Label: SHOPKO ABSOLUTE DAILY MOISTURIZING BROAD SPECTRUM SPF15 SUNSCREEN- avobenzone, homosalate, octinoxate, octocrylene cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 37012-626-11 - Packager: Shopko Stores Operating Co., LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 6, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
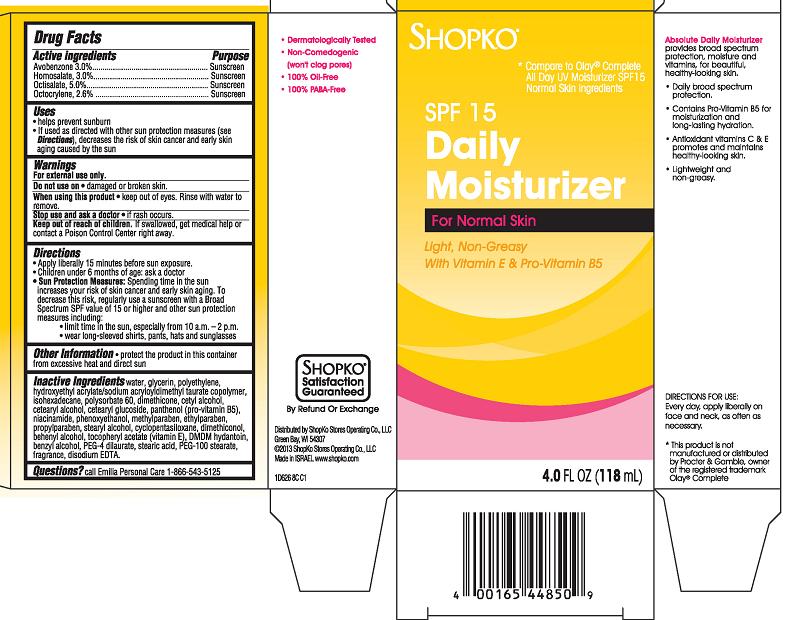
- Active ingredient
- Purpose
- Uses
- Warnings
- Do not use on
- When using this product
- Stop use and ask a doctor
- Keep Out of Reach of Children.
-
Directions
- Apply liberally 15 minutes before sun exposure.
- Children under 6 months of age: ask a doctor
-
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- Other Information
-
Inactive Ingredients
water, glycerin, polyethylene, hydroxyethyl acrylate/sodium acryloyldimethyl taurate copolymer, isohexadecane, polysorbate 60, dimethicone, cetyl alcohol, cetearyl alcohol, cetearyl glucoside, panthenol (pro-vitamin B5), niacinamide, phenoxythanol, methylparaben, ethylparaben, propylparaben, stearyl alcohol, cyclopentasiloxane, dimethiconol, behenyl alcohol, tocopheryl acetate (vitamin E), DMDM hydantoin, benzyl alcohol, PEG-4 dilaurate, stearic acid, PEG-100 stearate, fragrance, disodium EDTA
-
Package/Label Principal Display Panel - Carton Label
Shopko*
*Compare to Olay® Complete All Day UV Moisturizer SPF15 Normal Skin Ingredients
SPF 15 Daily Moisturizer For Normal Skin
Light, Non-Greasy
With Vitamin E & Pro-Vitamin B5- Dermatologically Tested
- Non-Comedogenic (won't clog pores)
- 100% Oil-Free
- 100% PABA-Free
Absolute Daily Moisturizer provides broad spectrum protection, moisture and vitamins, for beautiful, health-looking skin.- Daily broad spectrum protection.
- Contains Pro-Vitamin B5 for moisturization and long-lasting hydration.
- Antioxidant vitamins C & E promotes and maintains healthy-looking skin.
- Lightweight and non-greasy.
DIRECTIONS FOR USE: Every day, apply liberally on face and neck, as often as necessary.
*This product is not manufactured or distributed by Procter & Gamble, owner of the registered trademark Olay® Complete
4 FL OZ (118 mL)
Distributed by ShopKo Stores Operating Co., LLC
Green Bay, WI 54307
© 2013 ShopKo Stores Operating Co., LLC
Made in Israel. www.shopko.com1D626 8C C1
Carton Label
-
INGREDIENTS AND APPEARANCE
SHOPKO ABSOLUTE DAILY MOISTURIZING BROAD SPECTRUM SPF15 SUNSCREEN
avobenzone, homosalate, octinoxate, octocrylene creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:37012-626 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 mg in 100 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 3 mg in 100 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 5 mg in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 2.6 mg in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) HYDROXYETHYL ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER (45000 MPA.S AT 1%) (UNII: 86FQE96TZ4) ISOHEXADECANE (UNII: 918X1OUF1E) POLYSORBATE 60 (UNII: CAL22UVI4M) DIMETHICONE (UNII: 92RU3N3Y1O) CETYL ALCOHOL (UNII: 936JST6JCN) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) CETEARYL GLUCOSIDE (UNII: 09FUA47KNA) PANTHENOL (UNII: WV9CM0O67Z) NIACINAMIDE (UNII: 25X51I8RD4) PHENOXYETHANOL (UNII: HIE492ZZ3T) METHYLPARABEN (UNII: A2I8C7HI9T) ETHYLPARABEN (UNII: 14255EXE39) PROPYLPARABEN (UNII: Z8IX2SC1OH) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) DIMETHICONOL (100000 CST) (UNII: OSA9UP217S) DOCOSANOL (UNII: 9G1OE216XY) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) DMDM HYDANTOIN (UNII: BYR0546TOW) BENZYL ALCOHOL (UNII: LKG8494WBH) PEG-4 DILAURATE (UNII: KCR71CW036) STEARIC ACID (UNII: 4ELV7Z65AP) PEG-100 STEARATE (UNII: YD01N1999R) EDETATE DISODIUM (UNII: 7FLD91C86K) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:37012-626-11 1 in 1 CARTON 1 118 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 06/01/2013 Labeler - Shopko Stores Operating Co., LLC (023252638)