1)].
In heart failure patients with renal impairment, AUC0-72h was 36% and 65% higher in mild and moderate renal impairment, respectively. Cmax was 15% and 55% higher in mild and moderate renal impairment, respectively.
Pediatrics:
ATACAND pharmacokinetics have not been determined in children with renal insufficiency.
Hepatic Insufficiency
The pharmacokinetics of candesartan were compared in patients with mild and moderate hepatic impairment to matched healthy volunteers following a single oral dose of 16 mg candesartan cilexetil. The increase in AUC for candesartan was 30% in patients with mild hepatic impairment (Child-Pugh A) and 145% in patients with moderate hepatic impairment (Child-Pugh B). The increase in Cmax for candesartan was 56% in patients with mild hepatic impairment and 73% in patients with moderate hepatic impairment. The pharmacokinetics after candesartan cilexetil administration have not been investigated in patients with severe hepatic impairment. No initial dosage adjustment is necessary in patients with mild hepatic impairment. In hypertensive patients with moderate hepatic impairment, consideration should be given to initiation of ATACAND at a lower dose [see DOSAGE AND ADMINISTRATION (2.1)].
Heart Failure
The pharmacokinetics of candesartan were linear in patients with heart failure (NYHA class II and III) after candesartan cilexetil doses of 4, 8, and 16 mg. After repeated dosing, the AUC was approximately doubled in these patients compared with healthy, younger patients. The pharmacokinetics in heart failure patients is similar to that in healthy elderly volunteers [see DOSAGE AND ADMINISTRATION (2.3)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
There was no evidence of carcinogenicity when candesartan cilexetil was orally administered to mice and rats for up to 104 weeks at doses up to 100 and 1000 mg/kg/day, respectively. Rats received the drug by gavage, whereas mice received the drug by dietary administration. These (maximally-tolerated) doses of candesartan cilexetil provided systemic exposures to candesartan (AUCs) that were, in mice, approximately 7 times and, in rats, more than 70 times the exposure in man at the maximum recommended daily human dose (32 mg).
Candesartan and its O-deethyl metabolite tested positive for genotoxicity in the in vitro Chinese hamster lung (CHL) chromosomal aberration assay. Neither compound tested positive in the Ames microbial mutagenesis assay or the in vitro mouse lymphoma cell assay. Candesartan (but not its O-deethyl metabolite) was also evaluated in vivo in the mouse micronucleus test and in vitro in the Chinese hamster ovary (CHO) gene mutation assay, in both cases with negative results. Candesartan cilexetil was evaluated in the Ames test, the in vitro mouse lymphoma cell and rat hepatocyte unscheduled DNA synthesis assays and the in vivo mouse micronucleus test, in each case with negative results. Candesartan cilexetil was not evaluated in the CHL chromosomal aberration or CHO gene mutation assay.
Fertility and reproductive performance were not affected in studies with male and female rats given oral doses of up to 300 mg/kg/day (83 times the maximum daily human dose of 32 mg on a body surface area basis).
- 14 CLINICAL STUDIES
-
14.1 Hypertension
Adult
The antihypertensive effects of ATACAND were examined in 14 placebo-controlled trials of 4- to 12-weeks duration, primarily at daily doses of 2 to 32 mg per day in patients with baseline diastolic blood pressures of 95 to 114 mm Hg. Most of the trials were of candesartan cilexetil as a single agent, but it was also studied as add-on to hydrochlorothiazide and amlodipine. These studies included a total of 2350 patients randomized to one of several doses of candesartan cilexetil and 1027 to placebo. Except for a study in diabetics, all studies showed significant effects, generally dose related, of 2 to 32 mg on trough (24 hour) systolic and diastolic pressures compared to placebo, with doses of 8 to 32 mg giving effects of about 8-12/4-8 mm Hg. There were no exaggerated first-dose effects in these patients. Most of the antihypertensive effect was seen within 2 weeks of initial dosing and the full effect in 4 weeks. With once-daily dosing, blood pressure effect was maintained over 24 hours, with trough to peak ratios of blood pressure effect generally over 80%. Candesartan cilexetil had an additional blood pressure lowering effect when added to hydrochlorothiazide.
