Label: MEKINIST- trametinib tablet, film coated
MEKINIST- trametinib powder, for solution
- NDC Code(s): 0078-1105-15, 0078-1112-15, 0078-1112-94, 0078-1161-47
- Packager: Novartis Pharmaceuticals Corporation
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated December 3, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use MEKINIST safely and effectively. See full prescribing information for MEKINIST.
MEKINIST® (trametinib) tablets, for oral use
MEKINIST® (trametinib) for oral solution
Initial U.S. Approval: 2013RECENT MAJOR CHANGES
INDICATIONS AND USAGE
MEKINIST is a kinase inhibitor indicated as a single agent for the treatment of BRAF-inhibitor treatment-naïve patients with unresectable or metastatic melanoma with BRAF V600E or V600K mutations as detected by an FDA-approved test. (1.1, 2.1)
MEKINIST is indicated, in combination with dabrafenib, for:
- the treatment of patients with unresectable or metastatic melanoma with BRAF V600E or V600K mutations as detected by an FDA-approved test. (1.1, 2.1)
- the adjuvant treatment of patients with melanoma with BRAF V600E or V600K mutations, as detected by an FDA-approved test, and involvement of lymph node(s), following complete resection. (1.2, 2.1)
- the treatment of patients with metastatic non-small cell lung cancer (NSCLC) with BRAF V600E mutation as detected by an FDA-approved test. (1.3, 2.1)
- the treatment of patients with locally advanced or metastatic anaplastic thyroid cancer (ATC) with BRAF V600E mutation, as detected by an FDA-approved test, and with no satisfactory locoregional treatment options. (1.4, 2.1)
- the treatment of adult and pediatric patients 1 year of age and older with unresectable or metastatic solid tumors with BRAF V600E mutation who have progressed following prior treatment and have no satisfactory alternative treatment options. This indication is approved under accelerated approval based on overall response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s). (1.5, 2.1)
- the treatment of pediatric patients 1 year of age and older with low-grade glioma (LGG) with a BRAF V600E mutation who require systemic therapy. (1.6, 2.1)
Limitations of Use: MEKINIST is not indicated for treatment of patients with colorectal cancer because of known intrinsic resistance to BRAF inhibition. (1.7, 12.1)
DOSAGE AND ADMINISTRATION
- The recommended dosage of MEKINIST in adult patients is 2 mg orally once daily. The recommended dosage for MEKINIST in pediatric patients is based on body weight. (2)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- New Primary Malignancies, Cutaneous and Non-Cutaneous: Can occur when MEKINIST is used with dabrafenib. Monitor patients for new malignancies prior to, or while on therapy, and following discontinuation of treatment. (5.1)
- Hemorrhage: Major hemorrhagic events can occur. Monitor for signs and symptoms of bleeding. (5.2)
- Colitis and Gastrointestinal Perforation: Colitis and gastrointestinal perforation can occur in patients receiving MEKINIST. (5.3)
- Venous Thromboembolic Events: Deep vein thrombosis (DVT) and pulmonary embolism (PE) can occur in patients receiving MEKINIST. (5.4, 2.4)
- Cardiomyopathy: Assess left ventricular ejection fraction (LVEF) before treatment, after one month of treatment, then every 2 to 3 months thereafter. (5.5, 2.4)
- Ocular Toxicities: Perform ophthalmological evaluation for any visual disturbances. For Retinal Vein Occlusion (RVO), permanently discontinue MEKINIST. (5.6, 2.4)
- Interstitial Lung Disease (ILD)/Pneumonitis: Withhold MEKINIST for new or progressive unexplained pulmonary symptoms. Permanently discontinue MEKINIST for treatment-related ILD or pneumonitis. (5.7, 2.4)
- Serious Febrile Reactions: Can occur when MEKINIST is used with dabrafenib. (5.8, 2.4)
- Serious Skin Toxicities: Monitor for skin toxicities and for secondary infections. Permanently discontinue MEKINIST for intolerable Grade 2 or for Grade 3 or 4 rash not improving within 3 weeks despite interruption of MEKINIST. Permanently discontinue for severe cutaneous adverse reactions (SCARs). (5.9, 2.4)
- Hyperglycemia: Monitor serum glucose levels in patients with preexisting diabetes or hyperglycemia. (5.10)
- Hemophagocytic Lymphohistiocytosis (HLH): Interrupt treatment for suspected HLH. Discontinue treatment if HLH is confirmed. (5.12)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of potential risk to a fetus and to use effective contraception. (5.13, 8.1, 8.3)
ADVERSE REACTIONS
Most common adverse reactions (≥ 20%) for MEKINIST as a single agent include rash, diarrhea, and lymphedema. (6.1)
Most common adverse reactions (≥ 20%) for MEKINIST in combination with dabrafenib include:
- Unresectable or metastatic melanoma: pyrexia, nausea, rash, chills, diarrhea, vomiting, hypertension, and peripheral edema. (6.1)
- Adjuvant treatment of melanoma: pyrexia, fatigue, nausea, headache, rash, chills, diarrhea, vomiting, arthralgia, and myalgia. (6.1)
- NSCLC: pyrexia, fatigue, nausea, vomiting, diarrhea, dry skin, decreased appetite, edema, rash, chills, hemorrhage, cough, and dyspnea. (6.1)
- Adult patients with solid tumors: pyrexia, fatigue, nausea, rash, chills, headache, hemorrhage, cough, vomiting, constipation, diarrhea, myalgia, arthralgia, and edema. (6.1)
- Pediatric patients with solid tumors: pyrexia, rash, vomiting, fatigue, dry skin, cough, diarrhea, dermatitis acneiform, headache, abdominal pain, nausea, hemorrhage, constipation, and paronychia. (6.1)
- Pediatric patients with LGG: pyrexia, rash, headache, vomiting, musculoskeletal pain, fatigue, diarrhea, dry skin, nausea, hemorrhage, abdominal pain, and dermatitis acneiform. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Novartis Pharmaceuticals Corporation at 1-888-669-6682 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 4/2025
- the treatment of patients with unresectable or metastatic melanoma with BRAF V600E or V600K mutations as detected by an FDA-approved test. (1.1, 2.1)
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 BRAF V600E or V600K Mutation-Positive Unresectable or Metastatic Melanoma
1.2 Adjuvant Treatment of BRAF V600E or V600K Mutation-Positive Melanoma
1.3 BRAF V600E Mutation-Positive Metastatic NSCLC
1.4 BRAF V600E Mutation-Positive Locally Advanced or Metastatic Anaplastic Thyroid Cancer
1.5 BRAF V600E Mutation-Positive Unresectable or Metastatic Solid Tumors
1.6 BRAF V600E Mutation-Positive Low-Grade Glioma
1.7 Limitations of Use
2 DOSAGE AND ADMINISTRATION
2.1 Patient Selection
2.2 Recommended Dosage
2.3 Administration
2.4 Dosage Modifications for Adverse Reactions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 New Primary Malignancies
5.2 Hemorrhage
5.3 Colitis and Gastrointestinal Perforation
5.4 Venous Thromboembolic Events
5.5 Cardiomyopathy
5.6 Ocular Toxicities
5.7 Interstitial Lung Disease/Pneumonitis
5.8 Serious Febrile Reactions
5.9 Serious Skin Toxicities
5.10 Hyperglycemia
5.11 Risks Associated with Combination Treatment
5.12 Hemophagocytic Lymphohistiocytosis
5.13 Embryo-Fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 BRAF V600E or V600K Mutation-Positive Unresectable or Metastatic Melanoma
14.2 Adjuvant Treatment of BRAF V600E or V600K Mutation-Positive Melanoma
14.3 BRAF V600E Mutation-Positive Metastatic Non-Small Cell Lung Cancer
14.4 BRAF V600E Mutation-Positive Locally Advanced or Metastatic Anaplastic Thyroid Cancer
14.5 Lack of Clinical Activity in Metastatic Melanoma Following BRAF-Inhibitor Therapy
14.6 BRAF V600E Mutation-Positive Unresectable or Metastatic Solid Tumors
14.7 BRAF V600E Mutation-Positive Low-Grade Glioma
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1
INDICATIONS AND USAGE
1.1 BRAF V600E or V600K Mutation-Positive Unresectable or Metastatic Melanoma
MEKINIST® is indicated, as a single agent in BRAF-inhibitor treatment-naïve patients or in combination with dabrafenib, for the treatment of patients with unresectable or metastatic melanoma with BRAF V600E or V600K mutations, as detected by an FDA-approved test [see Dosage and Administration (2.1)].
1.2 Adjuvant Treatment of BRAF V600E or V600K Mutation-Positive Melanoma
MEKINIST is indicated, in combination with dabrafenib, for the adjuvant treatment of patients with melanoma with BRAF V600E or V600K mutations as detected by an FDA-approved test, and involvement of lymph node(s), following complete resection [see Dosage and Administration (2.1)].
1.3 BRAF V600E Mutation-Positive Metastatic NSCLC
MEKINIST is indicated, in combination with dabrafenib, for the treatment of patients with metastatic non-small cell lung cancer (NSCLC) with BRAF V600E mutation as detected by an FDA-approved test [see Dosage and Administration (2.1)].
1.4 BRAF V600E Mutation-Positive Locally Advanced or Metastatic Anaplastic Thyroid Cancer
MEKINIST is indicated, in combination with dabrafenib, for the treatment of patients with locally advanced or metastatic anaplastic thyroid cancer (ATC) with BRAF V600E mutation, as detected by an FDA-approved test, and with no satisfactory locoregional treatment options [see Dosage and Administration (2.1)].
1.5 BRAF V600E Mutation-Positive Unresectable or Metastatic Solid Tumors
MEKINIST is indicated, in combination with dabrafenib, for the treatment of adult and pediatric patients 1 year of age and older with unresectable or metastatic solid tumors with BRAF V600E mutation who have progressed following prior treatment and have no satisfactory alternative treatment options [see Dosage and Administration (2.1)]. This indication is approved under accelerated approval based on overall response rate and duration of response [see Clinical Studies (14.6)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
-
2
DOSAGE AND ADMINISTRATION
2.1 Patient Selection
Melanoma
- Confirm the presence of BRAF V600E or V600K mutation in tumor specimens prior to initiation of treatment with MEKINIST as a single agent or in combination with dabrafenib [see Clinical Studies (14.1, 14.2)].
- Information on FDA-approved tests for the detection of BRAF V600 mutations in melanoma is available at: http://www.fda.gov/CompanionDiagnostics.
NSCLC
- Confirm the presence of BRAF V600E mutation in tumor specimens prior to initiation of treatment with MEKINIST and dabrafenib [see Clinical Studies (14.3)].
- Information on FDA-approved tests for the detection of BRAF V600E mutations in NSCLC is available at: http://www.fda.gov/CompanionDiagnostics.
ATC
- Confirm the presence of BRAF V600E mutation in tumor specimens prior to initiation of treatment with MEKINIST and dabrafenib [see Clinical Studies (14.4)].
- Information on FDA-approved tests for the detection of BRAF V600E mutations in ATC is available at: http://www.fda.gov/CompanionDiagnostics.
Solid Tumors
- Confirm the presence of BRAF V600E mutation in tumor specimens prior to initiation of treatment with MEKINIST and dabrafenib [see Clinical Studies (14.6)]. An FDA-approved test for the detection of BRAF V600E mutation in solid tumors other than melanoma and NSCLC is not currently available.
Low-Grade Glioma
- Confirm the presence of BRAF V600E mutation in tumor specimens prior to initiation of treatment with MEKINIST and dabrafenib [see Clinical Studies (14.7)]. An FDA-approved test for the detection of BRAF V600E mutation in LGG is not currently available.
2.2 Recommended Dosage
MEKINIST Tablets
Adult Patients
The recommended dosage for MEKINIST tablets in adult patients is 2 mg orally taken once daily [see Dosage and Administration (2.3)].
Pediatric Patients
The recommended dosage for MEKINIST tablets in pediatric patients who weigh at least 26 kg is based on body weight (Table 1) [see Dosage and Administration (2.3)]. A recommended dosage of MEKINIST tablets has not been established in patients who weigh less than 26 kg.
Table 1. Recommended Dosage for MEKINIST Tablets in Pediatric Patients (Weight-based) Body Weight Recommended Dosage 26 to 37 kg 1 mg orally once daily 38 to 50 kg 1.5 mg orally once daily 51 kg or greater 2 mg orally once daily MEKINIST for Oral Solution
Adult and Pediatric Patients
The recommended dosage for MEKINIST for oral solution for adult and pediatric patients is based on body weight (Table 2) [see Dosage and Administration (2.3)].
