Label: NECK DECOLLETE HAND ARM DAY REPARATIVE HYDRATION RESCUE AND REPROGRAM BROAD SPECTRUM SPF 35 SUNSCREEN- neck and decollete day cream
- NDC Code(s): 72414-007-20, 72414-007-60
- Packager: Haute Custom Beauty
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 18, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PACKAGE LABEL
- Product
-
INGREDIENTS AND APPEARANCE
NECK DECOLLETE HAND ARM DAY REPARATIVE HYDRATION RESCUE AND REPROGRAM BROAD SPECTRUM SPF 35 SUNSCREEN
neck and decollete day creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72414-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 3 g in 100 g OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 6 g in 100 g ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 4 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 2.4 g in 100 g Inactive Ingredients Ingredient Name Strength CETEARYL ETHYLHEXANOATE (UNII: 9M64UO4C25) SODIUM HYDROXIDE (UNII: 55X04QC32I) PANTHENOL (UNII: WV9CM0O67Z) DIMETHYLMETHOXY CHROMANOL (UNII: XBH432G01F) DECARBOXY CARNOSINE HYDROCHLORIDE (UNII: 6X7K9I5QR7) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) STEARIC ACID (UNII: 4ELV7Z65AP) XANTHAN GUM (UNII: TTV12P4NEE) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PALMITOYL TRIPEPTIDE-5 (UNII: 2A3916MQHO) PROPANEDIOL (UNII: 5965N8W85T) GLYCERIN (UNII: PDC6A3C0OX) PENTYLENE GLYCOL (UNII: 50C1307PZG) CAPRYLIC/CAPRIC/LAURIC TRIGLYCERIDE (UNII: FJ1H6M2JG9) SESAME OIL (UNII: QX10HYY4QV) CETEARYL ISONONANOATE (UNII: P5O01U99NI) CETEARYL GLUCOSIDE (UNII: 09FUA47KNA) BELLIS PERENNIS (UNII: 2HU33I03UY) CAPRYLYL GLYCOL (UNII: 00YIU5438U) .ALPHA.-TOCOPHEROL ACETATE, D- (UNII: A7E6112E4N) FYTIC ACID (UNII: 7IGF0S7R8I) LIMOSILACTOBACILLUS FERMENTUM (UNII: 2C1F12C6AP) POTASSIUM CETYL PHOSPHATE (UNII: 03KCY6P7UT) ACEFYLLINE METHYLSILANOL MANNURONATE (UNII: OI1T4Z23RZ) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) ALUMINUM OXIDE (UNII: LMI26O6933) VIOLA ODORATA (UNII: AET12U8B74) SHEA BUTTER (UNII: K49155WL9Y) NIACINAMIDE (UNII: 25X51I8RD4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72414-007-60 60 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 08/18/2023 2 NDC:72414-007-20 200 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 08/18/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 08/18/2023 Labeler - Haute Custom Beauty (024442805) Registrant - Haute Custom Beauty (466493781) Establishment Name Address ID/FEI Business Operations Haute Custom Beauty SLU 466493781 manufacture(72414-007) , label(72414-007) , pack(72414-007)


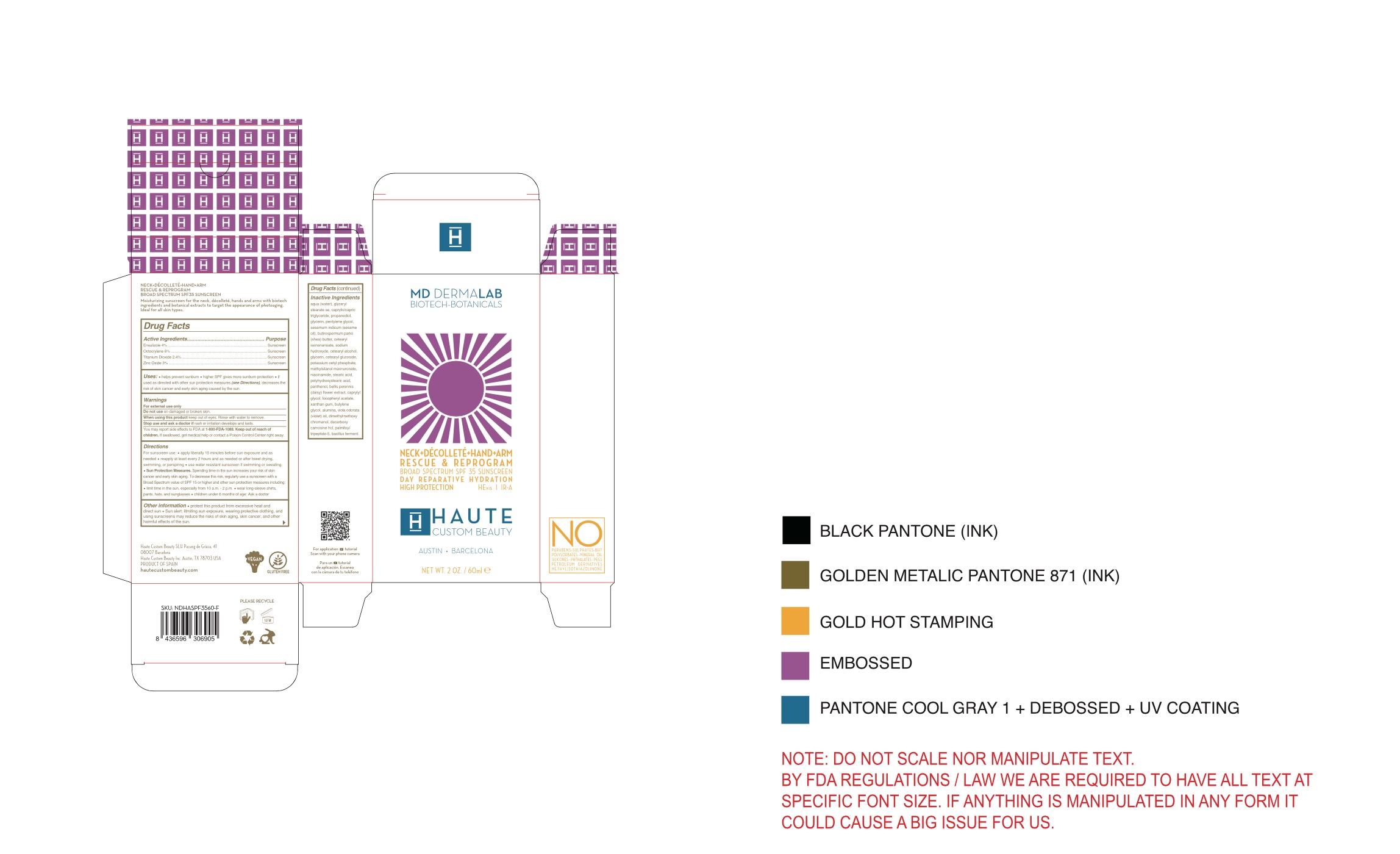 Package Label Principal display panel
Package Label Principal display panel
