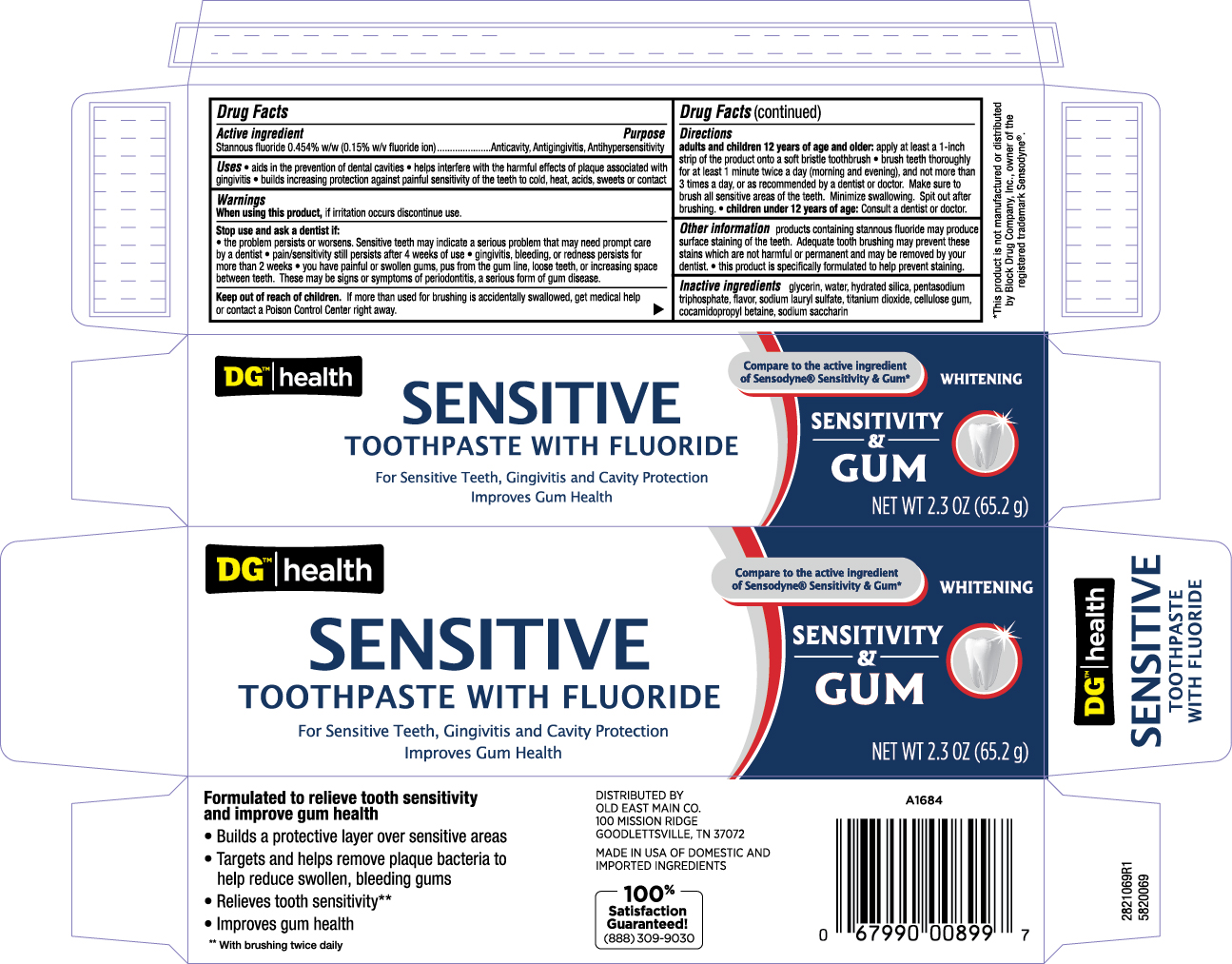
Label: SENSITIVITY AND GUM- stannous fluoride paste, dentifrice
- NDC Code(s): 55910-569-19
- Packager: DG Health (Rexall)
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
When using this product, if irritation occurs discontinue use.
Stop use and ask a dentist if: * the problem persists or worsens. Sensitive teeth may indicate a serious problem that may need prompt care by a dentist * pain/sensitivity still persists after 4 weeks of use * gingivitis, bleeding, or redness persists for more than 2 weeks * you have painful or swollen gums, pus from the gum line, loose teeth, or increasing space between teeth. These may be signs or symptoms of periodontitis, a serious form of gum disease.
- Keep out of reach of children
-
Directions
adults and children 12 years of age and older: apply at least a 1-inch strip of the product unto a soft bristle toothbrush * brush teeth thoroughly for at least 1 minute twice a day (morning and evening), and not more than 3 times a day, or as recommended by a dentist or doctor. Make sure to brush all sensitive areas of the teeth. Minimize swallowing. Spit out after brushing. * children under 12 years of age: consult a dentist or doctor.
- Other information
- Inactive ingredients
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SENSITIVITY AND GUM
stannous fluoride paste, dentifriceProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55910-569 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength STANNOUS FLUORIDE (UNII: 3FTR44B32Q) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.15 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) SACCHARIN SODIUM (UNII: SB8ZUX40TY) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) HYDRATED SILICA (UNII: Y6O7T4G8P9) SODIUM TRIPOLYPHOSPHATE ANHYDROUS (UNII: 9SW4PFD2FZ) SODIUM LAURYL SULFATE (UNII: 368GB5141J) Product Characteristics Color white Score Shape Size Flavor MINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55910-569-19 1 in 1 CARTON 01/11/2024 1 65.2 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M021 01/11/2024 Labeler - DG Health (Rexall) (068331990) Registrant - Lornamead (080046418) Establishment Name Address ID/FEI Business Operations Lornamead 080046418 manufacture(55910-569) , pack(55910-569)