Table 2. Recommended Dosage for MEKINIST for Oral Solution in Adult and Pediatric Patients (Weight-based) Body Weight Recommended Dosage
Total Volume of Oral Solution Once Daily
(Trametinib Content)8 kg 0.3 mg (6 mL) 9 kg 0.35 mg (7 mL) 10 kg 0.35 mg (7 mL) 11 kg 0.4 mg (8 mL) 12 to 13 kg 0.45 mg (9 mL) 14 to 17 kg 0.55 mg (11 mL) 18 to 21 kg 0.7 mg (14 mL) 22 to 25 kg 0.85 mg (17 mL) 26 to 29 kg 0.9 mg (18 mL) 30 to 33 kg 1 mg (20 mL) 34 to 37 kg 1.15 mg (23 mL) 38 to 41 kg 1.25 mg (25 mL) 42 to 45 kg 1.4 mg (28 mL) 46 to 50 kg 1.6 mg (32 mL) ≥ 51 kg 2 mg (40 mL) Duration of Treatment
- The recommended duration of treatment for patients with unresectable or metastatic melanoma or solid tumors, metastatic NSCLC, or locally advanced or metastatic anaplastic thyroid cancer is until disease progression or unacceptable toxicity.
- The recommended duration of treatment in the adjuvant melanoma setting is until disease recurrence or unacceptable toxicity for up to 1 year.
- The recommended duration of treatment for pediatric patients with LGG is until disease progression or until unacceptable toxicity.
Combination Therapy with Dabrafenib
Refer to the dabrafenib prescribing information for recommended dabrafenib dosing information.
2.3 Administration
- Take MEKINIST at the same time each day, approximately 24 hours apart.
- Do not take a missed dose of MEKINIST within 12 hours of the next dose of MEKINIST.
- If vomiting occurs after MEKINIST administration, do not take an additional dose. Take the next dose at its scheduled time.
MEKINIST Tablets
- Take MEKINIST tablets on an empty stomach (at least 1 hour before or 2 hours after a meal) [see Clinical Pharmacology (12.3)].
- Do not crush or break MEKINIST tablets.
MEKINIST for Oral Solution
- MEKINIST powder for oral solution must be reconstituted by a pharmacist or other healthcare provider prior to dispensing to the patient.
- MEKINIST for oral solution is intended for administration by a caregiver. Prior to use of the oral solution, ensure caregivers receive training on proper dosing and administration of MEKINIST for oral solution.
- When administering MEKINIST for oral solution as a single agent, take the oral solution with a low-fat meal or on an empty stomach [see Clinical Pharmacology (12.3)].
- When coadministering with dabrafenib, take the MEKINIST oral solution on an empty stomach (at least 1 hour before or 2 hours after a meal). Breastfeeding and/or baby formula may be given on demand if a pediatric patient is unable to tolerate the fasting conditions [see Clinical Pharmacology (12.3)].
Preparation and Administration
- To prepare MEKINIST for oral solution, tap the bottle until powder flows freely. Add 90 mL distilled or purified water to the powder in the bottle and invert or gently shake the bottle with re-attached cap for up to 5 minutes until powder is fully dissolved yielding a clear solution. Separate the bottle adapter from the oral syringe. Insert bottle adapter into bottle neck after reconstitution of the solution. Write the discard after date. Once reconstituted, MEKINIST for oral solution can be used for 35 days.
- The final concentration of the solution is 0.05 mg/mL.
- Administer MEKINIST for oral solution from an oral syringe or feeding tube (4 French gauge or larger).
- After reconstitution, store in original bottle below 25°C (77°F) and do not freeze.
2.4 Dosage Modifications for Adverse Reactions
Dose reductions for adverse reactions associated with MEKINIST are presented in Tables 3 and 4.
Table 3. Recommended Dosage Reductions for MEKINIST Tablets for Adverse Reactions Recommended Dosage 1 mg orally once daily 1.5 mg orally once daily 2 mg orally once daily First dose reduction 0.5 mg orally once daily 1 mg orally once daily 1.5 mg orally once daily Second dose reduction N/A 0.5 mg orally once daily 1 mg orally once daily Subsequent modification Permanently discontinue MEKINIST tablets if unable to tolerate a maximum of two dose reductions. Table 4. Recommended Dosage Reductions for MEKINIST for Oral Solution for Adverse Reactions Body Weight
(Recommended dosage once daily)First Dose Reduction
(Administer once daily)Second Dose Reduction
(Administer once daily)8 kg
[0.3 mg (6 mL)]0.25 mg (5 mL) 0.15 mg (3 mL) 9 kg
[0.35 mg (7 mL)]0.25 mg (5 mL) 0.2 mg (4 mL) 10 kg
[0.35 mg (7 mL)]0.25 mg (5 mL) 0.2 mg (4 mL) 11 kg
[0.4 mg (8 mL)]0.3 mg (6 mL) 0.2 mg (4 mL) 12 to 13 kg
[0.45 mg (9 mL)]0.35 mg (7 mL) 0.25 mg (5 mL) 14 to 17 kg
[0.55 mg (11 mL)]0.4 mg (8 mL) 0.3 mg (6 mL) 18 to 21 kg
[0.7 mg (14 mL)]0.55 mg (11 mL) 0.35 mg (7 mL) 22 to 25 kg
[0.85 mg (17 mL)]0.65 mg (13 mL) 0.45 mg (9 mL) 26 to 29 kg
[0.9 mg (18 mL)]0.7 mg (14 mL) 0.45 mg (9 mL) 30 to 33 kg
[1 mg (20 mL)]0.75 mg (15 mL) 0.5 mg (10 mL) 34 to 37 kg
[1.15 mg (23 mL)]0.85 mg (17 mL) 0.6 mg (12 mL) 38 to 41 kg
[1.25 mg (25 mL)]0.95 mg (19 mL) 0.65 mg (13 mL) 42 to 45 kg
[1.4 mg (28 mL)]1.05 mg (21 mL) 0.7 mg (14 mL) 46 to 50 kg
[1.6 mg (32 mL)]1.2 mg (24 mL) 0.8 mg (16 mL) ≥ 51 kg
[2 mg (40 mL)]1.5 mg (30 mL) 1 mg (20 mL) Permanently discontinue MEKINIST for oral solution if unable to tolerate a maximum of two dose reductions. Dosage modifications for adverse reactions associated with MEKINIST are presented in Table 5.
Table 5. Recommended Dosage Modifications for MEKINIST for Adverse Reactions aNational Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 4.0.
bSee Tables 3 and 4 for recommended dose reductions of MEKINIST.
cDose modifications are not recommended for MEKINIST when administered with dabrafenib for the following adverse reactions of dabrafenib: non-cutaneous malignancies and uveitis. Dose modification of MEKINIST is not required for new primary cutaneous malignancies.Severity of Adverse Reactiona Dosage Modification for MEKINISTb Hemorrhage [see Warnings and Precautions (5.2)] - Grade 3
Withhold MEKINIST.
- If improved, resume MEKINIST at lower dose.
- If not improved, permanently discontinue MEKINIST.
- Grade 4
Permanently discontinue MEKINIST. Venous Thromboembolic Events [see Warnings and Precautions (5.4)] - Uncomplicated deep venous thrombosis (DVT) or pulmonary embolism (PE)
Withhold MEKINIST for up to 3 weeks.
- If improved to Grade 0-1, resume MEKINIST at lower dose.
- If not improved, permanently discontinue MEKINIST.
- Life-threatening PE
Permanently discontinue MEKINIST. Cardiomyopathy [see Warnings and Precautions (5.5)] - Asymptomatic, absolute decrease in left ventricular ejection fraction (LVEF) of 10% or greater from baseline that is below the institutional lower limit of normal (LLN)
Withhold MEKINIST for up to 4 weeks. - If improved to normal LVEF value, resume MEKINIST at lower dose.
- If not improved to normal LVEF value, permanently discontinue MEKINIST.
- Symptomatic cardiomyopathy
- Absolute decrease in LVEF of greater than 20% from baseline that is below the institutional LLN
Permanently discontinue MEKINIST. Ocular Toxicities [see Warnings and Precautions (5.6)] - Retinal pigment epithelial detachments (RPED)
Withhold MEKINIST for up to 3 weeks. - If improved, resume MEKINIST at same or lower dose.
- If not improved, permanently discontinue MEKINIST or resume MEKINIST at lower dose.
- Retinal vein occlusion (RVO)
Permanently discontinue MEKINIST.
Pulmonary [see Warnings and Precautions (5.7)] - Interstitial lung disease (ILD)/pneumonitis
Permanently discontinue MEKINIST. Febrile Reactions [see Warnings and Precautions (5.8)] - Fever of 100.4°F to 104°F (or first symptoms in case of recurrence)
Withhold MEKINIST until fever resolves, then resume MEKINIST at same or lower dose. - Fever higher than 104°F
- Fever complicated by rigors, hypotension, dehydration, or renal failure
- Withhold MEKINIST until febrile reactions resolve for at least 24 hours, then resume MEKINIST at lower dose.
- Permanently discontinue MEKINIST.
Skin Toxicities [see Warnings and Precautions (5.9)] - Intolerable Grade 2
- Grade 3 or 4
Withhold MEKINIST for up to 3 weeks. - If improved, resume MEKINIST at lower dose.
- If not improved, permanently discontinue MEKINIST.
- Severe cutaneous adverse reactions (SCARs)
Permanently discontinue MEKINIST. Other Adverse Reactionsc - Intolerable Grade 2
- Any Grade 3
Withhold MEKINIST. - If improved to Grade 0-1, resume MEKINIST at lower dose.
- If not improved, permanently discontinue MEKINIST.
- First occurrence of any Grade 4
- Withhold MEKINIST until improves to Grade 0-1, then resume MEKINIST at lower dose.
- Permanently discontinue MEKINIST.
- Recurrent Grade 4
Permanently discontinue MEKINIST. Refer to the dabrafenib prescribing information for dose modifications for adverse reactions associated with dabrafenib.
-
3
DOSAGE FORMS AND STRENGTHS
MEKINIST tablets:
- 0.5 mg tablets: Yellow, ovaloid, biconvex, unscored film-coated tablets with beveled edges and with the Novartis logo debossed on one side and ‘TT’ on the other side.
- 2 mg tablets: Pink, round, biconvex, unscored film-coated tablets with beveled edges and with the Novartis logo debossed on one side and ‘LL’ on the other side.
MEKINIST for oral solution:
- White to almost white powder containing 4.7 mg of trametinib per bottle. Each mL of reconstituted strawberry-flavored trametinib solution contains 0.05 mg of trametinib.
- 4 CONTRAINDICATIONS
-
5
WARNINGS AND PRECAUTIONS
5.1 New Primary Malignancies
Cutaneous Malignancies
MEKINIST Administered with Dabrafenib (Adult): In the pooled safety population [see Adverse Reactions (6.1)], cutaneous squamous cell carcinomas (cuSCCs) and keratoacanthomas occurred in 2% of patients. Basal cell carcinoma and new primary melanoma occurred in 3% and < 1% of patients, respectively.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population, new primary melanoma occurred in < 1% of patients.
Perform dermatologic evaluations prior to initiation of MEKINIST when used with dabrafenib, every 2 months while on therapy, and for up to 6 months following discontinuation of the combination.
Non-Cutaneous Malignancies
Based on its mechanism of action, dabrafenib may promote growth and development of malignancies with activation of RAS through mutation or other mechanisms; refer to the prescribing information for dabrafenib.
In the pooled safety population of MEKINIST administered with dabrafenib, non-cutaneous malignancies occurred in 1% of patients.
Monitor patients receiving MEKINIST and dabrafenib closely for signs or symptoms of non-cutaneous malignancies. No dose modification is required for MEKINIST in patients who develop non-cutaneous malignancies.
5.2 Hemorrhage
Hemorrhages, including major hemorrhage defined as symptomatic bleeding in a critical area or organ, can occur with MEKINIST. Fatal cases have been reported.
MEKINIST Administered with Dabrafenib (Adult): In the pooled safety population [see Adverse Reactions (6.1)], hemorrhagic events occurred in 17% of patients; gastrointestinal hemorrhage occurred in 3% of patients; intracranial hemorrhage occurred in 0.6% of patients; fatal hemorrhage occurred in 0.5% of patients. The fatal events were cerebral hemorrhage and brainstem hemorrhage.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population, hemorrhagic events occurred in 25% of patients; the most common type of bleeding was epistaxis (16%). Serious events of bleeding occurred in 3.6% of patients and included gastrointestinal hemorrhage (1.2%), cerebral hemorrhage (0.6%), uterine hemorrhage (0.6%), post-procedural hemorrhage (0.6%), and epistaxis (0.6%).