The antihypertensive effects of candesartan cilexetil and losartan potassium at their highest recommended doses administered once— daily were compared in two randomized, double-blind trials. In a total of 1268 patients with mild to moderate hypertension who were not receiving other antihypertensive therapy, candesartan cilexetil 32 mg lowered systolic and diastolic blood pressure by 2 to 3 mm Hg on average more than losartan potassium 100 mg, when measured at the time of either peak or trough effect. The antihypertensive effects of twice daily dosing of either candesartan cilexetil or losartan potassium were not studied.
The antihypertensive effect was similar in men and women and in patients older and younger than 65. Candesartan was effective in reducing blood pressure regardless of race, although the effect was somewhat less in blacks (usually a low-renin population). This has been generally true for angiotensin II antagonists and ACE inhibitors.
In long-term studies of up to 1 year, the antihypertensive effectiveness of candesartan cilexetil was maintained, and there was no rebound after abrupt withdrawal.
There were no changes in the heart rate of patients treated with candesartan cilexetil in controlled trials.
Pediatric
The antihypertensive effects of ATACAND were evaluated in hypertensive children 1 to < 6 years old and 6 to < 17 years of age in two randomized, double-blind multicenter, 4-week dose ranging studies. There were 93 patients 1 to < 6 years of age, 74% of whom had renal disease, that were randomized to receive an oral dose of candesartan cilexetil suspension 0.05, 0.20 or 0.40 mg/kg once daily. The primary method of analysis was slope of the change in systolic blood pressure (SBP) as a function of dose. Since there was no placebo group, the change from baseline likely overestimates the true magnitude of blood pressure effect. Nevertheless, SBP and diastolic blood pressure (DBP) decreased 6.0/5.2 to 12.0/11.1 mmHg from baseline across the three doses of candesartan.
In children 6 to < 17 years, 240 patients were randomized to receive either placebo or low, medium, or high doses of ATACAND in a ratio of 1: 2: 2: 2. For children who weighed < 50 kg the doses of ATACAND were 2, 8, or 16 mg once daily. For those > 50 kg the ATACAND doses were 4, 16 or 32 mg once daily. Those enrolled were 47% Black and 29% were female; mean age +/- SD was 12.9 +/- 2.6 years.
The placebo subtracted effect at trough for sitting systolic blood pressure/sitting diastolic blood pressure for the different doses were from 4.9/3.0 to 7.5/6.2 mmHg.
In children 6 to < 17 years there was a trend for a lesser blood pressure effect for Blacks compared to other patients. There were too few individuals in the age group of 1 - 6 years old to determine whether Blacks respond differently than other patients to ATACAND.
-
14.2 Heart Failure
Candesartan was studied in two heart failure outcome studies: 1. The Candesartan in Heart failure: Assessment of Reduction in Mortality and morbidity trial in patients intolerant of ACE inhibitors (CHARM–Alternative), 2. CHARM–Added in patients already receiving ACE inhibitors. Both studies were international double-blind, placebo-controlled trials in patients with NYHA class II - IV heart failure and LVEF ≤40%. In both trials, patients were randomized to placebo or ATACAND (initially 4-8 mg once daily, titrated as tolerated to 32 mg once daily) and followed for up to 4 years. Patients with serum creatinine > 3 mg/dL, serum potassium > 5.5 mEq/L, symptomatic hypotension or known bilateral renal artery stenosis were excluded. The primary end point in both trials was time to either cardiovascular death or hospitalization for heart failure.
CHARM–Alternative included 2028 subjects not receiving an ACE inhibitor due to intolerance. The mean age was 67 years and 32% were female, 48% were NYHA II, 49% were NYHA III, 4% were NYHA IV, and the mean ejection fraction was 30%. Sixty-two percent had a history of myocardial infarction, 50% had a history of hypertension, and 27% had diabetes. Concomitant drugs at baseline were diuretics (85%), digoxin (46%), beta-blockers (55%), and spironolactone (24%). The mean daily dose of ATACAND was approximately 23 mg and 59% of subjects on treatment received 32 mg once daily.
After a median follow-up of 34 months, there was a 23% reduction in the risk of cardiovascular death or heart failure hospitalization on ATACAND (p<0.001), with both components contributing to the overall effect (Table 1).
