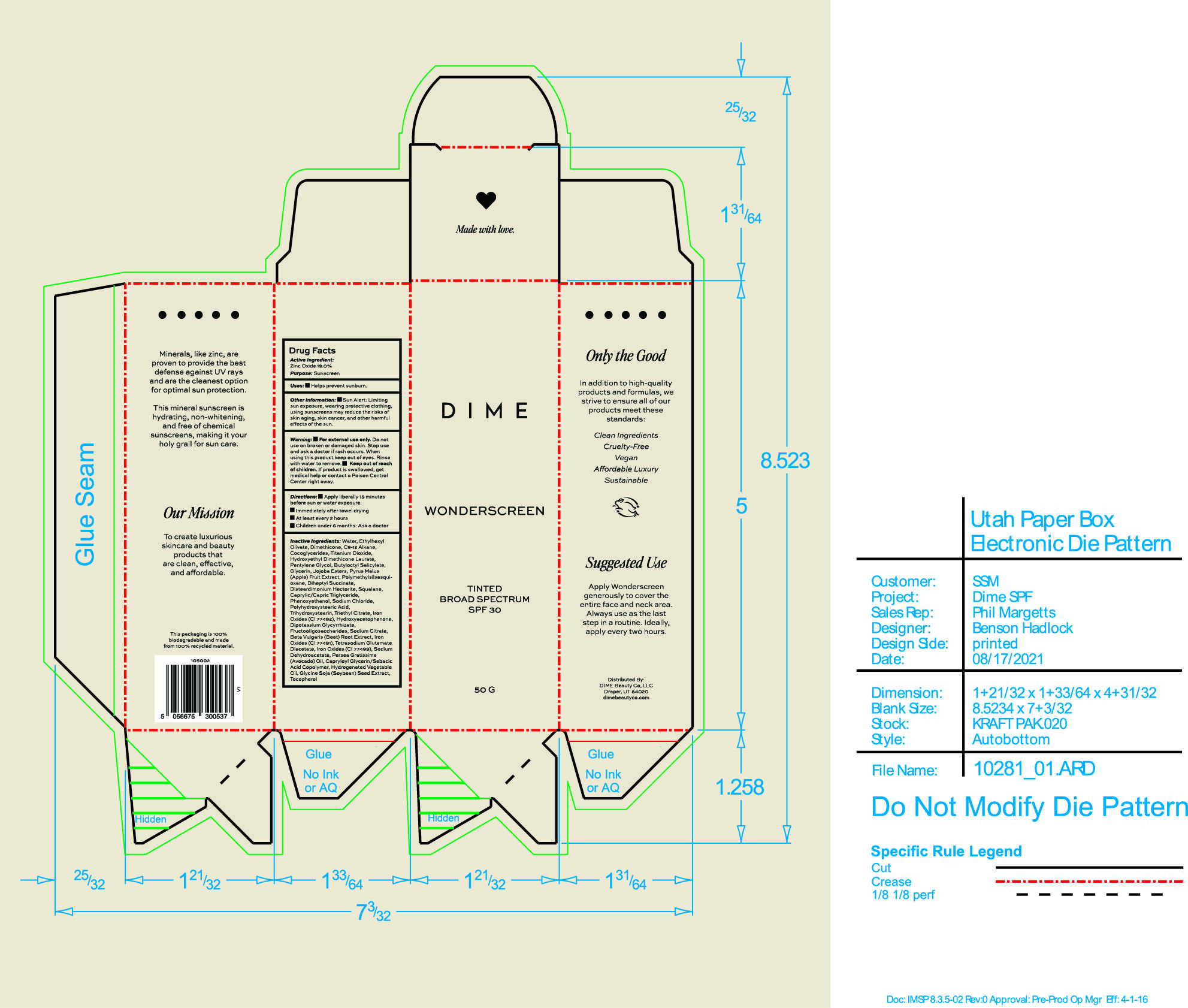
Label: WONDERSCREEN BROAD SPECTRUM SPF 30 SHADE 1- zinc oxide cream
- NDC Code(s): 82870-007-01
- Packager: Dime Beauty Co, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 6, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
-
INACTIVE INGREDIENT
Inactive Ingredients: Water, Ethylhexyl Olivate, Dimethicone, C9-12 Alkane, Cocoglycerides, Titanium Dioxide, Hydroxyethyl Dimethicone Laurate, Pentylene Glycol, Butyloctyl Salicylate, Glycerin, Jojoba Esters, Pyrus Malus (Apple) Fruit Extract, Polymethylsilsesquioxane, Diheptyl Succinate, Disteardimonium Hectorite, Squalane, Caprylic/Capric Triglyceride, Phenoxyethanol, Sodium Chloride, Polyhydroxystearic Acid, Trihydroxystearin, Triethyl Citrate, Iron Oxide (CI 77492), Hydroxyacetophenone, Dipotassium Glycyrrhizate, Fructooligosaccharides, Sodium Citrate, Beta Vulgaris (Beet) Root Extract, Iron Oxides (CI 77491), Tetrasodium Glutamate Diacetate, Iron Oxides (CI 77499), Sodium Dehydroacetate, Persea Gratissima (Avocado) Oil, Capryloyl Glycerin/Sebacic Acid Copolymer, Hydrogenated Vegetable Oil, Glycine Soja (Soybean) Seed Extract, Tocopherol
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
WONDERSCREEN BROAD SPECTRUM SPF 30 SHADE 1
zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82870-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 19 g in 100 g Inactive Ingredients Ingredient Name Strength C9-12 ALKANE (UNII: 7J5R5W72QM) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) SODIUM DEHYDROACETATE (UNII: 8W46YN971G) ETHYLHEXYL OLIVATE (UNII: HTC7G3S2PV) JOJOBA OIL, RANDOMIZED (UNII: 7F0EV20QYL) APPLE (UNII: B423VGH5S9) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AVOCADO OIL (UNII: 6VNO72PFC1) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) TETRASODIUM GLUTAMATE DIACETATE (UNII: 5EHL50I4MY) CAPRYLOYL GLYCERIN/SEBACIC ACID COPOLYMER (2000 MPA.S) (UNII: N7YC58165T) TOCOPHEROL (UNII: R0ZB2556P8) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) FERRIC OXIDE RED (UNII: 1K09F3G675) DIHEPTYL SUCCINATE (UNII: 057N7SS26Y) PHENOXYETHANOL (UNII: HIE492ZZ3T) PENTYLENE GLYCOL (UNII: 50C1307PZG) WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) COCO-GLYCERIDES (UNII: ISE9I7DNUG) TRIHYDROXYSTEARIN (UNII: 06YD7896S3) FERROSOFERRIC OXIDE (UNII: XM0M87F357) BEET (UNII: N487KM8COK) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) GLYCERIN (UNII: PDC6A3C0OX) SQUALANE (UNII: GW89575KF9) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM CHLORIDE (UNII: 451W47IQ8X) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82870-007-01 50 g in 1 BOTTLE; Type 0: Not a Combination Product 10/06/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 10/06/2023 Labeler - Dime Beauty Co, LLC (058459261)