Permanently discontinue MEKINIST for all Grade 4 hemorrhagic events and for any Grade 3 hemorrhagic events that do not improve. Withhold MEKINIST for Grade 3 hemorrhagic events; if improved, resume MEKINIST at the next lower dose level.
5.3 Colitis and Gastrointestinal Perforation
Colitis and gastrointestinal perforation, including fatal outcomes, have been reported in patients taking:
MEKINIST Monotherapy and Administered with Dabrafenib (Adult): In the pooled safety population [see Adverse Reactions (6.1)], colitis occurred in < 1% of patients and gastrointestinal perforation occurred in < 1% of patients.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population, colitis events occurred in < 1% of patients.
Monitor patients closely for colitis and gastrointestinal perforations.
5.4 Venous Thromboembolic Events
MEKINIST Administered with Dabrafenib (Adult): In the pooled safety population [see Adverse Reactions (6.1)], deep vein thrombosis (DVT) and pulmonary embolism (PE) occurred in 2% of patients.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population, embolism events occurred in < 1% of patients.
Advise patients to immediately seek medical care if they develop symptoms of DVT or PE, such as shortness of breath, chest pain, or arm or leg swelling. Permanently discontinue MEKINIST for life-threatening PE. Withhold MEKINIST for uncomplicated DVT and PE for up to 3 weeks; if improved, MEKINIST may be resumed at a lower dose level [see Dosage and Administration (2.4)].
5.5 Cardiomyopathy
Cardiomyopathy, including cardiac failure, can occur with MEKINIST.
MEKINIST Administered with Dabrafenib (Adult): In the pooled safety population [see Adverse Reactions (6.1)], cardiomyopathy, defined as a decrease in left ventricular ejection fraction (LVEF) ≥ 10% from baseline and below the institutional lower limit of normal (LLN), occurred in 6% of patients. Development of cardiomyopathy resulted in dose interruption or discontinuation of MEKINIST in 3% and < 1% of patients, respectively. Cardiomyopathy resolved in 45 of 50 patients who received MEKINIST administered with dabrafenib.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population, cardiomyopathy, defined as a decrease in LVEF ≥ 10% from baseline and below the institutional LLN, occurred in 9% of patients.
Assess LVEF by echocardiogram or multigated acquisition (MUGA) scan before initiation of MEKINIST as a single agent or with dabrafenib, one month after initiation, and then at 2- to 3-month intervals while on treatment. For an asymptomatic absolute decrease in LVEF of 10% or greater from baseline that is below the institutional LLN, withhold MEKINIST for up to 4 weeks. If improved to normal LVEF value, resume MEKINIST at a lower dose. If no improvement to normal LVEF value within 4 weeks, permanently discontinue MEKINIST. For symptomatic cardiomyopathy or an absolute decrease in LVEF of greater than 20% from baseline that is below the institutional LLN, permanently discontinue MEKINIST [see Dosage and Administration (2.4)].
5.6 Ocular Toxicities
Retinal Vein Occlusion
In the pooled safety population [see Adverse Reactions (6.1)] of MEKINIST monotherapy, the incidence of retinal vein occlusion (RVO) was 0.6%. In the pooled safety population [see Adverse Reactions (6.1)] of MEKINIST administered with dabrafenib, there were no cases of RVO. RVO may lead to macular edema, decreased visual function, neovascularization, and glaucoma.
Urgently (within 24 hours) perform ophthalmological evaluation for patient-reported loss of vision or other visual disturbances. Permanently discontinue MEKINIST in patients with documented RVO [see Dosage and Administration (2.4)].
Retinal Pigment Epithelial Detachment
Retinal pigment epithelial detachment (RPED) can occur with MEKINIST. Retinal detachments may be bilateral and multifocal, occurring in the central macular region of the retina or elsewhere in the retina. In melanoma and NSCLC trials, routine monitoring of patients to detect asymptomatic RPED was not conducted; therefore, the true incidence of this finding is unknown.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population, RPED events occurred in < 1% of patients.
Perform ophthalmological evaluation periodically and at any time a patient reports visual disturbances. Withhold MEKINIST if RPED is diagnosed. If resolution of the RPED is documented on repeat ophthalmological evaluation within 3 weeks, resume MEKINIST at same or reduced dose. If no improvement after 3 weeks, resume MEKINIST at reduced dose or permanently discontinue MEKINIST [see Dosage and Administration (2.4)].
5.7 Interstitial Lung Disease/Pneumonitis
In the pooled safety population [see Adverse Reactions (6.1)] of MEKINIST monotherapy, interstitial lung disease or pneumonitis occurred in 2% of patients. In the pooled safety population [see Adverse Reactions (6.1)] of MEKINIST administered with dabrafenib, ILD or pneumonitis occurred in 1% of patients.
Withhold MEKINIST in patients presenting with new or progressive pulmonary symptoms and findings, including cough, dyspnea, hypoxia, pleural effusion, or infiltrates, pending clinical investigations. Permanently discontinue MEKINIST for patients diagnosed with treatment-related ILD or pneumonitis [see Dosage and Administration (2.4)].
5.8 Serious Febrile Reactions
Serious febrile reactions and fever of any severity accompanied by hypotension, rigors or chills, dehydration, or renal failure, can occur when MEKINIST is administered with dabrafenib.
MEKINIST Administered with Dabrafenib (Adult): In the pooled safety population [see Adverse Reactions (6.1)], fever occurred in 58% of patients. Serious febrile reactions and fever of any severity complicated by hypotension, rigors or chills, dehydration or renal failure occurred in 5% of patients. Fever was complicated by hypotension in 4%, dehydration in 3%, syncope in 2%, renal failure in 1%, and severe chills/rigors in < 1% of patients.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population [see Adverse Reactions (6.1)], pyrexia occurred in 66% of patients.
Withhold MEKINIST when used as monotherapy, and both MEKINIST and dabrafenib when used in combination, if the patient’s temperature is ≥ 100.4°F. In case of recurrence, therapy can also be interrupted at the first symptom of pyrexia [see Adverse Reactions (6.1)]. Fever may be complicated by hypotension, rigors or chills, dehydration, or renal failure. Evaluate for signs and symptoms of infection and monitor serum creatinine and other evidence of renal function during and following severe pyrexia. If appropriate, MEKINIST, or both MEKINIST and dabrafenib when used in combination, may be restarted if the patient has recovered from the febrile reaction for at least 24 hours, either at same or lower dose [see Dosage and Administration (2.4)]. Administer antipyretics as secondary prophylaxis when resuming MEKINIST if patient had a prior episode of severe febrile reaction or fever associated with complications. Administer corticosteroids (e.g., prednisone 10 mg daily) for at least 5 days for second or subsequent pyrexia if temperature does not return to baseline within 3 days of onset of pyrexia, or for pyrexia associated with complications, such as dehydration, hypotension, renal failure, or severe chills/rigors, and there is no evidence of active infection.
5.9 Serious Skin Toxicities
Severe cutaneous adverse reactions (SCARs), including Stevens-Johnson syndrome (SJS) and drug reaction with eosinophilia and systemic symptoms (DRESS), which can be life-threatening or fatal, have been reported during treatment with MEKINIST administered with dabrafenib [see Adverse Reactions (6.2)].
MEKINIST Administered with Dabrafenib (Adult): In the pooled safety population [see Adverse Reactions (6.1)], other serious skin toxicity occurred in < 1% of patients.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population, serious adverse events of skin and subcutaneous tissue disorders occurred in 1.8% of patients.
Monitor for new or worsening serious skin reactions. Permanently discontinue MEKINIST for SCARs [see Dosage and Administration (2.4)]. For other skin toxicities, withhold MEKINIST for intolerable or severe skin toxicity. Resume MEKINIST at a lower dose in patients with improvement or recovery from skin toxicity within 3 weeks. Permanently discontinue MEKINIST if skin toxicity has not improved in 3 weeks [see Dosage and Administration (2.4)].
5.10 Hyperglycemia
MEKINIST Administered with Dabrafenib (Adult): In the pooled safety population [see Adverse Reactions (6.1)], 15% of patients with a history of diabetes who had received MEKINIST with dabrafenib required more intensive hypoglycemic therapy. Grade 3 and Grade 4 hyperglycemia occurred in 2% of patients.
MEKINIST Administered with Dabrafenib (Pediatric): In the pooled safety population, Grade 3 and Grade 4 hyperglycemia events occurred in < 1% of patients.
Monitor serum glucose levels upon initiation and as clinically appropriate when MEKINIST is administered with dabrafenib in patients with preexisting diabetes or hyperglycemia. Initiate or optimize anti-hyperglycemic medications as clinically indicated.
5.11 Risks Associated with Combination Treatment
MEKINIST is indicated for use in combination with dabrafenib. Review the prescribing information for dabrafenib for information on the serious risks of dabrafenib prior to initiation of MEKINIST with dabrafenib.
5.12 Hemophagocytic Lymphohistiocytosis
Hemophagocytic lymphohistiocytosis (HLH) has been observed in the post-marketing setting when MEKINIST was administered with dabrafenib. If HLH is suspected, interrupt treatment. If HLH is confirmed, discontinue treatment and initiate appropriate management of HLH.
5.13 Embryo-Fetal Toxicity
Based on findings from animal studies and its mechanism of action, MEKINIST can cause fetal harm when administered to a pregnant woman. Trametinib was embryotoxic and abortifacient in rabbits at doses greater than or equal to those resulting in exposures approximately 0.3 times the human exposure at the recommended adult clinical dose. Advise pregnant women of the potential risk to a fetus. Advise female patients of reproductive potential to use effective contraception during treatment with MEKINIST and for 4 months after treatment [see Use in Specific Populations (8.1, 8.3)].
-
6
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- New Primary Malignancies [see Warnings and Precautions (5.1)]
- Hemorrhage [see Warnings and Precautions (5.2)]
- Colitis and Gastrointestinal Perforation [see Warnings and Precautions (5.3)]
- Venous Thromboembolic Events [see Warnings and Precautions (5.4)]
- Cardiomyopathy [see Warnings and Precautions (5.5)]
- Ocular Toxicities [see Warnings and Precautions (5.6)]
- Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.7)]
- Serious Febrile Reactions [see Warnings and Precautions (5.8)]
- Serious Skin Toxicities [see Warnings and Precautions (5.9)]
- Hyperglycemia [see Warnings and Precautions (5.10)]
- Hemophagocytic Lymphohistiocytosis [see Warnings and Precautions (5.12)]
There are additional adverse reactions associated with dabrafenib. Refer to the dabrafenib prescribing information for additional information.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adult Safety Pools
The pooled safety population described in the WARNINGS AND PRECAUTIONS reflects exposure to MEKINIST 2 mg orally, once daily as a single agent in 329 patients with various solid tumors enrolled in METRIC, MEK113583, and MEK111054. Among these 329 patients who received MEKINIST as a single agent, 33% were exposed for 6 months or longer and 9% were exposed for greater than one year.
The pooled safety population described in the WARNINGS AND PRECAUTIONS reflects exposure to MEKINIST 2 mg orally, once daily, administered in combination with dabrafenib 150 mg orally, twice daily, in 1087 patients enrolled in COMBI-d, COMBI-v, COMBI-AD, and BRF113928 with unresectable or metastatic melanoma, adjuvant melanoma, or NSCLC. Among these 1087 patients who received MEKINIST administered with dabrafenib, 70% were exposed for 6 months or longer and 21% were exposed for greater than one year.
Pediatric Safety Pool
The pediatric pooled safety population described in the WARNINGS AND PRECAUTIONS reflects exposure to weight-based MEKINIST orally, once daily administered in combination with dabrafenib in 166 pediatric patients across two trials: a multi-center, open-label, multi-cohort study in pediatric patients with BRAF V600E mutation-positive glioma requiring systemic therapy (Study G2201; n = 123) and a multi-center, open-label, multi-cohort study in pediatric patients with refractory or recurrent solid tumors with MAPK pathway activation (Study X2101; n = 43) [see Clinical Studies (14.6, 14.7)]. Among 166 patients who received MEKINIST administered with dabrafenib, 85% were exposed for 6 months and 69% were exposed for greater than one year. The most common (> 20%) adverse reactions were pyrexia (66%), rash (54%), headache (40%), vomiting (38%), musculoskeletal pain (36%), fatigue (31%), dry skin (31%), diarrhea (30%), nausea (26%), epistaxis and other bleeding events (25%), abdominal pain (24%), and dermatitis acneiform (23%). The most common (> 2%) Grade 3 or 4 laboratory abnormalities were decreased neutrophil count (20%), increased alanine aminotransferase (3.1%), and increased aspartate aminotransferase (3.1%).
