Label: COLD-EEZE COLD REMEDY- zinc gluconate, rosa canina, echinacea purpurea chewable gel
- NDC Code(s): 71179-341-25
- Packager: Vespyr Brands Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated September 14, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
-
Active ingredients
Active ingredients (per lozenge ) . . . . . . . . . . . . . . . . . . . . . . Purposes
Zinc Gluconate 1X HPUS † (8.8%) (13.2 mg zinc).................... Cold Remedy
Rosa Caina (Rose Hips) 2X HPUS † (1.0%) ............................ Promotes immune support*
Echinacea Purpurea 1X HPUS † (0.02%)................................... Promotes immune support*
† The letters ‘HPUS’ indicate that the active ingredient is in the official Homeopathic Pharmacopeia of the United States. - Purposes
- Uses
-
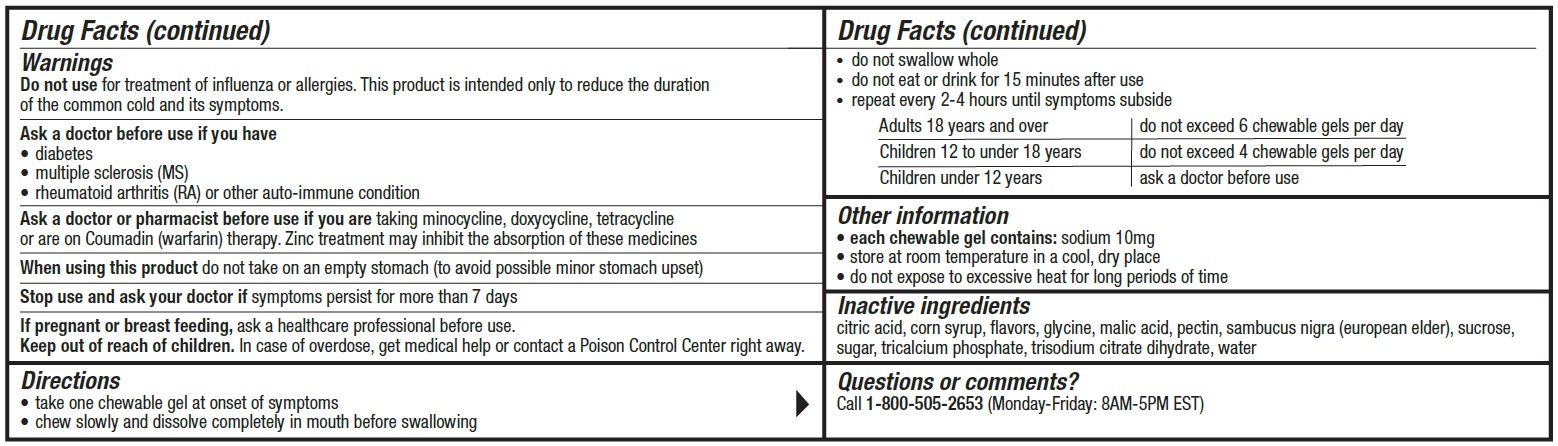
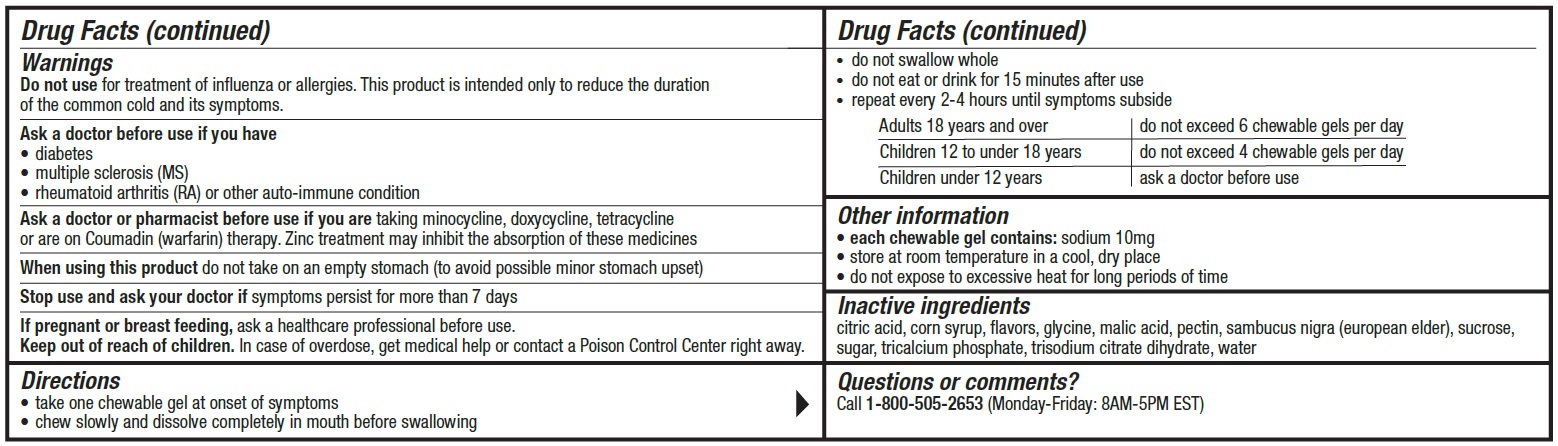
Warnings
Do not use for treatment of influenza or allergies. This product is intended only to reduce the duration of the common cold and provide relief of cold and flu symptoms.
Ask a doctor before use if you have
• diabetes• multiple sclerosis (MS)
• rheumatoid arthritis (RA) or other auto-immune condition
-
Directions
• take one chewable gel at onset of symptoms
• chew slowly and dissolve completely in mouth before swallowing
• do not swallow whole
• do not eat or drink for 15 minutes after use
• repeat every 2-4 hours until symptoms subsideAdults 18 years and over do not exceed 6 chewable gels per day Children 12 to under 18 years do not exceed 4 chewable gels per day Children under 12 years ask a doctor before use - Other information
- Inactive ingredients
- Questions?
-
PRINCIPAL DISPLAY PANEL
