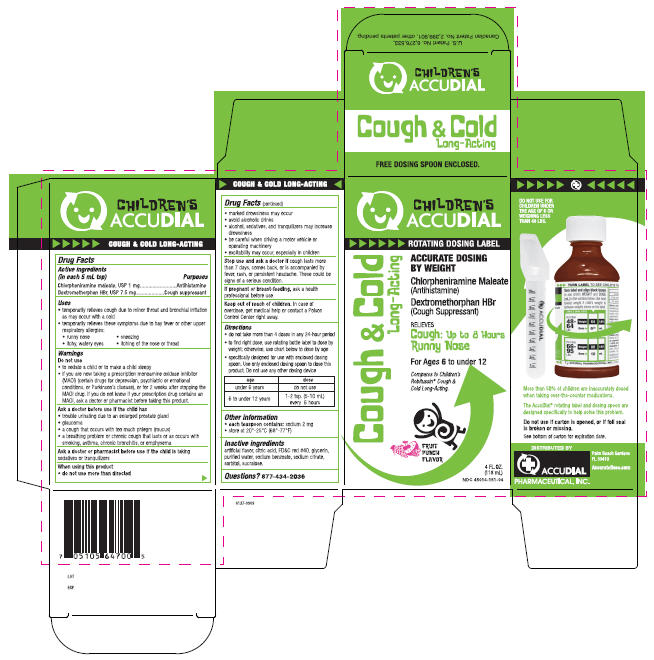
Label: COUGH AND COLD- chlorpheniramine maleate and dextromethorphan hydrobromide liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 45014-151-04 - Packager: Accudial Pharmaceutical, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 11, 2009
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
-
Warnings
Do not use
- to sedate a child or to make a child sleepy
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if the child has
- trouble urinating due to an enlarged prostate gland
- glaucoma
- a cough that occurs with too much phlegm (mucus)
- a breathing problem or chronic cough that lasts or as occurs with smoking, asthma, chronic bronchitis, or emphysema
When using this product
- do not use more than directed
- marked drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
-
Directions
- do not take more than 4 doses in any 24-hour period
- to find right dose, use rotating bottle label to dose by weight; otherwise, use chart below to dose by age
- specifically designed for use with enclosed dosing spoon. Use only enclosed dosing spoon to dose this product. Do not use any other dosing device
age dose under 6 years do not use 6 to under 12 years 1-2 tsp. (5-10 mL) every 6 hours - Other information
- Inactive ingredients
- Questions?
-
PRINCIPAL DISPLAY PANEL - 118 mL Carton
CHILDREN'S
ACCUDIALROTATING DOSING LABEL
Cough & Cold
Long-ActingACCURATE DOSING
BY WEIGHTChlorpheniramine Maleate
(Antihistamine)Dextromethorphan HBr
(Cough Suppressant)RELIEVES
Cough: Up to 8 Hours
Runny NoseFor Ages 6 to under 12
Compares to Children's
Robitussin® Cough &
Cold Long-Acting.FRUIT
PUNCH
FLAVOR4 FL. OZ.
(118 mL)
NDC 45014-151-04
-
INGREDIENTS AND APPEARANCE
COUGH AND COLD
chlorpheniramine maleate and dextromethorphan hydrobromide liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:45014-151 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Chlorpheniramine Maleate (UNII: V1Q0O9OJ9Z) (Chlorpheniramine - UNII:3U6IO1965U) Chlorpheniramine Maleate 1 mg in 5 mL Dextromethorphan Hydrobromide (UNII: 9D2RTI9KYH) (Dextromethorphan - UNII:7355X3ROTS) Dextromethorphan Hydrobromide 7.5 mg in 5 mL Inactive Ingredients Ingredient Name Strength Fruit (UNII: C2AIY4ERZC) Citric Acid Monohydrate (UNII: 2968PHW8QP) FD&C RED NO. 40 (UNII: WZB9127XOA) Glycerin (UNII: PDC6A3C0OX) Water (UNII: 059QF0KO0R) Sodium Benzoate (UNII: OJ245FE5EU) Sodium Citrate (UNII: 1Q73Q2JULR) Sorbitol (UNII: 506T60A25R) Sucralose (UNII: 96K6UQ3ZD4) Product Characteristics Color RED (Reddish-Pink) , PINK Score Shape Size Flavor FRUIT PUNCH Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:45014-151-04 118 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part341 11/25/2009 Labeler - Accudial Pharmaceutical, Inc. (831999201)