Unresectable or Metastatic BRAF V600E or V600K Mutation-Positive Melanoma
MEKINIST as a Single Agent
The safety of MEKINIST was evaluated in the METRIC study, a randomized, open-label trial of patients with BRAF V600E or V600K mutation-positive unresectable or metastatic melanoma who received MEKINIST (N = 211) 2 mg orally once daily or chemotherapy (N = 99) (either dacarbazine 1000 mg/m2 every 3 weeks or paclitaxel 175 mg/m2 every 3 weeks) [see Clinical Studies (14.1)]. Patients with abnormal LVEF, history of acute coronary syndrome within 6 months, or current evidence of Class II or greater congestive heart failure (New York Heart Association) were excluded. The median duration of treatment with MEKINIST was 4.3 months.
In this study, 9% of patients who received MEKINIST experienced adverse reactions resulting in permanent discontinuation of trial medication. The most frequent adverse reactions resulting in permanent discontinuation of MEKINIST were decreased LVEF, pneumonitis, renal failure, diarrhea, and rash. Adverse reactions led to dose reductions in 27% of patients treated with MEKINIST. Rash and decreased LVEF were the most frequent reasons cited for dose reductions of MEKINIST. Table 6 and Table 7 present adverse reactions and laboratory abnormalities, respectively, of MEKINIST as a single agent in the METRIC study.
Table 6. Select Adverse Reactions Occurring in ≥ 10% of Patients Who Received MEKINIST and at a Higher Incidence (≥ 5%) Than in the Chemotherapy Arm or ≥ 2% (Grades 3 or 4) Adverse Reactions in the METRIC Study aNCI CTCAE version 4.0.
bGrade 4 adverse reactions limited to rash (n = 1) in trametinib arm and diarrhea (n = 1) in chemotherapy arm.
cIncludes stomatitis, aphthous stomatitis, mouth ulceration, and mucosal inflammation.
dIncludes abdominal pain, lower abdominal pain, upper abdominal pain, and abdominal tenderness.
eIncludes lymphedema, edema, and peripheral edema.
fIncludes epistaxis, gingival bleeding, hematochezia, rectal hemorrhage, melena, vaginal hemorrhage, hemorrhoidal hemorrhage, hematuria, and conjunctival hemorrhage.Adverse Reactions MEKINIST Chemotherapy N = 211 N = 99 All
Gradesa
(%)Grades
3 and 4b
(%)All
Gradesa
(%)Grades
3 and 4b
(%)Skin and subcutaneous tissue Rash 57 8 10 0 Acneiform dermatitis 19 < 1 1 0 Dry skin 11 0 0 0 Pruritus 10 2 1 0 Paronychia 10 0 1 0 Gastrointestinal Diarrhea 43 0 16 2 Stomatitisc 15 2 2 0 Abdominal paind 13 1 5 1 Vascular Lymphedemae 32 1 4 0 Hypertension 15 12 7 3 Hemorrhagef 13 < 1 0 0 Table 7. Laboratory Abnormalities Occurring at a Higher Incidence in Patients Who Received MEKINIST in the METRIC Study [Between-Arm Difference of ≥ 5% (All Grades) or ≥ 2% (Grades 3 or 4)a] Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aOnly Grade 3 adverse reactions were reported in either treatment arm.Laboratory Abnormality MEKINIST Chemotherapy N = 211 N = 99 All
Grades
(%)Grades
3 and 4
(%)All
Grades
(%)Grades
3 and 4
(%)Increased AST 60 2 16 1 Hypoalbuminemia 42 2 23 1 Increased ALT 39 3 20 3 Anemia 38 2 26 3 Increased alkaline phosphatase 24 2 18 3 Other clinically important adverse reactions for MEKINIST in a pool of MEKINIST monotherapy clinical studies observed in less than 10% of patients who received MEKINIST were:
Cardiac: Bradycardia, atrioventricular block, bundle branch block
Gastrointestinal: Dry mouth
Infections and Infestations: Folliculitis, rash pustular, cellulitis
Musculoskeletal and Connective Tissue: Rhabdomyolysis
Nervous System: Dizziness, dysgeusia, peripheral neuropathy
Ocular: Blurred vision, dry eye
MEKINIST with Dabrafenib
The safety of MEKINIST when administered with dabrafenib was evaluated in 559 patients with previously untreated, unresectable or metastatic, BRAF V600 mutation-positive melanoma who received MEKINIST in two trials, the COMBI-d study (n = 209), a multi-center, double-blind, randomized (1:1), active-controlled trial and the COMBI-v study (n = 350), a multi-center, open-label, randomized (1:1), active-controlled trial. In both trials, patients received MEKINIST 2 mg orally once daily and dabrafenib 150 mg orally twice daily until disease progression or unacceptable toxicity. Both trials excluded patients with abnormal LVEF, history of acute coronary syndrome within 6 months, history of Class II or greater congestive heart failure (New York Heart Association), history of RVO or RPED, QTcB interval ≥ 480 msec, uncontrolled hypertension, uncontrolled arrhythmias, active brain metastases, or known history of glucose-6-phosphate dehydrogenase deficiency [see Clinical Studies (14.1)].
Among these 559 patients, 197 (35%) were exposed to MEKINIST for > 6 months to 12 months while 185 (33%) were exposed to MEKINIST for > 1 year. The median age was 55 years (range: 18 to 91), 57% were male, and 98% were White, 72% had baseline ECOG performance status of 0 and 28% had ECOG performance status of 1, 64% had M1c disease, 35% had elevated lactate dehydrogenase (LDH) at baseline, and 0.5% had a history of brain metastases.
The most common adverse reactions (≥ 20%) for MEKINIST in patients who received MEKINIST plus dabrafenib in the COMBI-d and COMBI-v studies were: pyrexia, nausea, rash, chills, diarrhea, vomiting, hypertension, and peripheral edema.
The demographics and baseline tumor characteristics of patients enrolled in the COMBI-d study are summarized in Clinical Studies [see Clinical Studies (14.1)]. Patients who received MEKINIST plus dabrafenib had a median duration of exposure of 11 months (range: 3 days to 30 months) to MEKINIST. Among the 209 patients who received MEKINIST plus dabrafenib, 26% were exposed to MEKINIST for > 6 months to 12 months while 46% were exposed to MEKINIST for > 1 year.
In the COMBI-d study, adverse reactions leading to discontinuation of MEKINIST occurred in 11% of patients who received MEKINIST plus dabrafenib; the most frequent were pyrexia (1.4%) and decreased ejection fraction (1.4%). Adverse reactions leading to dose reductions of MEKINIST occurred in 18% of patients who received MEKINIST plus dabrafenib; the most frequent were pyrexia (2.9%), neutropenia (1.9%), decreased ejection fraction (1.9%), and rash (1.9%). Adverse reactions leading to dose interruptions of MEKINIST occurred in 46% of patients who received MEKINIST plus dabrafenib; the most frequent were pyrexia (18%), chills (7%), vomiting (6%), and decreased ejection fraction (4.8%).
Table 8 and Table 9 present selected adverse reactions and laboratory abnormalities, respectively, of MEKINIST observed in the COMBI-d study.
Table 8. Adverse Reactions Occurring in ≥ 10% (All Grades) of Patients Who Received MEKINIST with Dabrafenib and at a Higher Incidence* Than in Patients Who Received Single-Agent Dabrafenib in COMBI-da * ≥ 5% for All Grades or ≥ 2% for Grades 3–4 incidence in patients who received MEKINIST with dabrafenib compared with patients who received dabrafenib as a single agent.
aNCI CTCAE version 4.0.
bIncludes peripheral edema, edema, lymphedema, localized edema, and generalized edema.
cIncludes abdominal pain, upper abdominal pain, lower abdominal pain, and abdominal discomfort.
dIncludes rash, generalized rash, pruritic rash, erythematous rash, papular rash, vesicular rash, macular rash, maculo-papular rash, and follicular rash.
eMost common events (≥ 1%) include epistaxis, hematochezia, decreased hemoglobin, purpura, and rectal hemorrhage. Grade 4 events were limited to hepatic hematoma and duodenal ulcer hemorrhage (each n = 1 in the pooled combination arm).Adverse Reactions Pooled MEKINIST plus
Dabrafenib
N = 559COMBI-d Study MEKINIST plus
Dabrafenib N = 209Dabrafenib
N = 211All
Grades
(%)Grades
3 and 4
(%)All
Grades
(%)Grades
3 and 4
(%)All
Grades
(%)Grades
3 and 4
(%)General Pyrexia 54 5 57 7 33 1.9 Chills 31 0.5 31 0 17 0.5 Peripheral edemab 21 0.7 25 1.4 11 0.5 Gastrointestinal Nausea 35 0.4 34 0.5 27 1.4 Diarrhea 31 1.3 30 1.4 16 0.9 Vomiting 27 1.1 25 1.0 14 0.5 Abdominal painc 18 0.9 26 1.0 14 2.4 Skin and subcutaneous tissue Rashd 32 1.1 42 0 27 1.4 Vascular Hypertension 26 11 25 6 16 6 Hemorrhagee 18 2.0 19 1.9 15 1.9 Nervous system Dizziness 11 0.2 14 0 7 0 Other clinically important adverse reactions for MEKINIST across the COMBI-d and COMBI-v studies (N = 559) observed in less than 10% of patients who received MEKINIST in combination with dabrafenib were:
Cardiac: Bradycardia, atrioventricular block, bundle branch block
Immune System: Sarcoidosis
Musculoskeletal and Connective Tissue: Rhabdomyolysis
Nervous System: Peripheral neuropathy, Guillain-Barré syndrome
Skin and Subcutaneous Tissue: Photosensitivity
Table 9. Laboratory Abnormalities Worsening from Baseline Occurring at ≥ 10% (All Grades) of Patients Who Received MEKINIST with Dabrafenib and at a Higher Incidence* Than in Patients Who Received Single-Agent Dabrafenib in COMBI-d Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
* ≥ 5% for All Grades or ≥ 2% for Grades 3–4 incidence in patients who received MEKINIST with dabrafenib compared with patients who received dabrafenib as a single agent.
aFor these laboratory tests, the denominator is 556.
bFor these laboratory tests, the denominator is 208 for the combination arm, 207-209 for the dabrafenib arm.
cGrade 4 adverse reactions limited to lymphopenia and hyperglycemia (each n = 4), increased ALT and increased AST (each n = 3), neutropenia (n = 2), and hyponatremia (n = 1) in the pooled combination arm; neutropenia, lymphopenia, increased ALT, increased AST, and hyperglycemia (each n = 1) in the COMBI-d study combination arm; neutropenia, thrombocytopenia, increased ALT, and increased AST (each n = 1) in the dabrafenib arm.Laboratory Abnormality Pooled MEKINIST plus
Dabrafenib
N = 559aCOMBI-d Study MEKINIST plus
Dabrafenib N = 209bDabrafenib
N = 211bAll
Grades
(%)Grades
3 and 4c
(%)All
Grades
(%)Grades
3 and 4c
(%)All
Grades
(%)Grades
3 and 4c
(%)Chemistry Hyperglycemia 60 4.7 65 6 57 4.3 Hypoalbuminemia 48 1.1 53 1.4 27 0 Hyponatremia 25 8 24 6 14 2.9 Hepatic Increased AST 59 4.1 60 4.3 21 1.0 Increased blood alkaline phosphatase 49 2.7 50 1.0 25 0.5 Increased ALT 48 4.5 44 3.8 28 1.0 Hematology Neutropenia 46 7 50 6 16 1.9 Anemia 43 2.3 43 2.4 38 4.3 Lymphopenia 32 8 38 9 28 7 Thrombocytopenia 21 0.7 19 0.5 10 0.5 Adjuvant Treatment of BRAF V600E or V600K Mutation-Positive Melanoma
The safety of MEKINIST when administered with dabrafenib was evaluated in 435 patients with Stage III melanoma with BRAF V600E or V600K mutations following complete resection who received at least one dose of study therapy in the COMBI-AD study [see Clinical Studies (14.2)]. Patients received MEKINIST 2 mg orally once daily and dabrafenib 150 mg orally twice daily for 12 months. The trial excluded patients with abnormal LVEF; history of acute coronary syndromes, coronary angioplasty, or stenting within 6 months; Class II or greater congestive heart failure (New York Heart Association); QTc interval ≥ 480 msec; treatment refractory hypertension; uncontrolled arrhythmias; or history of RVO.
