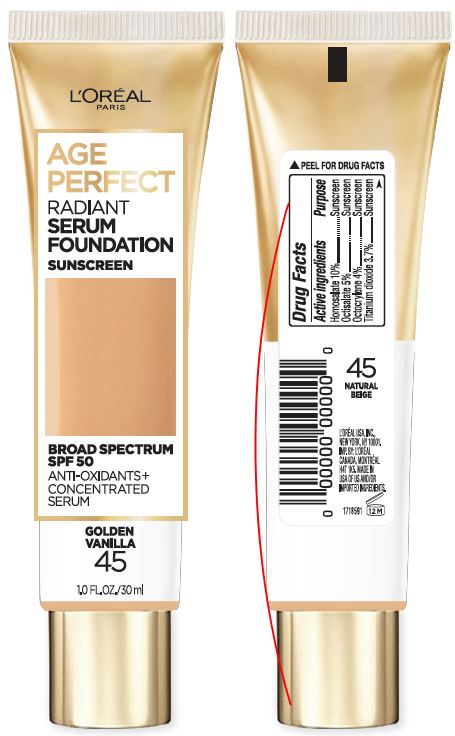
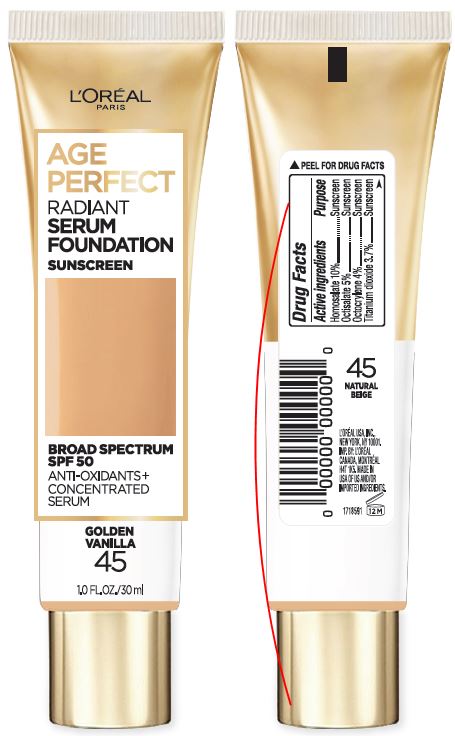
Label: LOREAL PARIS AGE PERFECT RADIANT SERUM FOUNDATION SPF 50- homosalate, octisalate and octocrylene cream
- NDC Code(s): 49967-942-01
- Packager: L'Oreal USA Products Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
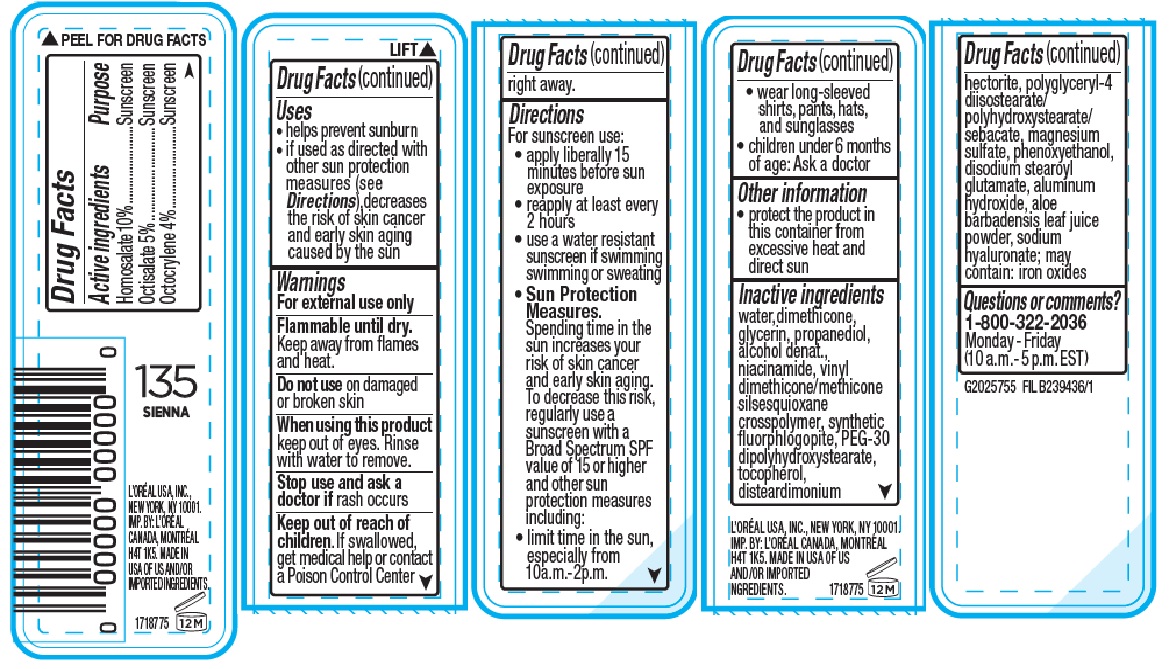
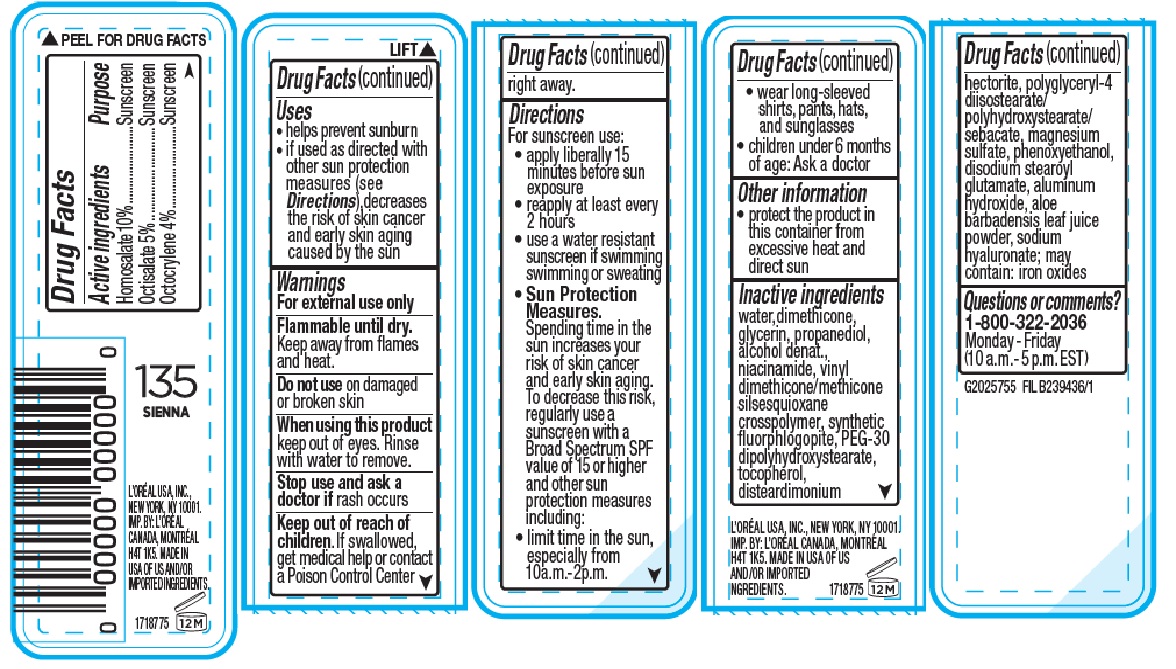
- Active ingredients
- Purpose
- Uses
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
For sunscreen use:
• apply liberally 15 minutes before sun exposure
• reapply at least every 2 hours
• use a water resistant suncreen if swimming or sweating
• Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m. - 2 p.m.
• wear long-sleeved shrits, pants, hat and sunglasses
• children under 6 months of age: Ask a doctor - Other information
-
Inactive ingredients
water, dimethicone, glycerin, propanediol, alcohol denat., niacinamide, vinyl dimethicone/methicone silsesquioxane crosspolymer, synthetic fluorphlogopite, PEG-30 dipolyhydroxystearate, tocopherol, disteardimonium hectorite, polyglyceryl-4 diisostearate/polyhydroxystearate/sebacate, magnesium sulfate, phenoxyethanol, disodium stearoyl glutamate, aluminum hydroxide, aloe barbadensis leaf juice powder, sodium hyaluronate; may contain: iron oxides
