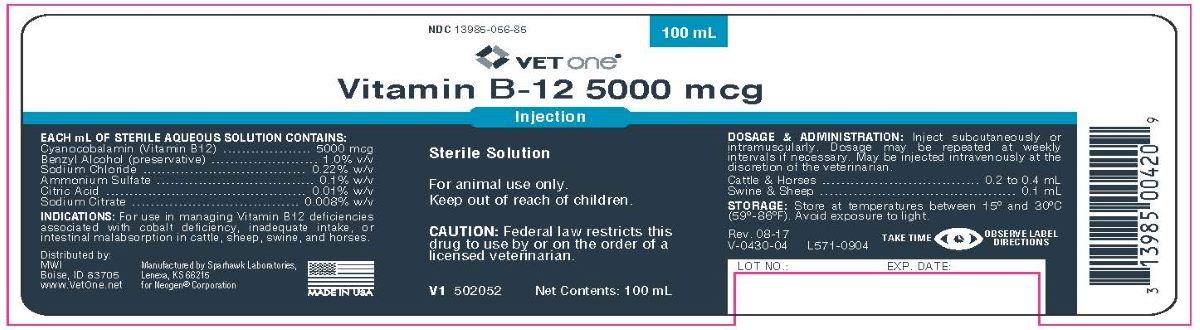
Label: VITAMIN B-12 5000 MCG- cyanocobalamin injection
- NDC Code(s): 13985-056-85
- Packager: MWI/Vet One
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- INDICATIONS:
-
EACH mL OF STERILE AQUEOUS SOLUTION CONTAINS:
Cyanocobalamin (Vitamin B12)..............5000 mcg
Benzyl Alcohol (preservative).................1.0% v/v
Sodium Chloride.................................0.22% w/v
Ammonium Sulfate...............................0.1% w/v
Citric Acid..........................................0.01% w/v
Sodium Citrate..................................0.008% w/v
- DOSAGE & ADMINISTRATION:
- STORAGE:
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
VITAMIN B-12 5000 MCG
cyanocobalamin injectionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:13985-056 Route of Administration INTRAVENOUS, INTRAMUSCULAR, SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) AMMONIUM SULFATE (UNII: SU46BAM238) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) SODIUM CITRATE (UNII: 1Q73Q2JULR) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13985-056-85 100 mL in 1 VIAL, MULTI-DOSE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/01/2017 Labeler - MWI/Vet One (019926120) Registrant - Sparhawk Laboratories, Inc. (147979082) Establishment Name Address ID/FEI Business Operations Sparhawk Laboratories, Inc. 147979082 analysis, manufacture Establishment Name Address ID/FEI Business Operations Hebei Huarong Pharmaceutical Co., Ltd. 529658734 api manufacture