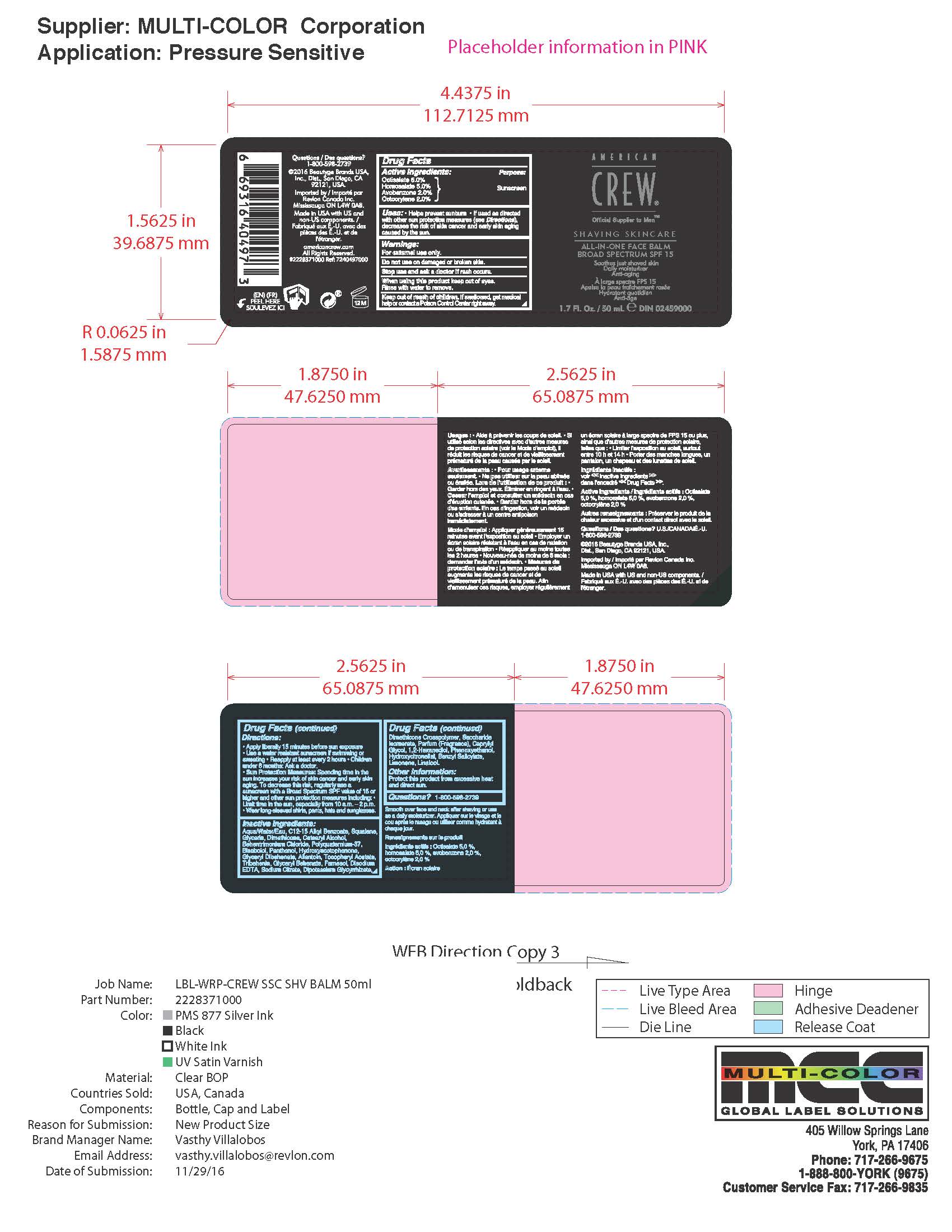
Label: AMERICAN CREW ALL IN ONE FACE BALM BROAD SPECTRUM SPF 15- octisalate,homosalate,avobenzone,octocrylene liquid
- NDC Code(s): 10967-651-24, 10967-651-51
- Packager: Revlon Consumer Products Corp
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 11, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- Directions
-
Sun protection measures
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
•Limit time in the sun, especially from 10 am - 2 pm
•Wear long-sleeved shirts, pants, hats and sunglasses. -
Inactive Ingredients
Aqua/Water/Eau, C12-15 Alkyl Benzoate, Squalane,
Glycerin, Dimethicone, Cetearyl Alcohol, Behentrimonium
Chloride, Polyquaternium-37, Bisabolol, Panthenol,
Hydroxyacetophenone, Glyceryl Dibehenate, Allantoin,
Tocopheryl Acetate, Tribehenin, Glyceryl Behenate,
Farnesol, Disodium EDTA, Sodium Citrate, Dipotassium
Glycyrrhizate, Dimethicone Crosspolymer, Saccharide
Isomerate, Parfum (Fragrance), Caprylyl Glycol,
1,2-Hexanediol, Phenoxyethanol, Hydroxycitronellal, - Questions?
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
AMERICAN CREW ALL IN ONE FACE BALM BROAD SPECTRUM SPF 15
octisalate,homosalate,avobenzone,octocrylene liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10967-651 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 5 mg in 1 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 2 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 2 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) SQUALANE (UNII: GW89575KF9) GLYCERIN (UNII: PDC6A3C0OX) DIMETHICONE (UNII: 92RU3N3Y1O) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) BEHENTRIMONIUM CHLORIDE (UNII: X7GNG3S47T) POLYQUATERNIUM-37 (10000 MPA.S) (UNII: 41QWS48DFN) .ALPHA.-BISABOLOL, (+/-)- (UNII: 36HQN158VC) DEXPANTHENOL (UNII: 1O6C93RI7Z) 4-HYDROXY ACETOPHENONE (UNII: G1L3HT4CMH) GLYCERYL DIBEHENATE (UNII: R8WTH25YS2) ALLANTOIN (UNII: 344S277G0Z) CAPRYLYL GLYCOL (UNII: 00YIU5438U) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) TRIBEHENIN (UNII: 8OC9U7TQZ0) FARNESOL (UNII: EB41QIU6JL) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) PHENOXYETHANOL (UNII: HIE492ZZ3T) SACCHARIDE ISOMERATE (UNII: W8K377W98I) HYDROXYCITRONELLAL (UNII: 8SQ0VA4YUR) BENZYL SALICYLATE (UNII: WAO5MNK9TU) LIMONENE, (+)- (UNII: GFD7C86Q1W) LINALOOL, (+/-)- (UNII: D81QY6I88E) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) EUGENOL (UNII: 3T8H1794QW) GERANIOL (UNII: L837108USY) BENZYL BENZOATE (UNII: N863NB338G) CITRAL (UNII: T7EU0O9VPP) CINNAMYL ALCOHOL (UNII: SS8YOP444F) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10967-651-51 170 mL in 1 PACKAGE; Type 0: Not a Combination Product 04/01/2017 2 NDC:10967-651-24 50 mL in 1 PACKAGE; Type 0: Not a Combination Product 04/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 04/01/2017 Labeler - Revlon Consumer Products Corp (788820165) Establishment Name Address ID/FEI Business Operations REVLON, INC. 809725570 manufacture(10967-651)