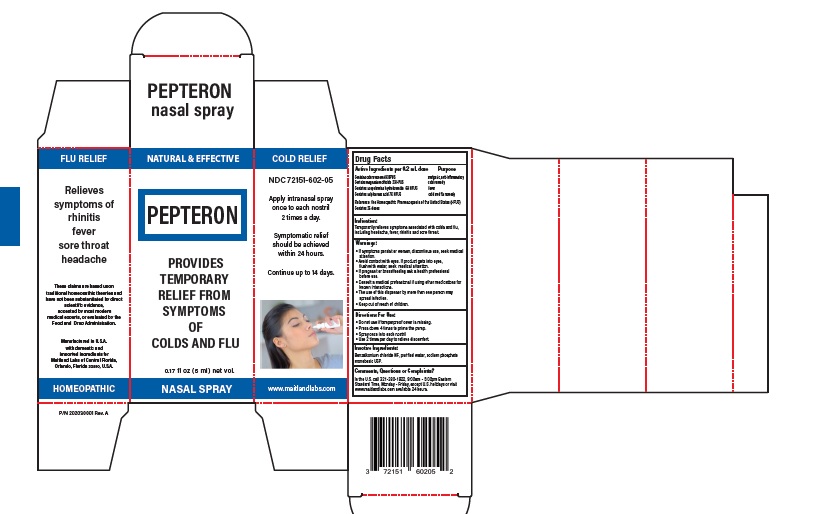
Label: PEPTERON- homeopathic nasal spray spray, metered
-
Contains inactivated NDC Code(s)
NDC Code(s): 72151-602-05 - Packager: Maitland Labs of Central Florida
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 26, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients per dose Purpose
-
Warning
- If symptoms persist or worsen, discontinue use, seek medical attention
- Avoid contact with eyes. If product gets into eyes,flush with water, seek medical attention
- If pregnant or breastfeeding ask a health professional before use
- Consult a medical professional if using other medications for known interactions
- The use of this dispenser by more than one person may spread infection.
- keep out of reach of children.
- Purpose
- Indication
- Directions For Use
- KEEP OUT OF REACH OF CHILDREN
- Comments, Questions or Complaints?
- Inactive ingredient section
- Product Label
-
INGREDIENTS AND APPEARANCE
PEPTERON
homeopathic nasal spray spray, meteredProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72151-602 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NAJA NAJA WHOLE (UNII: D279BDR31E) (NAJA NAJA WHOLE - UNII:D279BDR31E) NAJA NAJA WHOLE 4 [hp_X] in 0.2 mL MAGNESIUM CHLORIDE (UNII: 02F3473H9O) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM CHLORIDE 2 [hp_X] in 0.2 mL SCOPOLAMINE HYDROBROMIDE (UNII: 451IFR0GXB) (SCOPOLAMINE - UNII:DL48G20X8X) SCOPOLAMINE HYDROBROMIDE 6 [hp_X] in 0.2 mL SULFUROUS ACID (UNII: J1P7893F4J) (SULFUROUS ACID - UNII:J1P7893F4J) SULFUROUS ACID 7 [hp_X] in 0.2 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) WATER (UNII: 059QF0KO0R) SODIUM PHOSPHATE, MONOBASIC, ANHYDROUS (UNII: KH7I04HPUU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72151-602-05 5 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/26/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/26/2020 Labeler - Maitland Labs of Central Florida (080974960) Establishment Name Address ID/FEI Business Operations Maitland Labs of Central Florida 080974960 manufacture(72151-602)