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LOREAL PARIS AGE PERFECT RADIANT SERUM FOUNDATION SPF 50
homosalate, octisalate and octocrylene creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49967-942 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 100 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 40 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCERIN (UNII: PDC6A3C0OX) PROPANEDIOL (UNII: 5965N8W85T) ALCOHOL (UNII: 3K9958V90M) NIACINAMIDE (UNII: 25X51I8RD4) VINYL DIMETHICONE/METHICONE SILSESQUIOXANE CROSSPOLYMER (UNII: 9NH1UDD2RR) PEG-30 DIPOLYHYDROXYSTEARATE (UNII: 9713Q0S7FO) TOCOPHEROL (UNII: R0ZB2556P8) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) MAGNESIUM SULFATE, UNSPECIFIED FORM (UNII: DE08037SAB) PHENOXYETHANOL (UNII: HIE492ZZ3T) DISODIUM STEAROYL GLUTAMATE (UNII: 45ASM2L11M) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) ALOE VERA LEAF (UNII: ZY81Z83H0X) HYALURONATE SODIUM (UNII: YSE9PPT4TH) BROWN IRON OXIDE (UNII: 1N032N7MFO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49967-942-01 30 mL in 1 TUBE; Type 0: Not a Combination Product 10/01/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 10/01/2019 Labeler - L'Oreal USA Products Inc (002136794) Establishment Name Address ID/FEI Business Operations L'OREAL USA PRODUCTS, INC. 624244349 manufacture(49967-942) , pack(49967-942)