Patients who received MEKINIST in combination with dabrafenib had a median duration of exposure of 11 months (range: 0 to 12) to MEKINIST. Among the 435 patients who received MEKINIST in combination with dabrafenib, 72% were exposed to MEKINIST for > 6 months. The median age of patients who received MEKINIST in combination with dabrafenib was 50 years (range: 18 to 89), 56% were male, 99% were White, 92% had baseline ECOG performance status of 0, and 8% had baseline ECOG performance status of 1.
The most common adverse reactions (≥ 20%) in patients who received MEKINIST in combination with dabrafenib were: pyrexia, fatigue, nausea, headache, rash, chills, diarrhea, vomiting, arthralgia, and myalgia.
Adverse reactions resulting in discontinuation and dose interruptions of MEKINIST occurred in 24% and 54% of patients, respectively; the most frequent for each were pyrexia and chills. Adverse reactions leading to dose reductions of MEKINIST occurred in 23% of patients; the most frequent were pyrexia and decreased ejection fraction.
Table 10 summarizes the adverse reactions that occurred in at least 20% of the patients who received MEKINIST in combination with dabrafenib.
Table 10. Adverse Reactions Occurring in ≥ 20% of Patients in the COMBI-AD Studya aNCI CTCAE version 4.0.
bIncludes pyrexia and hyperpyrexia.
cIncludes fatigue, asthenia, and malaise.
dIncludes headache and tension headache.
eIncludes rash, rash maculo-papular, rash macular, rash generalized, rash erythematous, rash papular, rash pruritic, nodular rash, rash vesicular, and rash pustular.
fIncludes myalgia, musculoskeletal pain, and musculoskeletal chest pain.Adverse Reactions MEKINIST plus Dabrafenib
N = 435Placebo
N = 432All
Grades
(%)Grades
3 and 4
(%)All
Grades
(%)Grades
3 and 4
(%)General Pyrexiab 63 5 11 < 1 Fatiguec 59 5 37 < 1 Chills 37 1 4 0 Gastrointestinal Nausea 40 < 1 20 0 Diarrhea 33 < 1 15 < 1 Vomiting 28 < 1 10 0 Nervous system Headached 39 1 24 0 Skin and subcutaneous tissue Rashe 37 < 1 16 < 1 Musculoskeletal and connective tissue Arthralgia 28 < 1 14 0 Myalgiaf 20 < 1 14 0 Other clinically important adverse reactions for MEKINIST in the COMBI-AD study observed in less than 20% of patients who received MEKINIST in combination with dabrafenib were: blurred vision (6%), decreased ejection fraction (5%), peripheral neuropathy (2.5%), rhabdomyolysis (< 1%), atrioventricular block (< 1%), Guillain-Barré syndrome (< 1%), and sarcoidosis (< 1%).
The laboratory abnormalities are summarized in Table 11.
Table 11. Laboratory Abnormalities Worsening from Baseline Occurring in ≥ 20% of Patients in the COMBI-AD Study Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aThe incidence is based on the number of patients who had both a baseline and at least one on-study laboratory measurement:
MEKINIST plus Dabrafenib (range: 429 to 431) and Placebo arm (range: 426 to 428).Laboratory Abnormality MEKINIST plus Dabrafeniba
N = 435Placeboa
N = 432All
Grades
(%)Grades
3 and 4
(%)All
Grades
(%)Grades
3 and 4
(%)Chemistry Hyperglycemia 63 3 47 2 Hypophosphatemia 42 7 10 < 1 Hypoalbuminemia 25 < 1 < 1 0 Hepatic Increased AST 57 6 11 < 1 Increased ALT 48 5 18 < 1 Increased blood alkaline phosphatase 38 1 6 < 1 Hematology Neutropenia 47 6 12 < 1 Lymphopenia 26 5 6 < 1 Anemia 25 < 1 6 < 1 Trial COMBI-APlus (Pyrexia Management Study)
COMBI-APlus evaluated the impact of pyrexia-related outcomes of a revised pyrexia management algorithm in patients who received dabrafenib administered with trametinib in the adjuvant treatment of BRAF V600 mutation-positive melanoma after complete resection. The pyrexia management algorithm interrupted both dabrafenib and trametinib when patient’s temperature is ≥ 100.4°F.
Grade 3-4 pyrexia occurred in 4.3% of patients, hospitalizations due to pyrexia occurred in 5.1% of patients, pyrexia with complications (dehydration, hypotension, renal dysfunction, syncope, severe chills) occurred in 2.2% of patients, and treatment discontinuation due to pyrexia occurred in 2.5% of patients.
Metastatic, BRAF V600E Mutation-Positive Non-Small Cell Lung Cancer
The safety of MEKINIST when administered with dabrafenib was evaluated in 93 patients with previously untreated (n = 36) and previously treated (n = 57) metastatic BRAF V600E mutation-positive NSCLC in a multi-center, multi-cohort, non-randomized, open-label trial (Study BRF113928). Patients received MEKINIST 2 mg orally once daily and dabrafenib 150 mg orally twice daily until disease progression or unacceptable toxicity. The trial excluded patients with abnormal LVEF, history of acute coronary syndrome within 6 months, history of Class II or greater congestive heart failure (New York Heart Association), QTc interval ≥ 480 msec, treatment refractory hypertension, uncontrolled arrhythmias, active brain metastases, history of ILD or pneumonitis, or history or current RVO [see Clinical Studies (14.3)].
Among these 93 patients, 53 (57%) were exposed to MEKINIST and dabrafenib for > 6 months and 27 (29%) were exposed to MEKINIST and dabrafenib for ≥ 1 year. The median age was 65 years (range: 41 to 91), 46% were male, 85% were White; 32% had baseline ECOG performance status of 0 and 61% had ECOG performance status of 1; 98% had non-squamous histology; and 12% were current smokers, 60% were former smokers, and 28% had never smoked.
The most common adverse reactions (≥ 20%) in these 93 patients were: pyrexia, fatigue, nausea, vomiting, diarrhea, dry skin, decreased appetite, edema, rash, chills, hemorrhage, cough, and dyspnea.
Adverse reactions leading to discontinuation of MEKINIST occurred in 19% of patients; the most frequent were pyrexia (2.2%), decreased ejection fraction (2.2%), and respiratory distress (2.2%). Adverse reactions leading to dose reductions of MEKINIST occurred in 30% of patients; the most frequent were pyrexia (5%), nausea (4.3%), vomiting (4.3%), diarrhea (3.2%), and neutropenia (3.2%). Adverse reactions leading to dose interruptions of MEKINIST occurred in 57% of patients; the most frequent were pyrexia (16%), vomiting (10%), neutropenia (8%), nausea (5%), and decreased ejection fraction (5%).
Table 12 and Table 13 present adverse reactions and laboratory abnormalities, respectively, of MEKINIST in combination with dabrafenib in Study BRF113928.
Table 12. Adverse Reactions Occurring in ≥ 20% (All Grades) of Patients Treated with MEKINIST plus Dabrafenib in Study BRF113928a aNCI CTCAE version 4.0.
bIncludes fatigue, malaise, and asthenia.
cIncludes peripheral edema, edema, and generalized edema.
dIncludes rash, rash generalized, rash papular, rash macular, rash maculo-papular, and rash pustular.
eIncludes hemoptysis, hematoma, epistaxis, purpura, hematuria, subarachnoid hemorrhage, gastric hemorrhage, urinary bladder hemorrhage, contusion, hematochezia, injection site hemorrhage, pulmonary hemorrhage, and retroperitoneal hemorrhage.Adverse Reactions MEKINIST plus Dabrafenib
N = 93All
Grades
(%)Grades
3 and 4
(%)General Pyrexia 55 5 Fatigueb 51 5 Edemac 28 0 Chills 23 1.1 Gastrointestinal Nausea 45 0 Vomiting 33 3.2 Diarrhea 32 2.2 Decreased appetite 29 0 Skin and subcutaneous tissue Dry skin 31 1.1 Rashd 28 3.2 Vascular Hemorrhagee 23 3.2 Respiratory system Cough 22 0 Dyspnea 20 5 Other clinically important adverse reactions for MEKINIST in Study BRF113928 observed in less than 20% of patients who received MEKINIST administered with dabrafenib were:
Cardiac: Atrioventricular block
Nervous System: Peripheral neuropathy
Table 13. Treatment-Emergent Laboratory Abnormalities Occurring in ≥ 20% (All Grades) of Patients Who Received MEKINIST plus Dabrafenib in Study BRF113928 Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aFor these laboratory tests, the denominator is 90.
bFor these laboratory tests, the denominator is 91.Laboratory Abnormality MEKINIST plus Dabrafenib
N = 93All
Grades
(%)Grades
3 and 4
(%)Chemistrya Hyperglycemia 71 9 Hyponatremia 57 17 Hypophosphatemia 36 7 Increased creatinine 21 1.1 Hepatica Increased blood alkaline phosphatase 64 0 Increased AST 61 4.4 Increased ALT 32 6 Hematologyb Leukopenia 48 8 Anemia 46 10 Neutropenia 44 8 Lymphopenia 42 14 Advanced BRAF V600E Mutation-Positive Tumors
Study BRF117019
The safety of MEKINIST when administered with dabrafenib was evaluated in a multi-cohort, multi-center, non-randomized, open-label study in adult patients with cancers with the BRAF V600E mutation (Study BRF117019). A total of 206 patients were enrolled in the trial, 36 of whom were enrolled in the ATC cohort, 105 were enrolled in specific solid tumor cohorts, and 65 in other malignancies [see Clinical Studies (14.4, 14.6)]. Patients received MEKINIST 2 mg orally once daily and dabrafenib 150 mg orally twice daily until disease progression or unacceptable toxicity.
Among these 206 patients, 101 (49%) were exposed to MEKINIST for ≥ 1 year and 103 (50%) were exposed to dabrafenib for ≥ 1 year. The median age was 60 years (range: 18 to 89); 56% were male; 79% were White; and 34% had baseline ECOG performance status of 0 and 60% had ECOG performance status of 1.
Serious adverse reactions occurred in 45% of patients who received MEKINIST in combination with dabrafenib. Serious adverse reactions in > 5% of patients included pyrexia (11%) and pneumonia (6%). Fatal adverse reactions occurred in 3.9% of patients who received MEKINIST in combination with dabrafenib. Fatal adverse reactions that occurred in > 1% of patients included sepsis (1.9%).
Permanent treatment discontinuation due to an adverse reaction occurred in 13% of patients. Adverse reactions which resulted in permanent treatment discontinuation in > 1% of patients included nausea (1.5%).
Dosage interruptions due to an adverse reaction occurred in 55% of patients. Adverse reactions which required dosage interruption in > 5% of patients included pyrexia (22%), chills (9%), fatigue (6%), neutropenia (6%), and nausea (5%).
Dose reductions due to an adverse reaction occurred in 44% of patients. Adverse reactions which required dose reductions in > 5% of patients included pyrexia (18%), chills (8%), and fatigue (6%).
The most common (≥ 20%) adverse reactions, including laboratory abnormalities, are listed in Table 14 and Table 15.
Table 14 summarizes the adverse reactions in Study BRF117019.
Table 14. Adverse Reactions (≥ 20%) in Adult Patients Treated with MEKINIST Plus Dabrafenib in Study BRF117019 aNCI CTCAE version 4.0.
bIncludes fatigue, asthenia, and malaise.
cIncludes peripheral edema and peripheral swelling.
dIncludes rash, rash maculo-papular, rash erythematous, rash pustular, and rash papular.
eIncludes epistaxis, hematuria, contusion, hematoma, hemoptysis, conjunctival hemorrhage, hematochezia, rectal hemorrhage, hemorrhoidal hemorrhage, melaena, purpura, eye contusion, eye hemorrhage, gastric hemorrhage, gingival bleeding, hematemesis, hemorrhage intracranial, hemorrhagic stroke, hemothorax, increased tendency to bruise, large intestinal hemorrhage, mouth hemorrhage, petechiae, pharyngeal hemorrhage, prothrombin time prolonged, pulmonary hematoma, retinal hemorrhage, vaginal hemorrhage, and vitreous hemorrhage.
fIncludes cough and productive cough.
gIncludes myalgia, musculoskeletal chest pain, and musculoskeletal pain.Adverse Reactions MEKINIST plus Dabrafeniba
(N = 206)All Grades
(%)Grade 3 or 4
(%)General Pyrexia 55 4.95 Fatigueb 50 5 Chills 30 0.5 Peripheral edemac 22 0 Gastrointestinal Nausea 40 1.5 Constipation 27 0 Vomiting 27 1.5 Diarrhea 26 2.93 Skin and subcutaneous tissue Rashd 40 2.4 Nervous system Headache 30 1.5 Vascular Hemorrhagee 29 4.4 Respiratory system Coughf 29 0 Musculoskeletal and connective tissue Myalgiag 24 0.5 Arthralgia 23 0.5 Clinically relevant adverse reactions for MEKINIST in Study BRF117019 observed in less than 20% of patients who received MEKINIST in combination with dabrafenib were: peripheral neuropathy (9%), decreased ejection fraction (8%), atrioventricular block (2.9%), uveitis (1.9%), hypersensitivity (1.9%) and Guillain-Barré syndrome (< 1%).