Table 1. CHARM — Alternative: Primary Endpoint and its Components Endpoint (time to first event)
ATACAND (n= 1013)
Placebo (n=1015)
Hazard Ratio (95% CI)
p-value (logrank)
CV death or heart failure hospitalization
334
406
0.77
(0.67–0.89)
<0.001
CV death
219
252
0.85
(0.71–1.02)
0.072
Heart failure hospitalization
207
286
0.68
(0.57–0.81)
<0.001
In CHARM–Added, 2548 subjects receiving an ACE inhibitor were randomized to ATACAND or placebo. The specific ACE inhibitor and dose were at the discretion of the investigators, who were encouraged to titrate patients to doses known to be effective in clinical outcome trials, subject to patient tolerability. Forced titration to maximum tolerated doses of ACE inhibitor was not required.
The mean age was 64 years and 21% were female, 24% were NYHA II, 73% were NYHA III, 3% were NYHA IV, and the mean ejection fraction was 28%. Fifty-six percent had a history of myocardial infarction, 48% had a history of hypertension, and 30% had diabetes. Concomitant drugs at baseline in addition to ACE inhibitors were diuretics (90%), digoxin (58%), beta-blockers (55%), and spironolactone (17%). The mean daily dose of ATACAND was approximately 24 mg and 61% of subjects on treatment received 32 mg once daily.
After a median follow-up of 41 months, there was a 15% reduction in the risk of cardiovascular death or heart failure hospitalization on ATACAND (p=0.011), with both components contributing to the overall effect (Table 2). There was no evident relationship between dose of ACE inhibitor and the benefit of ATACAND.
Table 2. CHARM — Added: Primary Endpoint and its Components Endpoint (time to first event)
ATACAND (n=1276)
Placebo (n=1272)
Hazard Ratio (95% CI)
p-value (logrank)
CV death or heart failure hospitalization
483
538
0.85 (0.75–0.96)
0.011
CV death
302
347
0.84 (0.72–0.98)
0.029
Heart failure hospitalization
309
356
0.83 (0.71–0.96)
0.014
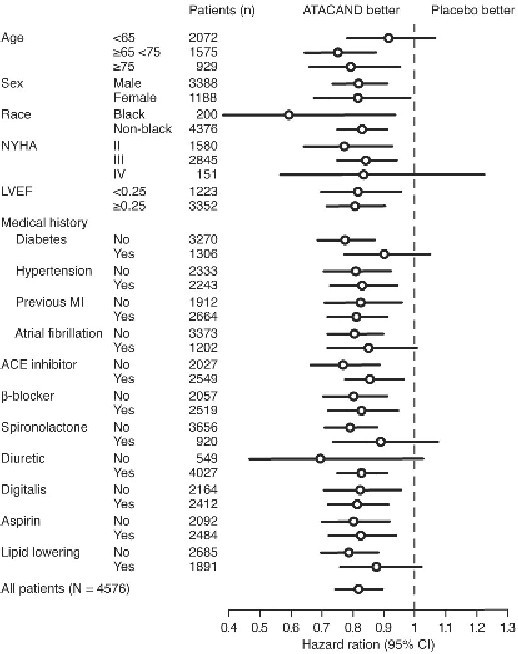
In these two studies, the benefit of ATACAND in reducing the risk of CV death or heart failure hospitalization (18% p<0.001) was evident in major subgroups (see Figure), and in patients on other combinations of cardiovascular and heart failure treatments, including ACE inhibitors and beta-blockers.
Figure. CV Death or Heart Failure Hospitalization in Subgroups – LV Systolic Dysfunction Trials
-
16 HOW SUPPLIED/STORAGE AND HANDLING
No. 3782 — Tablets ATACAND, 4 mg, are white to off-white, circular/biconvex-shaped, non-film-coated scored tablets, coded ACF on one side and 004 on the other. They are supplied as follows:
Bottles of 30
NDC 54868-5591-0
No. 3780 — Tablets ATACAND, 8 mg, are light pink, circular/biconvex-shaped, non-film-coated scored tablets, coded ACG on one side and 008 on the other. They are supplied as follows:
Bottles of 30
NDC 54868-5489-0
No. 3781 — Tablets ATACAND, 16 mg, are pink, circular/biconvex-shaped, non-film-coated scored tablets, coded ACH on one side and 016 on the other. They are supplied as follows:
Bottles of 30
NDC 54868-4413-0
No. 3791 — Tablets ATACAND, 32 mg, are pink, circular/biconvex-shaped, non-film-coated scored tablets, coded ACL on one side and 032 on the other. They are supplied as follows:
Bottles of 30
NDC 54868-4612-0
Storage
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature].Keep container tightly closed.
