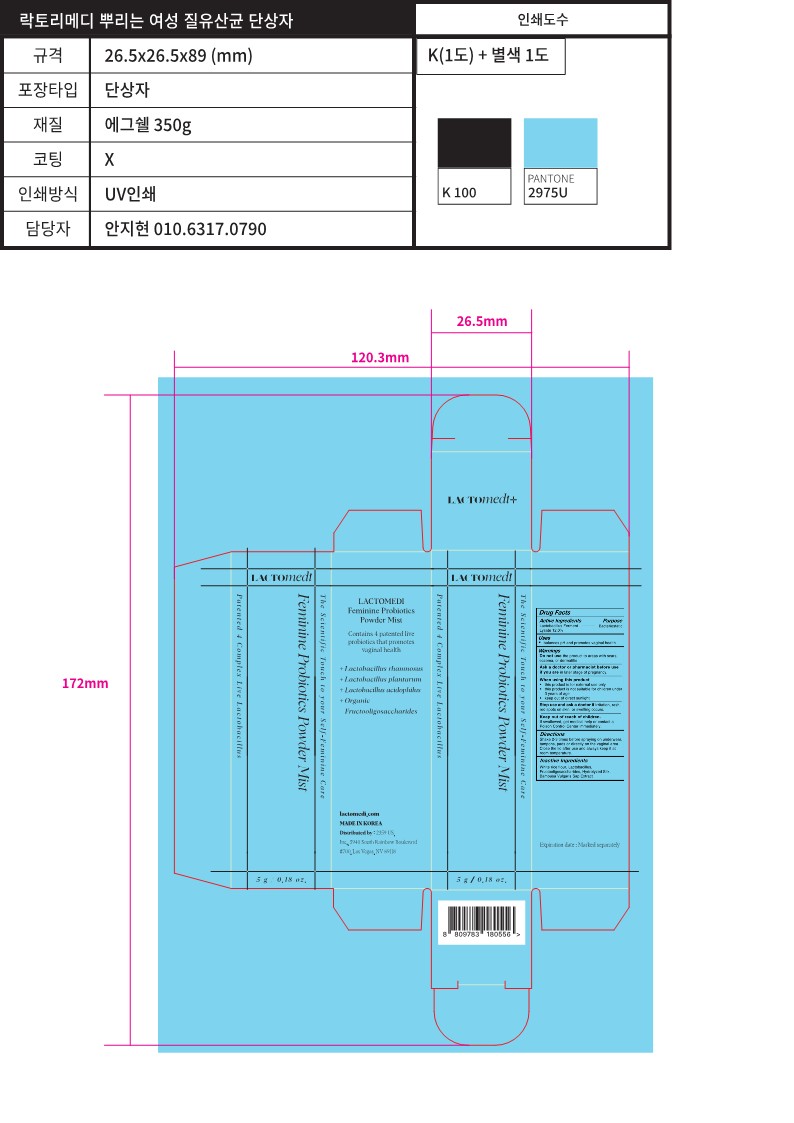
Label: LACTOMEDI FERMININE PROBIOTICS POWDER MIST- lactobacillus ferment lysate spray
- NDC Code(s): 83490-318-01
- Packager: 2359 US INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated July 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purposes
- Uses
- Warnings
- Warnings
- Warnings
- Warnings
-
Warnings
Do not use the product to areas with scars, eczema, or dermatitis.
Ask a doctor or pharmacist before use if you are In later stage of pregnancy.
When using this product
• this product is for external use only
• this product is not suitable for children under 3 years of age
• keep out of direct sunlight
Stop use and ask a doctor if irritation, rash, red spots on skin, or swelling occurs.
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center immediately. - Directions
- Inactive Ingredients
- Label
-
INGREDIENTS AND APPEARANCE
LACTOMEDI FERMININE PROBIOTICS POWDER MIST
lactobacillus ferment lysate sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83490-318 Route of Administration VAGINAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIMOSILACTOBACILLUS FERMENTUM (UNII: 2C1F12C6AP) (LIMOSILACTOBACILLUS FERMENTUM - UNII:2C1F12C6AP) LIMOSILACTOBACILLUS FERMENTUM 12 g in 100 g Inactive Ingredients Ingredient Name Strength BETA VULGARIS ROOT FRUCTOOLIGOSACCHARIDES (UNII: N2UDM48ZD6) SILK, ACID HYDROLYZED (1000 MW) (UNII: 8549W658QV) LACTICASEIBACILLUS RHAMNOSUS (UNII: 9601IVB87J) BAMBUSA VULGARIS SAP (UNII: 26W4035XBO) WHITE RICE (UNII: A195V20H7A) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83490-318-01 5 g in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 08/31/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 08/24/2023 Labeler - 2359 US INC (118974080) Registrant - 2359 US INC (118974080) Establishment Name Address ID/FEI Business Operations Isamogu Inc. 695695834 manufacture(83490-318)