Table 15 summarizes the laboratory abnormalities in Study BRF117019.
Table 15. Select Laboratory Abnormalities (≥ 20%) That Worsened from Baseline in Adult Patients Treated with MEKINIST Plus Dabrafenib in Study BRF117019 Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aThe denominator used to calculate the rate varied from 199 to 202 based on the number of patients with a baseline value and at least one post-treatment value.Laboratory Abnormality MEKINIST plus Dabrafeniba All Grades
(%)Grade 3 or 4
(%)Chemistry Hyperglycemia 61 8 Decreased sodium 35 10 Decreased magnesium 24 0 Increased creatinine 21 1.5 Hepatic Increased alkaline phosphatase 51 5 Increased AST 51 4.6 Increased ALT 39 3 Hematology Decreased hemoglobin 44 9 BRAF V600E Mutation-Positive Solid Tumors in Pediatric Patients
Study CTMT212X2101 (X2101)
The safety of MEKINIST when administered with dabrafenib was evaluated in Study X2101, a multi-center, open-label, multi-cohort study in pediatric patients (n = 48) with refractory or recurrent solid tumors activation [see Clinical Studies (14.6)]. The median duration of exposure to MEKINIST in Parts C (dose escalation) and D (cohort expansion) was 20.8 and 24.4 months, respectively. The median duration of exposure to dabrafenib in Parts C and D was 20.8 and 24.9 months, respectively. The median age of pediatric patients who received MEKINIST with dabrafenib was 9 years (range: 1 to 17).
Serious adverse reactions occurred in 46% of patients who received MEKINIST in combination with dabrafenib. Serious adverse reactions in > 5% of patients included pyrexia (25%) and decreased ejection fraction (6%). Permanent treatment discontinuation due to an adverse reaction occurred in 21% of patients. Adverse reactions which resulted in permanent treatment discontinuation in > 3% of patients included increased ALT (6%), increased AST (4.2%) and decreased ejection fraction (4.2%). Dosage interruptions due to an adverse reaction occurred in 73% of patients. Adverse reactions which required dosage interruption in > 5% of patients included pyrexia (56%), vomiting (19%), neutropenia (13%), rash (13%), decreased ejection fraction (6%), and uveitis (6%). Dose reductions due to an adverse reaction occurred in 25% of patients. Adverse reactions which required dose reductions in > 5% of patients included pyrexia (13%).
The most common (≥ 20%) adverse reactions, including laboratory abnormalities, are listed in Table 16 and Table 17.
Table 16 summarizes the adverse reactions in Study X2101.
Table 16. Adverse Reactions (≥ 20%) in Pediatric Patients Treated with MEKINIST Plus Dabrafenib in Study X2101 aNCI CTCAE version 4.0.
bIncludes fatigue, asthenia, and malaise.
cIncludes rash, rash maculo-papular, rash erythematous, rash papular, rash pustular, and rash macular.
dIncludes dermatitis acneiform and acne.
eIncludes abdominal pain and abdominal pain upper.
fIncludes epistaxis, hematuria, contusion, hematoma, petechiae, rectal hemorrhage, and red blood cell count decreased.Adverse Reactions MEKINIST plus Dabrafeniba
(N = 48)All Grades
(%)Grade 3 or 4
(%)General Pyrexia 75 17 Fatigueb 48 0 Skin and subcutaneous tissue Rashc 73 2.1 Dry skin 48 0 Dermatitis acneiformd 40 0 Gastrointestinal Vomiting 52 4.2 Diarrhea 42 2.1 Abdominal paine 33 4.2 Nausea 33 2.1 Constipation 23 0 Respiratory system Cough 44 0 Nervous system Headache 35 0 Vascular Hemorrhagef 33 0 Infections and infestations Paronychia 23 0 Clinically relevant adverse reactions for MEKINIST in Study X2101 observed in less than 20% of patients (N=48) who received MEKINIST in combination with dabrafenib were: atrioventricular block (2.1%).
Table 17 summarizes the laboratory abnormalities in Study X2101.
Table 17. Select Laboratory Abnormalities (≥ 20%) That Worsened from Baseline in Pediatric Patients Treated with MEKINIST Plus Dabrafenib in Study X2101 Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aThe denominator used to calculate the rate varied from 39 to 48 based on the number of patients with a baseline value and at least one post-treatment value.Laboratory Abnormality MEKINIST plus Dabrafeniba All Grades
(%)Grade 3 or 4
(%)Chemistry Hyperglycemia 65 2.2 Hypoalbuminemia 48 2.1 Hypocalcemia 40 2.1 Decreased phosphate 38 0 Decreased magnesium 33 2.1 Hypernatremia 27 0 Hypokalemia 21 2.1 Hepatic Increased AST 55 4.2 Increased ALT 40 6 Increased alkaline phosphatase 28 6 Increased total bilirubin 21 2.1 Hematology Decreased hemoglobin 60 6 Decreased neutrophils 49 28 BRAF V600E Mutation-Positive Low-Grade Glioma in Pediatric Patients
Study CDRB436G2201 (G2201)
The safety of MEKINIST in combination with dabrafenib was evaluated in pediatric patients 1 to < 18 years of age in Study G2201. Patients with low-grade glioma (LGG) who required first systemic therapy were randomized (2:1) to MEKINIST plus dabrafenib (n = 73) or carboplatin plus vincristine (n = 33). Nine patients crossed over from the carboplatin plus vincristine arm to the MEKINIST and dabrafenib arm. Pediatric patients received weight-based MEKINIST orally once daily administered in combination with dabrafenib until disease progression or intolerable toxicity. Patients in the control arm received carboplatin and vincristine at doses of 175 mg/m2 and 1.5 mg/m2, respectively in 10-week induction course followed by eight 6-week cycles of maintenance therapy or until disease progression or intolerable toxicity. Among patients with low-grade glioma who were randomized to MEKINIST plus dabrafenib (n = 73), 95% were exposed for 6 months or longer and 71% were exposed for greater than one year.
The median age of these patients was 10 years (range: 1 to 17); 60% female; 75% White, 7% Asian, 2.7% Black or African American, 4% other race, and 11% where race was unknown or not reported.
Serious adverse reactions occurred in 40% of these patients. Serious adverse reactions in > 3% of patients included pyrexia (14%) and vomiting (4%).
Permanent discontinuation of MEKINIST due to an adverse reaction occurred in 4% of patients. Adverse reactions which resulted in permanent discontinuation of MEKINIST included chills, fatigue, pyrexia, weight increased, and headache.
Dosage interruptions of MEKINIST due to an adverse reaction occurred in 70% of patients. Adverse reactions which required a dosage interruption in > 5% of patients included pyrexia (52%).
Dose reductions of MEKINIST due to an adverse reaction occurred in 12% of patients. Adverse reactions which required dose reductions in > 2% of patients included weight increased (2.7%).
The most common (≥ 15%) adverse reactions were pyrexia (68%), rash (51%), headache (47%), vomiting (34%), musculoskeletal pain (34%), fatigue (33%), diarrhea (29%), dry skin (26%), nausea (25%), hemorrhage (25%), abdominal pain (25%), dermatitis acneiform (22%), dizziness (15%), upper respiratory tract infection (15%), and weight increased (15%).
The most common (≥ 20%) laboratory abnormalities that worsened from baseline were leukopenia (59%), increased alkaline phosphatase (55%), anemia (46%), decreased neutrophils (44%), increased AST (37%), decreased magnesium (34%), increased magnesium (32%), decreased platelets (30%), increased ALT (29%), and increased lymphocytes (24%).
Table 18 summarizes the adverse reactions in Study G2201.
Table 18. Adverse Reactions (≥ 15%) in Pediatric LGG Patients Who Received MEKINIST in Combination with Dabrafenib in Study G2201a aNCI CTCAE version 4.03.
bIncludes diarrhea, colitis, enterocolitis, and enteritis.
cIncludes abdominal pain and upper abdominal pain.
dIncludes stomatitis, cheilitis, mouth ulceration, aphthous ulcer, and glossitis.
eIncludes pyrexia and body temperature increased.
fIncludes fatigue and asthenia.
gIncludes headache and migraine with aura.
hIncludes dizziness and vertigo.
iIncludes peripheral neuropathy, peripheral motor neuropathy, peripheral sensorimotor neuropathy, paresthesia, neuralgia, hypoaesthesia, and peripheral sensory neuropathy.
jIncludes epistaxis, post-procedural hemorrhage, hematuria, upper gastrointestinal hemorrhage, and hemorrhage intracranial.
kIncludes rash, rash macular, rash maculo-papular, rash pustular, rash papular, rash erythematous, eczema, erythema multiforme, dermatitis, dermatitis exfoliative, skin exfoliation, palmar-plantar erythrodysaesthesia syndrome, and dermatitis bullous.
lIncludes dermatitis acneiform, acne, and acne pustular.
mIncludes back pain, myalgia, pain in extremity, arthralgia, bone pain, non-cardiac chest pain, neck pain, and musculoskeletal stiffness.Adverse Reactions MEKINIST plus Dabrafenib
N = 73Carboplatin plus Vincristine
N = 33All Grades
(%)Grade ≥ 3
(%)All Grades
(%)Grade ≥ 3
(%)Gastrointestinal Vomiting 34 1 48 3 Diarrheab 29 0 18 6 Nausea 25 0 45 0 Abdominal painc 25 0 24 0 Constipation 12 0 36 0 Stomatitisd 10 0 18 0 General Pyrexiae 68 8 18 3 Fatiguef 33 0 39 0 Nervous system Headacheg 47 1 33 3 Dizzinessh 15 0 9 3 Peripheral neuropathyi 7 0 45 6 Vascular Hemorrhagej 25 0 12 0 Skin and subcutaneous tissue Rashk 51 2.7 18 3 Dry skin 26 0 3 0 Dermatitis acneiforml 22 0 0 0 Alopecia 3 0 24 0 Musculoskeletal and connective tissue Musculoskeletal painm 34 0 30 0 Pain in jaw 1.4 0 18 0 Metabolism and nutrition Decreased appetite 5 0 24 0 Respiratory, thoracic and mediastinal Oropharyngeal pain 11 0 18 0 Psychiatric Anxiety 1.4 0 15 3 Immune system Hypersensitivity 0 0 15 3 Infections and infestations Upper respiratory tract infection 15 0 6 0 Injury, poisoning and procedural complications Infusion-related reaction 0 0 15 3 Investigations Weight increased 15 7 0 0 Table 19 summarizes the laboratory abnormalities in Study G2201.
Table 19. Select Laboratory Abnormalities (≥ 20%) That Worsened from Baseline in Pediatric LGG Patients Who Received MEKINIST in Combination with Dabrafenib in Study G2201a Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aThe denominator used to calculate the rate varied from 70 to 73 in D + T arm and 9 to 33 in C + V arm based on the number of patients with a baseline value and at least one post-treatment value.Laboratory Abnormality MEKINIST plus Dabrafenib
N = 73Carboplatin plus Vincristine
N = 33All Grades
(%)Grade 3 or 4
(%)All Grades
(%)Grade 3 or 4
(%)Hepatic Increased alkaline phosphatase 55 0 13 0 Increased AST 37 1.4 55 0 Increased ALT 29 3 61 9 Chemistry Decreased magnesium 34 4.1 76 6 Increased magnesium 32 0 24 3 Increased potassium 15 4.2 21 6 Decreased calcium 14 4.1 22 9 Decreased potassium 8 1.4 70 0 Decreased phosphate 7 2.7 33 3 Decreased sodium 5 1.4 27 6 Increased serum fasting glucose 0 0 44 0 Hematology Decreased leukocytes 59 0 91 18 Decreased hemoglobin 46 0 94 36 Decreased neutrophils 44 17 84 75 Decreased platelets 30 0 73 18 Increased lymphocytes 24 0 13 3.1 Decreased lymphocytes 16 1.4 56 6 6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of MEKINIST in combination with dabrafenib. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac: Atrioventricular block complete. This adverse reaction was also observed with MEKINIST monotherapy.
