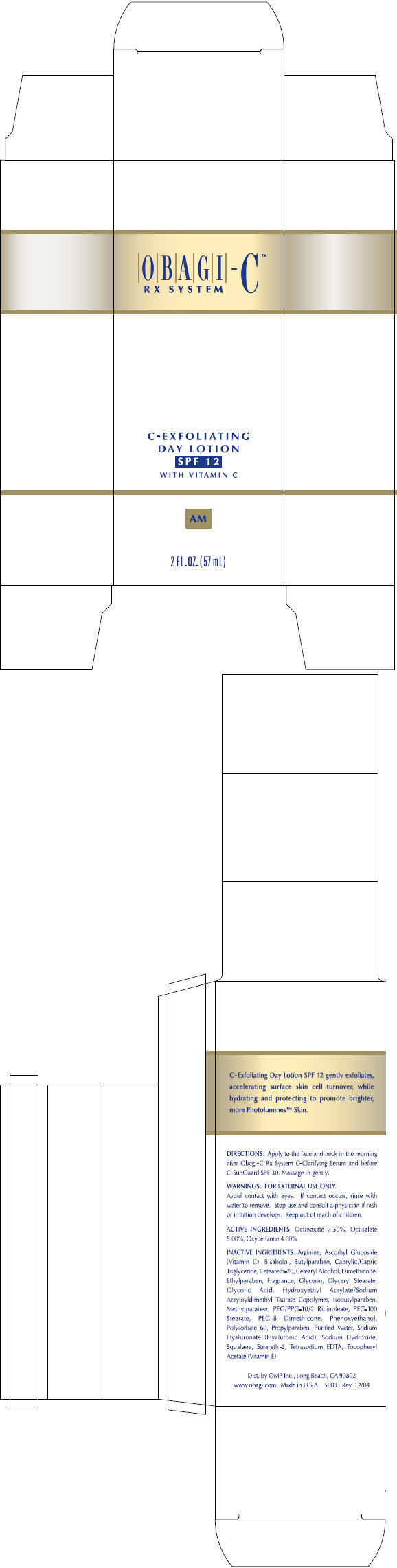
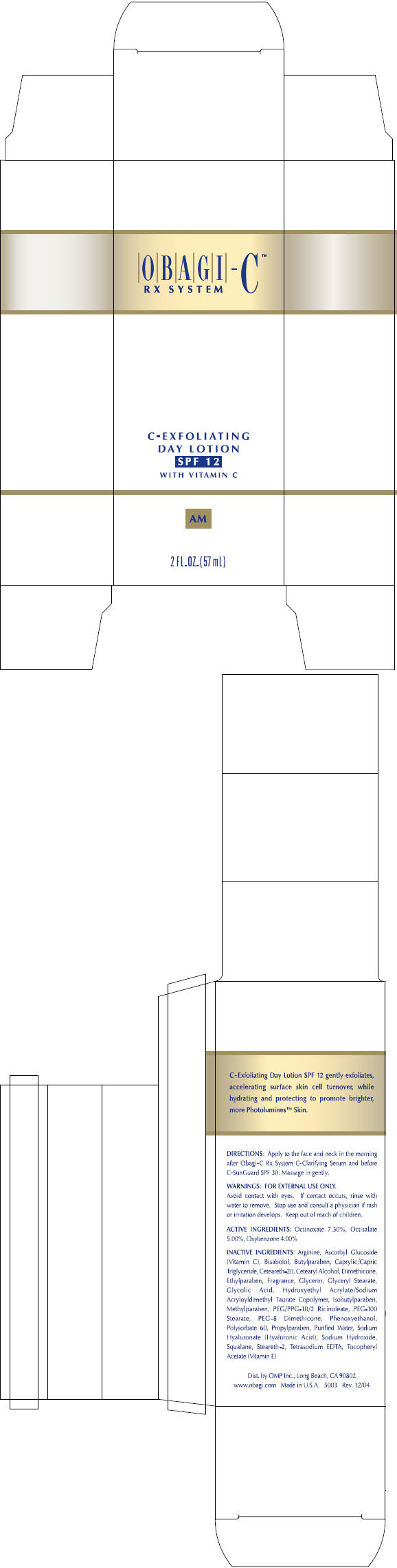
Label: OBAGI-C RX SYSTEM C-EXFOLIATING DAY WITH VITAMIN C- octisalate, octinoxate, and oxybenzone lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 62032-108-36 - Packager: OMP, INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 2, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DIRECTIONS
- WARNINGS
- ACTIVE INGREDIENTS
-
INACTIVE INGREDIENTS
Arginine, Ascorbyl Glucoside (Vitamin C), Bisabolol, Butylparaben, Caprylic/Capric Triglyceride, Ceteareth-20, Cetearyl Alcohol, Dimethicone, Ethylparaben, Fragrance, Glycerin, Glyceryl Stearate, Glycolic Acid, Hydroxyethyl Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Isobutylparaben, Methylparaben, PEG/PPG-10/2 Ricinoleate, PEG-100 Stearate, PEG-8 Dimethicone, Phenoxyethanol, Polysorbate 60, Propylparaben, Purified Water, Sodium Hyaluronate (Hyaluronic Acid), Sodium Hydroxide, Squalane, Steareth-2, Tetrasodium EDTA, Tocopheryl Acetate (Vitamin E)
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 57 mL Carton
-
INGREDIENTS AND APPEARANCE
OBAGI-C RX SYSTEM C-EXFOLIATING DAY WITH VITAMIN C
octisalate, octinoxate, and oxybenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62032-108 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 mL OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 mL Inactive Ingredients Ingredient Name Strength ISOBUTYLPARABEN (UNII: 0QQJ25X58G) STEARETH-2 (UNII: V56DFE46J5) ASCORBYL GLUCOSIDE (UNII: 2V52R0NHXW) LEVOMENOL (UNII: 24WE03BX2T) GLYCERIN (UNII: PDC6A3C0OX) EDETATE DISODIUM (UNII: 7FLD91C86K) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) DIMETHICONE (UNII: 92RU3N3Y1O) ARGININE (UNII: 94ZLA3W45F) WATER (UNII: 059QF0KO0R) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) GLYCOLIC ACID (UNII: 0WT12SX38S) SQUALANE (UNII: GW89575KF9) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) HYALURONATE SODIUM (UNII: YSE9PPT4TH) SODIUM HYDROXIDE (UNII: 55X04QC32I) PHENOXYETHANOL (UNII: HIE492ZZ3T) ETHYLPARABEN (UNII: 14255EXE39) BUTYLPARABEN (UNII: 3QPI1U3FV8) POLYSORBATE 60 (UNII: CAL22UVI4M) PEG-8 DIMETHICONE (UNII: GIA7T764OD) PEG-8 RICINOLEATE (UNII: DM36F4D2OU) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62032-108-36 1 in 1 CARTON 1 57 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 01/01/2004 Labeler - OMP, INC. (790553353) Establishment Name Address ID/FEI Business Operations PURETEK CORPORATION 785961046 MANUFACTURE(62032-108) , LABEL(62032-108) , PACK(62032-108)