Cold-EEZE ®
COLD REMEDY
HOMEOPATHIC
SHORTENS YOUR COLD
Plus DEFENSE*
PROMOTES Immune Health*
NATURAL FLAVOR CITRUS WITH ELDERBERRY
25 Chewable Gels
©2022 Vespyr Brands ®, Inc.
Cold-EEZE ® is a registred trademark of Vespyr Brands ®, Inc.
Distributed by Vespyr Brands ®, Inc.
East Windsor, NJ 08520
This product has not been evaluated by the FDA
*Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.

-
INGREDIENTS AND APPEARANCE
COLD-EEZE COLD REMEDY
zinc gluconate, rosa canina, echinacea purpurea chewable gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71179-341 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ROSA CANINA FRUIT (UNII: 3TNW8D08V3) (ROSA CANINA FRUIT - UNII:3TNW8D08V3) ROSA CANINA FRUIT 2 mg ECHINACEA PURPUREA (UNII: QI7G114Y98) (ECHINACEA PURPUREA - UNII:QI7G114Y98) ECHINACEA PURPUREA 1 mg ZINC GLUCONATE (UNII: U6WSN5SQ1Z) (ZINC CATION - UNII:13S1S8SF37) ZINC GLUCONATE 13.2 mg Inactive Ingredients Ingredient Name Strength SUGARCANE (UNII: 81H2R5AOH3) SAMBUCUS NIGRA FLOWER (UNII: 07V4DX094T) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) GLYCINE (UNII: TE7660XO1C) SUCROSE (UNII: C151H8M554) CORN SYRUP (UNII: 9G5L16BK6N) MALIC ACID (UNII: 817L1N4CKP) TRICALCIUM PHOSPHATE (UNII: K4C08XP666) WATER (UNII: 059QF0KO0R) PECTIN (UNII: 89NA02M4RX) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Product Characteristics Color purple Score Shape ROUND Size 23mm Flavor CITRUS (with Elderberry) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71179-341-25 25 in 1 BOTTLE; Type 0: Not a Combination Product 08/31/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 08/31/2023 Labeler - Vespyr Brands Inc (118094893)