-
17 PATIENT COUNSELING INFORMATION
Pregnancy – Female patients of childbearing age should be told about the consequences of second- and third-trimester exposure to drugs that act on the renin-angiotensin system, and they should also be told that these consequences do not appear to have resulted from intrauterine drug exposure that has been limited to the first trimester. These patients should be asked to report pregnancies to their physicians as soon as possible.
Post-menarche adolescents should be questioned on a regular basis as to changes in menstrual pattern and the possibility of pregnancy.
Manufactured under the license
from Takeda Pharmaceutical Company, Ltd.
by: AstraZeneca AB, S-151 85 Södertälje, Sweden
for: AstraZeneca LP, Wilmington, DE 19850
ATACAND is a trademark of the AstraZeneca group of companies.
Rev. 04/11
©AstraZeneca 2009
Relabeling and Repackaging by:
Physicians Total Care, Inc.
Tulsa, Oklahoma 74146
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ATACAND
candesartan cilexetil tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-5591(NDC:0186-0004) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CANDESARTAN CILEXETIL (UNII: R85M2X0D68) (CANDESARTAN - UNII:S8Q36MD2XX) CANDESARTAN CILEXETIL 4 mg Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) STARCH, CORN (UNII: O8232NY3SJ) CARBOXYMETHYLCELLULOSE CALCIUM (UNII: UTY7PDF93L) MAGNESIUM STEARATE (UNII: 70097M6I30) FERRIC OXIDE RED (UNII: 1K09F3G675) Product Characteristics Color WHITE (white to off-white) Score 2 pieces Shape ROUND (circular biconvex shaped) Size 7mm Flavor Imprint Code ACF;004 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-5591-0 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020838 05/16/2006 06/30/2011 ATACAND
candesartan cilexetil tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-5489(NDC:0186-0008) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CANDESARTAN CILEXETIL (UNII: R85M2X0D68) (CANDESARTAN - UNII:S8Q36MD2XX) CANDESARTAN CILEXETIL 8 mg Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) STARCH, CORN (UNII: O8232NY3SJ) CARBOXYMETHYLCELLULOSE CALCIUM (UNII: UTY7PDF93L) MAGNESIUM STEARATE (UNII: 70097M6I30) FERRIC OXIDE RED (UNII: 1K09F3G675) Product Characteristics Color PINK ( light pink) Score 2 pieces Shape ROUND (circular biconvex-sharped) Size 7mm Flavor Imprint Code ACG;008 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-5489-0 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020838 05/01/2006 06/30/2011 ATACAND
candesartan cilexetil tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-4413(NDC:0186-0016) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CANDESARTAN CILEXETIL (UNII: R85M2X0D68) (CANDESARTAN - UNII:S8Q36MD2XX) CANDESARTAN CILEXETIL 16 mg Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) STARCH, CORN (UNII: O8232NY3SJ) CARBOXYMETHYLCELLULOSE CALCIUM (UNII: UTY7PDF93L) MAGNESIUM STEARATE (UNII: 70097M6I30) FERRIC OXIDE RED (UNII: 1K09F3G675) Product Characteristics Color PINK Score 2 pieces Shape ROUND (circular biconvex shaped) Size 7mm Flavor Imprint Code ACH;016 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-4413-0 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020838 11/21/2000 ATACAND
candesartan cilexetil tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:54868-4612(NDC:0186-0032) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CANDESARTAN CILEXETIL (UNII: R85M2X0D68) (CANDESARTAN - UNII:S8Q36MD2XX) CANDESARTAN CILEXETIL 32 mg Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOLS (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE (UNII: RFW2ET671P) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) STARCH, CORN (UNII: O8232NY3SJ) CARBOXYMETHYLCELLULOSE CALCIUM (UNII: UTY7PDF93L) MAGNESIUM STEARATE (UNII: 70097M6I30) FERRIC OXIDE RED (UNII: 1K09F3G675) Product Characteristics Color PINK Score 2 pieces Shape ROUND (circular; biconvex-shaped) Size 10mm Flavor Imprint Code ACL;032 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54868-4612-0 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020838 04/17/2002 06/30/2013 Labeler - Physicians Total Care, Inc. (194123980) Establishment Name Address ID/FEI Business Operations Physicians Total Care, Inc. 194123980 repack(54868-5591, 54868-5489, 54868-4612, 54868-4413) , relabel(54868-4413)








