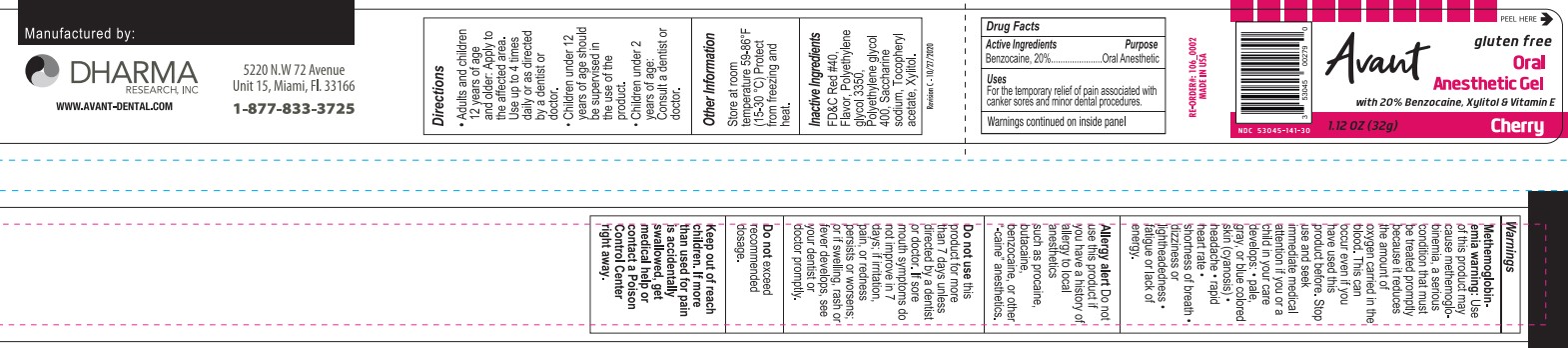
Label: AVANT- benzocaine gel
- NDC Code(s): 53045-141-30
- Packager: Dharma Research, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
-
Warnings
Methemoglobinemia warning
Use of this product may cause methemoglobinemia, a serious condition that must be treated promptly because it reduces the amount of oxygen carried in the blood. This can occur even if you have used this product before.
Stop use and seek medical attention if you or a child in your care develops:
• pale, gray, or blue colored skin (cyanosis)
• headache
• rapid heart rate
• shortness of breath
• dizziness or lightheadedness
• fatigue or lack of energyAlergy alert Do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine, or other "-caine" anesthetics.
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other Information
- Inactive Ingredients
- Avant Oral Anesthetic Gel
-
INGREDIENTS AND APPEARANCE
AVANT
benzocaine gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53045-141 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 6.4 g in 100 g Inactive Ingredients Ingredient Name Strength FD&C RED NO. 40 (UNII: WZB9127XOA) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) SACCHARIN SODIUM (UNII: SB8ZUX40TY) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) XYLITOL (UNII: VCQ006KQ1E) Product Characteristics Color Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53045-141-30 32 g in 1 BOTTLE; Type 0: Not a Combination Product 01/22/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M022 01/22/2021 Labeler - Dharma Research, Inc. (078444642) Establishment Name Address ID/FEI Business Operations Dharma Research, Inc. 078444642 manufacture(53045-141)