Label: CIPHERKEY HAIR TONIC- black soybean extracts liquid
- NDC Code(s): 54296-0002-1
- Packager: Jeuncell Co.,Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated September 22, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
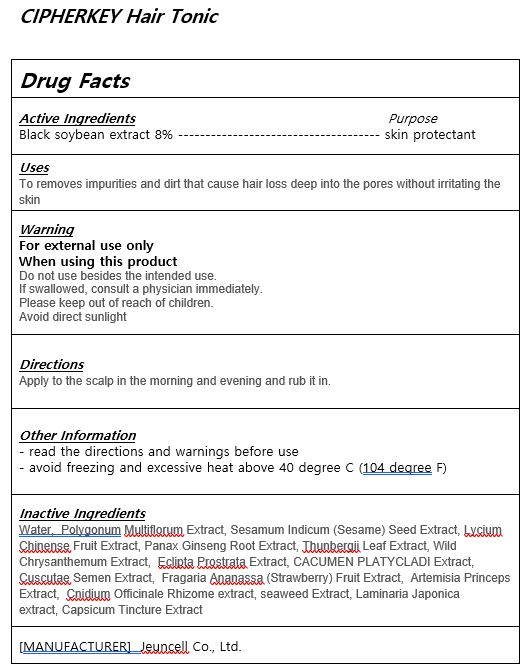
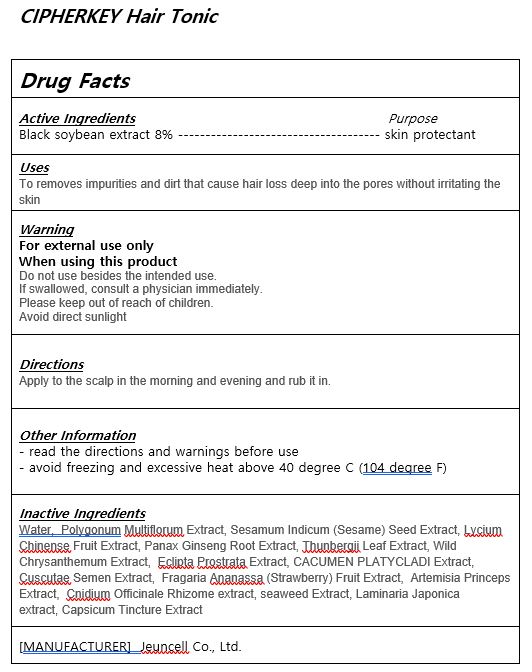
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Water, Polygonum Multiflorum Extract, Sesamum Indicum (Sesame) Seed Extract, Lycium Chinense Fruit Extract, Panax Ginseng Root Extract, Thunbergii Leaf Extract, Wild Chrysanthemum Extract, Eclipta Prostrata Extract, CACUMEN PLATYCLADI Extract,
Cuscutae Semen Extract, Fragaria Ananassa (Strawberry) Fruit Extract, Artemisia Princeps Extract, Cnidium Officinale Rhizome extract, seaweed Extract, Laminaria Japonica extract, Capsicum Tincture Extract - PURPOSE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- Uses
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CIPHERKEY HAIR TONIC
black soybean extracts liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54296-0002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SOYBEAN OIL (UNII: 241ATL177A) (SOYBEAN OIL - UNII:241ATL177A) SOYBEAN OIL 8 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PANAX GINSENG ROOT OIL (UNII: P9T4K47OM0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54296-0002-1 100 mL in 1 CONTAINER; Type 0: Not a Combination Product 09/21/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/21/2023 Labeler - Jeuncell Co.,Ltd (688212024) Registrant - Jeuncell Co.,Ltd (688212024) Establishment Name Address ID/FEI Business Operations Jeuncell Co.,Ltd 688212024 manufacture(54296-0002)