Immune System: Hemophagocytic lymphohistiocytosis (HLH) [see Warnings and Precautions (5.12)]
Skin and Subcutaneous Tissue: SCAR (including DRESS and SJS) [see Warnings and Precautions (5.9)]
- New Primary Malignancies [see Warnings and Precautions (5.1)]
- 7 DRUG INTERACTIONS
-
8
USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on its mechanism of action [see Clinical Pharmacology (12.1)] and findings from animal reproduction studies, MEKINIST can cause fetal harm when administered to a pregnant woman. There is insufficient data in pregnant women exposed to MEKINIST to assess the risks. Trametinib was embryotoxic and abortifacient in rabbits at doses greater than or equal to those resulting in exposures approximately 0.3 times the human exposure at the recommended adult clinical dose (see Data). Advise pregnant women of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In reproductive toxicity studies, administration of trametinib to rats during the period of organogenesis resulted in decreased fetal weights at doses greater than or equal to 0.031 mg/kg/day [approximately 0.3 times the human exposure at the recommended adult dose based on area under the curve (AUC)]. In rats, at a dose resulting in exposures 1.8-fold higher than the human exposure at the recommended adult dose, there was maternal toxicity and an increase in post-implantation loss.
In pregnant rabbits, administration of trametinib during the period of organogenesis resulted in decreased fetal body weight and increased incidence of variations in ossification at doses greater than or equal to 0.039 mg/kg/day (approximately 0.08 times the human exposure at the recommended adult dose based on AUC). In rabbits administered trametinib at 0.15 mg/kg/day (approximately 0.3 times the human exposure at the recommended adult dose based on AUC) there was an increase in post-implantation loss, including total loss of pregnancy, compared with control animals.
8.2 Lactation
Risk Summary
There are no data on the presence of trametinib in human milk, or the effects of trametinib on the breastfed child or on milk production. Because of the potential for serious adverse reactions in breastfed children, advise women not to breastfeed during treatment with MEKINIST and for 4 months following the last dose.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to initiating MEKINIST.
Contraception
Based on data from animal studies and its mechanism of action, MEKINIST can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1)].
Females
Advise female patients of reproductive potential to use effective contraception during treatment with MEKINIST and for 4 months after the last dose.
Males
To avoid potential drug exposure to pregnant partners and female partners of reproductive potential, advise male patients (including those who have had vasectomies) with female partners of reproductive potential to use condoms during treatment with MEKINIST and for 4 months after the last dose.
Infertility
Females
Advise female patients of reproductive potential that MEKINIST may impair fertility. Increased follicular cysts and decreased corpora lutea were observed in female rats at dose exposures equivalent to 0.3 times the human exposure at the recommended adult dose [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
BRAF V600E Mutation-Positive Unresectable or Metastatic Solid Tumors and LGG
The safety and effectiveness of MEKINIST in combination with dabrafenib have been established in pediatric patients 1 year of age and older with unresectable or metastatic solid tumors with BRAF V600E mutation who have progressed following prior treatment and have no satisfactory alternative treatment options; or with LGG with BRAF V600E mutation who require systemic therapy. Use of MEKINIST in combination with dabrafenib for these indications is supported by evidence from studies X2101 and G2201 that enrolled 171 patients (1 to < 18 years of age) with BRAF V600 mutation-positive advanced solid tumors, of which 4 (2.3%) patients were 1 to < 2 years of age, 39 (23%) patients were 2 to < 6 years of age, 54 (32%) patients were 6 to < 12 years of age, and 74 (43%) patients were 12 to < 18 years of age [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.6, 14.7)].
The safety and effectiveness of MEKINIST in combination with dabrafenib have not been established for these indications in pediatric patients less than 1 year old.
The safety and effectiveness of MEKINIST as a single agent in pediatric patients have not been established.
Juvenile Animal Toxicity Data
In a repeat-dose toxicity study in juvenile rats, decreased bone length and corneal dystrophy were observed at doses resulting in exposures as low as 0.3 times the human exposure at the recommended adult dose based on AUC. Additionally, a delay in sexual maturation was noted at doses resulting in exposures as low as 1.6 times the human exposure at the recommended adult dose based on AUC.
8.5 Geriatric Use
Of the 214 patients with melanoma who received single agent MEKINIST in the METRIC study, 27% were aged 65 years and older and 4% were over 75 years old [see Clinical Studies (14.1)]. This study of single agent MEKINIST in melanoma did not include sufficient numbers of geriatric patients to determine whether they respond differently from younger adults.
Of the 994 patients with melanoma who received MEKINIST plus dabrafenib in the COMBI-d, COMBI-v, and COMBI-AD studies [see Clinical Studies (14.1, 14.2)], 21% were aged 65 years and older and 5% were aged 75 years and older. No overall differences in the effectiveness of MEKINIST plus dabrafenib were observed in geriatric patients as compared to younger adults across these melanoma studies. The incidences of peripheral edema (26% vs. 12%) and anorexia (21% vs. 9%) increased in geriatric patients as compared to younger adults in these studies.
Of the 93 patients with NSCLC who received MEKINIST in Study BRF113928, there were insufficient numbers of geriatric patients aged 65 years and older to determine whether they respond differently from younger adults [see Clinical Studies (14.4)].
Of the 26 patients with ATC who received MEKINIST in Study BRF117019, 77% were aged 65 years and older and 31% were aged 75 years and older [see Clinical Studies (14.4)]. This study in ATC did not include sufficient numbers of younger adults to determine whether they respond differently compared to geriatric patients.
8.6 Hepatic Impairment
No dose adjustment is recommended in patients with mild (bilirubin ≤ upper limit of normal (ULN) and aspartate aminotransferase (AST) > ULN or bilirubin > 1x to 1.5x ULN and any AST) hepatic impairment.
A recommended dosage of MEKINIST has not been established for patients with moderate (bilirubin > 1.5x to 3x ULN and any AST) or severe (bilirubin > 3x to 10x ULN and any AST) hepatic impairment. Consider the risk-benefit profile of MEKINIST related to dosing prior to determining whether to administer MEKINIST to patients with moderate or severe hepatic impairment.
In patients with moderate hepatic impairment, 3 patients who received a starting dose of 1.5 mg orally once daily and two patients who received a starting dose of 2 mg orally once daily did not experience dose limiting toxicities (DLTs) during the first cycle of therapy.
In patients with severe hepatic impairment, 3 patients who received a starting dose of 1 mg orally once daily did not experience DLTs during the first cycle; one patient who received a starting dose of 1.5 mg orally once daily experienced a DLT (grade 3 acneiform rash).
Compared to patients with normal hepatic function, there was no increase in exposure of trametinib in patients with moderate or severe hepatic impairment [see Clinical Pharmacology (12.3)].
-
10
OVERDOSAGE
The highest doses of MEKINIST evaluated in clinical trials were 4 mg orally once daily and 10 mg administered orally once daily on 2 consecutive days followed by 3 mg once daily. In seven patients treated on one of these two schedules, there were two cases of RPEDs for an incidence of 28%.
Since trametinib is highly bound to plasma proteins, hemodialysis is likely to be ineffective in the treatment of overdose with MEKINIST.
-
11
DESCRIPTION
Trametinib dimethyl sulfoxide is a kinase inhibitor. The chemical name is acetamide, N-[3-[3-cyclopropyl-5-[(2-fluoro-4- iodophenyl)amino]-3,4,6,7-tetrahydro-6,8-dimethyl- 2,4,7-trioxopyrido[4,3-d]pyrimidin-1(2H)-yl]phenyl]-, compound with 1,1’-sulfinylbis[methane] (1:1). It has a molecular formula C26H23FIN5O4•C2H6OS with a molecular mass of 693.53 g/mol. Trametinib dimethyl sulfoxide has the following chemical structure:
Trametinib dimethyl sulfoxide is a white to almost white powder. It is practically insoluble in the pH range of 2 to 8 in aqueous media.
MEKINIST (trametinib) tablets for oral use are supplied as 0.5 mg and 2 mg tablets for oral administration. Each 0.5 mg tablet contains 0.5635 mg trametinib dimethyl sulfoxide equivalent to 0.5 mg of trametinib non-solvated parent. Each 2 mg tablet contains 2.254 mg trametinib dimethyl sulfoxide equivalent to 2 mg of trametinib non-solvated parent.
The inactive ingredients of MEKINIST tablets are: Tablet Core: colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate (vegetable source), mannitol, microcrystalline cellulose, and sodium lauryl sulfate. Coating: hypromellose, iron oxide red (2 mg tablets), iron oxide yellow (0.5 mg tablets), polyethylene glycol, polysorbate 80 (2 mg tablets), and titanium dioxide.
MEKINIST (trametinib) for oral solution is a white or almost white powder which produces a clear colorless solution when reconstituted with water. Each bottle contains 4.7 mg of trametinib equivalent to 5.3 mg trametinib dimethyl sulfoxide. Each mL of reconstituted trametinib solution contains 0.05 mg of trametinib non-solvated parent. The inactive ingredients of MEKINIST for oral solution are betadex sulfobutyl ether sodium, citric acid monohydrate, dibasic sodium phosphate, methylparaben, potassium sorbate, sucralose, and strawberry flavor.
-
12
CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Trametinib is a reversible inhibitor of mitogen-activated extracellular signal-regulated kinase 1 (MEK1) and MEK2 activation and of MEK1 and MEK2 kinase activity. MEK proteins are upstream regulators of the extracellular signal-related kinase (ERK) pathway, which promotes cellular proliferation. BRAF V600E mutations result in constitutive activation of the BRAF pathway which includes MEK1 and MEK2. Trametinib inhibits cell growth of various BRAF V600 mutation-positive tumors in vitro and in vivo.
Trametinib and dabrafenib target two different kinases in the RAS/RAF/MEK/ERK pathway. Use of trametinib and dabrafenib in combination resulted in greater growth inhibition of BRAF V600 mutation-positive tumor cell lines in vitro and prolonged inhibition of tumor growth in BRAF V600 mutation-positive tumor xenografts compared with either drug alone.
In the setting of BRAF-mutant colorectal cancer, induction of EGFR-mediated MAPK pathway re-activation has been identified as a mechanism of intrinsic resistance to BRAF inhibitors [see Indications and Usage (1.7)].
12.2 Pharmacodynamics
Administration of MEKINIST tablets 1 mg and 2 mg to patients with BRAF V600 mutation-positive melanoma resulted in dose-dependent changes in tumor biomarkers, including inhibition of phosphorylated ERK, inhibition of Ki67 (a marker of cell proliferation), and increases in p27 (a marker of apoptosis).
Cardiac Electrophysiology
The heart rate-corrected QT (QTc) prolongation potential of trametinib was assessed in a dedicated study in 32 patients who received placebo on Day 1 and MEKINIST tablets 2 mg once daily on Days 2-14 followed by MEKINIST tablets 3 mg on Day 15. No large changes in the mean QTc interval (i.e., > 20 ms) were detected in the study.
A decrease from baseline in HR by 9 beats/min (90% CI: -11.4 to -6.1) and an increase from baseline in PR by 20 ms (90% CI: 13.0 to 27.4) relative to placebo was observed at two hours post-dose in the same study.
In clinical trials in patients who received MEKINIST with dabrafenib, QTc prolongation > 500 ms occurred in 0.8% of patients and QTc increased by > 60 ms from baseline in 3.8% of patients.
12.3 Pharmacokinetics
The pharmacokinetics of trametinib were characterized following a single dose and multiple doses in patients with solid tumors and BRAF V600 mutation-positive metastatic melanoma. Following administration of MEKINIST tablets 0.125 mg (0.0625 times the approved recommended adult dosage) to 4 mg (2 times the approved recommended adult dosage) daily, both Cmax and AUC increase proportionally with dose. Intersubject variability in AUC and Cmax at steady state is 22% and 28%, respectively.
Absorption
The median time to achieve peak plasma concentrations (Tmax) is 1.5 hours post-dose. The mean absolute bioavailability of MEKINIST tablets is 72% and MEKINIST for oral solution is 81%.
