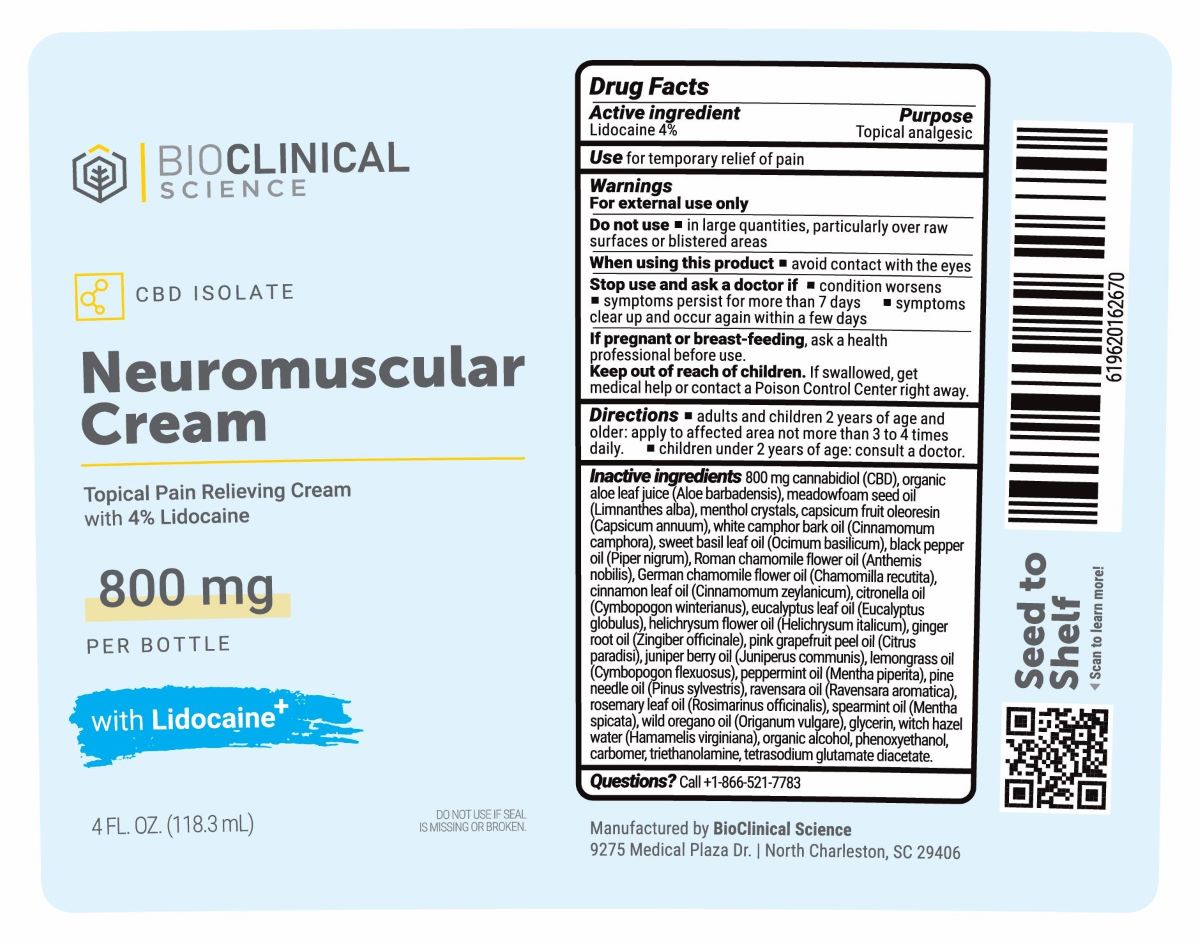
Label: NEUROMUSCULAR CREAM- lidocaine 4% cream
- NDC Code(s): 82984-0001-2, 82984-0001-4
- Packager: BioClinical Science, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- 59.2 mL Active Ingredient
- 59.2 mL Purpose Section
- 59.2 mL Indications and Usage Section
- 59.2 mL Warnings Section
- 59.2 mL Do Not Use Section
- 59.2 mL When Using Section
- 59.2 mL Stop Use Section
- 59.2 mL Ask Doctor Section
- 59.2 mL Pregnancy or Breast Feeding Section
- 59.2 mL Keep out of reach of children section
- 59.2 mL Dosage and Administration Section
- 59.2 mL Inactive Ingredient Section
- 59.2 mL Questions Section
- 118.3 mL Active Ingredient Section
- 118.3 mL Purpose Section
- 118.3 mL Indications and Usage Section
- 118.3 mL Warnings Section
- 118.3 mL Do Not Use Section
- 118.3 mL When Using Section
- 118.3 mL Stop Use Section
- 118.3 mL Ask Doctor Section
- 118.3 mL Pregnancy or Breast Feeding Section
- 118.3 mL Keep out of reach of children section
- 118.3 mL Dosage and Administration Section
- 118.3 mL Inactive ingredient section
- 118.3 mL Questions section
- 59.2 mL Bottle
- 118.3 mL Bottle
-
INGREDIENTS AND APPEARANCE
NEUROMUSCULAR CREAM
lidocaine 4% creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82984-0001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 4 g in 100 g Inactive Ingredients Ingredient Name Strength SPEARMINT OIL (UNII: C3M81465G5) HAMAMELIS VIRGINIANA LEAF WATER (UNII: 8FP93ED6H2) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERIN (UNII: PDC6A3C0OX) CAMPHOR OIL (UNII: 75IZZ8Y727) ROSEMARY OIL (UNII: 8LGU7VM393) EAST INDIAN LEMONGRASS OIL (UNII: UP0M8M3VZW) MENTHOL (UNII: L7T10EIP3A) CHAMAEMELUM NOBILE FLOWER OIL (UNII: UB27587839) CITRONELLA OIL (UNII: QYO8Q067D0) HELICHRYSUM ITALICUM FLOWER OIL (UNII: O97ZV7726K) JUNIPER BERRY OIL (UNII: SZH16H44UY) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) PEPPERMINT OIL (UNII: AV092KU4JH) CRYPTOCARYA AGATHOPHYLLA LEAF OIL (UNII: XM00Z00H98) EUCALYPTUS OIL (UNII: 2R04ONI662) ALOE (UNII: V5VD430YW9) CAPSICUM OLEORESIN (UNII: UW86K581WY) PINE NEEDLE OIL (PINUS SYLVESTRIS) (UNII: 5EXL5H740Y) BASIL OIL (UNII: Z129UMU8LE) TROLAMINE (UNII: 9O3K93S3TK) BLACK PEPPER OIL (UNII: U17J84S19Z) GRAPEFRUIT OIL (UNII: YR377U58W9) OREGANO LEAF OIL (UNII: 7D0CGR40U1) CANNABIDIOL (UNII: 19GBJ60SN5) CINNAMON LEAF OIL (UNII: S92U8SQ71V) TETRASODIUM GLUTAMATE DIACETATE (UNII: 5EHL50I4MY) CHAMOMILE FLOWER OIL (UNII: 60F80Z61A9) ALCOHOL (UNII: 3K9958V90M) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) GINGER OIL (UNII: SAS9Z1SVUK) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82984-0001-2 59.2 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 10/12/2023 2 NDC:82984-0001-4 118.3 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 10/12/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 10/12/2023 Labeler - BioClinical Science, LLC (117606575) Establishment Name Address ID/FEI Business Operations BioClinical Science, LLC 117606575 manufacture(82984-0001)