Effect of Food
Following administration of MEKINIST tablets, a high-fat, high-calorie meal (approximately 1000 calories) decreased trametinib AUC by 24%, Cmax by 70%, and delayed Tmax by approximately 4 hours as compared with fasted conditions.
Administration of a single 2 mg dose of MEKINIST for oral solution with a low-fat, low-calorie meal (approximately 500 calories, 14 grams fat, 80 grams carbohydrates, and 12 grams protein) showed no clinically significant changes on both AUC and Cmax of trametinib as compared with fasted conditions.
Distribution
Trametinib is 97.4% bound to human plasma proteins. The apparent volume of distribution (Vc/F) is 214 L.
Elimination
The estimated elimination half-life is 3.9 to 4.8 days. The apparent clearance is 4.9 L/h.
Metabolism
Trametinib is metabolized predominantly via deacetylation alone or with mono-oxygenation or in combination with glucuronidation biotransformation pathways in vitro. Deacetylation is mediated by carboxylesterases (i.e., carboxylesterase 1b/c and 2) and may also be mediated by other hydrolytic enzymes.
Following a single dose of [14C]-trametinib, approximately 50% of circulating radioactivity is represented as the parent compound; however, ≥ 75% of drug-related material in plasma is the parent compound based on metabolite profiling after repeat dosing of trametinib.
Excretion
Following oral administration of [14C]-trametinib, greater than 80% of excreted radioactivity was recovered in the feces while less than 20% of excreted radioactivity was recovered in the urine with less than 0.1% of the excreted dose as parent.
Specific Populations
Age (18 to 93 years), sex, body weight (36 to 170 kg), and renal impairment (eGFR 15 to 89 mL/min/1.73 m2) have no clinically significant effect on the exposure of trametinib. There are insufficient data to evaluate potential differences in the exposure of trametinib by race or ethnicity.
Pediatric Patients: The pharmacokinetics of trametinib in glioma and other solid tumors were evaluated in 244 patients aged 1 to < 18 years following a single dose or multiple doses. Pharmacokinetic parameters in patients aged 1 to < 18 years are within range of values previously observed in adults given the same dose based on weight. Weight (6 to 156 kg) was found to have a statistically significant effect on trametinib oral clearance in this population.
Patients with Hepatic Impairment: Hepatic impairment (defined by bilirubin and AST levels) had no significant effect in trametinib exposure or apparent drug clearance compared with patients with normal hepatic function.
Drug Interaction Studies
Effect of Dabrafenib on Trametinib: Coadministration of MEKINIST tablets 2 mg daily with dabrafenib resulted in no change in AUC of trametinib.
Effect of Trametinib on CYP Substrates: Coadministration of MEKINIST tablets 2 mg once daily with a sensitive CYP3A4 substrate had no clinically relevant effect on the AUC and Cmax of the sensitive CYP3A4 substrate.
Based on in vitro studies, trametinib is an inhibitor of CYP2C8, but is not an inhibitor of CYP1A2, CYP2A6, CYP2B6, CYP2C9, CYP2C19, or CYP2D6 at a clinically relevant systemic concentration.
Effect of Transporters on Trametinib: Trametinib is a substrate of P-glycoprotein (P-gp) and BSEP. Inhibition of P-gp is unlikely to result in a clinically important increase in trametinib concentrations as trametinib exhibits high passive permeability and bioavailability. Trametinib is not a substrate of BCRP, OATP1B1, OATP1B3, OATP2B1, OCT1, MRP2, or MATE1 in vitro.
Effect of Trametinib on Transporters: Based on in vitro studies, trametinib is not an inhibitor of P-gp, BCRP, OATP1B1, OATP1B3, OAT1, OAT3, OCT2, BSEP, MRP2, or MATE1 at a clinically relevant systemic concentration.
-
13
NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies with trametinib have not been conducted. Trametinib was not genotoxic in studies evaluating reverse mutations in bacteria, chromosomal aberrations in mammalian cells, and micronuclei in the bone marrow of rats.
Trametinib may impair fertility in humans. In female rats given trametinib for up to 13 weeks, increased follicular cysts and decreased corpora lutea were observed at doses ≥ 0.016 mg/kg/day (approximately 0.3 times the human exposure at the recommended adult dose based on AUC). In rat and dog toxicity studies up to 13 weeks in duration, there were no treatment effects observed on male reproductive tissues [see Use in Specific Populations (8.3)].
-
14
CLINICAL STUDIES
14.1 BRAF V600E or V600K Mutation-Positive Unresectable or Metastatic Melanoma
MEKINIST as a Single Agent
The safety and efficacy of MEKINIST were evaluated in an international, multi-center, randomized (2:1), open-label, active-controlled trial (the METRIC study; NCT01245062) in 322 patients with BRAF V600E or V600K mutation-positive, unresectable or metastatic melanoma. In the METRIC study, patients were not permitted to have more than one prior chemotherapy regimen for advanced or metastatic disease; prior treatment with a BRAF inhibitor or MEK inhibitor was not permitted. Patients were randomized to receive MEKINIST 2 mg orally once daily (N = 214) or chemotherapy (N = 108) consisting of either dacarbazine 1000 mg/m2 intravenously every 3 weeks or paclitaxel 175 mg/m2 intravenously every 3 weeks. Treatment continued until disease progression or unacceptable toxicity. Randomization was stratified according to prior use of chemotherapy for advanced or metastatic disease (yes vs. no) and LDH level (normal vs. greater than ULN). Tumor tissue was evaluated for BRAF mutations at a central testing site using a clinical trial assay. Tumor samples from 289 patients (196 patients treated with MEKINIST and 93 chemotherapy-treated patients) were also tested retrospectively using an FDA-approved companion diagnostic test, THxID®-BRAF assay. The major efficacy outcome measure was progression-free survival (PFS) as assessed by the investigator.
The median age for randomized patients was 54 years, 54% were male, greater than 99% were White, and all patients had baseline ECOG performance status of 0 or 1. Most patients had metastatic disease (94%), had M1c disease (64%), had elevated LDH (36%), had no history of brain metastasis (97%), and received no prior chemotherapy for advanced or metastatic disease (66%). The distribution of BRAF V600 mutations was BRAF V600E (87%), V600K (12%), or both (less than 1%). The median durations of follow-up prior to initiation of alternative treatment were 4.9 months for patients treated with MEKINIST and 3.1 months for patients treated with chemotherapy. Fifty-one (47%) patients crossed over from the chemotherapy arm at the time of disease progression to receive MEKINIST.
The METRIC study demonstrated a statistically significant increase in PFS in the patients treated with MEKINIST. Table 20 and Figure 1 summarize the PFS results.
Table 20. Efficacy Results in the METRIC Study Abbreviations: CI, confidence interval; DoR, duration of response; HR, hazard ratio; NR, not reached.
aPike estimator.Investigator-Assessed Endpoints MEKINIST
N = 214Chemotherapy
N = 108Progression-Free Survival Number of events (%) 117 (55%) 77 (71%) Progressive disease 107 (50%) 70 (65%) Death 10 (5%) 7 (6%) Median, months (95% CI) 4.8 (4.3, 4.9) 1.5 (1.4, 2.7) HRa (95% CI) 0.47 (0.34, 0.65) P-value (log-rank test) < 0.0001 Confirmed Tumor Responses Overall response rate (95% CI) 22% (17%, 28%) 8% (4%, 15%) Complete response, n (%) 4 (2%) 0 Partial response, n (%) 43 (20%) 9 (8%) Duration of Response Median DoR, months (95% CI) 5.5 (4.1, 5.9) NR (3.5, NR) 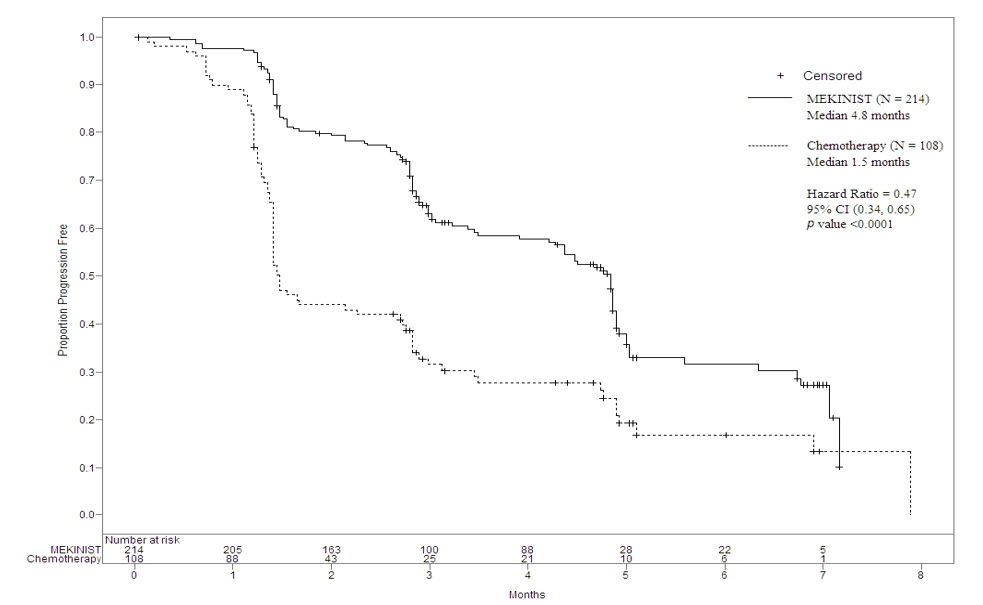
Figure 1. Kaplan-Meier Curves of Investigator-Assessed Progression-Free Survival (ITT Population) in the METRIC Study
In supportive analyses based on independent radiologic review committee (IRRC) assessment, the PFS results were consistent with those of the primary efficacy analysis.
MEKINIST with Dabrafenib
COMBI-d Study
The safety and efficacy of MEKINIST administered with dabrafenib were evaluated in an international, randomized, double-blind, active-controlled trial (the COMBI-d study; NCT01584648). The COMBI-d study compared dabrafenib plus MEKINIST to dabrafenib plus placebo as first-line treatment for patients with unresectable (Stage IIIC) or metastatic (Stage IV) BRAF V600E or V600K mutation-positive cutaneous melanoma. Patients were randomized (1:1) to receive MEKINIST 2 mg once daily plus dabrafenib 150 mg twice daily or dabrafenib 150 mg twice daily plus matching placebo. Randomization was stratified by LDH level (> ULN vs. ≤ ULN) and BRAF mutation subtype (V600E vs. V600K). The major efficacy outcome measure was investigator-assessed progression-free survival (PFS) per RECIST v1.1 with additional efficacy outcome measures of overall survival (OS) and confirmed overall response rate (ORR).
In the COMBI-d study, 423 patients were randomized to MEKINIST plus dabrafenib (n = 211) or dabrafenib plus placebo (n = 212). The median age was 56 years (range: 22 to 89), 53% were male, > 99% were White, 72% had ECOG performance status of 0, 4% had Stage IIIC, 66% had M1c disease, 65% had normal LDH, and 2 patients had a history of brain metastases. All patients had tumor containing BRAF V600E or V600K mutations as determined by centralized testing with the FDA-approved companion diagnostic test; 85% had BRAF V600E mutation-positive melanoma and 15% had BRAF V600K mutation-positive melanoma.
The COMBI-d study demonstrated statistically significant improvements in PFS and OS. Table 21 and Figure 2 summarize the efficacy results.
Table 21. Efficacy Results in the COMBI-d Study Abbreviations: CI, confidence interval; DoR, duration of response; HR, hazard ratio; NR, not reached; ORR, overall response rate.
aPFS and ORR were assessed by investigator.
bBased on stratified log-rank test.Endpoint MEKINIST plus Dabrafenib
N = 211Placebo plus Dabrafenib
N = 212Progression-Free Survivala Number of events (%) 102 (48%) 109 (51%) Median, months (95% CI) 9.3 (7.7, 11.1) 8.8 (5.9, 10.9) HR (95% CI) 0.75 (0.57, 0.99) P-valueb 0.035 Overall Survival Number of deaths (%) 99 (47%) 123 (58%) Median, months (95% CI) 25.1 (19.2, NR) 18.7 (15.2, 23.1) HR (95% CI) 0.71 (0.55, 0.92) P-valueb 0.01 Overall Response Ratea ORR (95% CI) 66% (60%, 73%) 51% (44%, 58%) P-value < 0.001 Complete response 10% 8% Partial response 56% 42% Median DoR, months (95% CI) 9.2 (7.4, NR) 10.2 (7.5, NR) 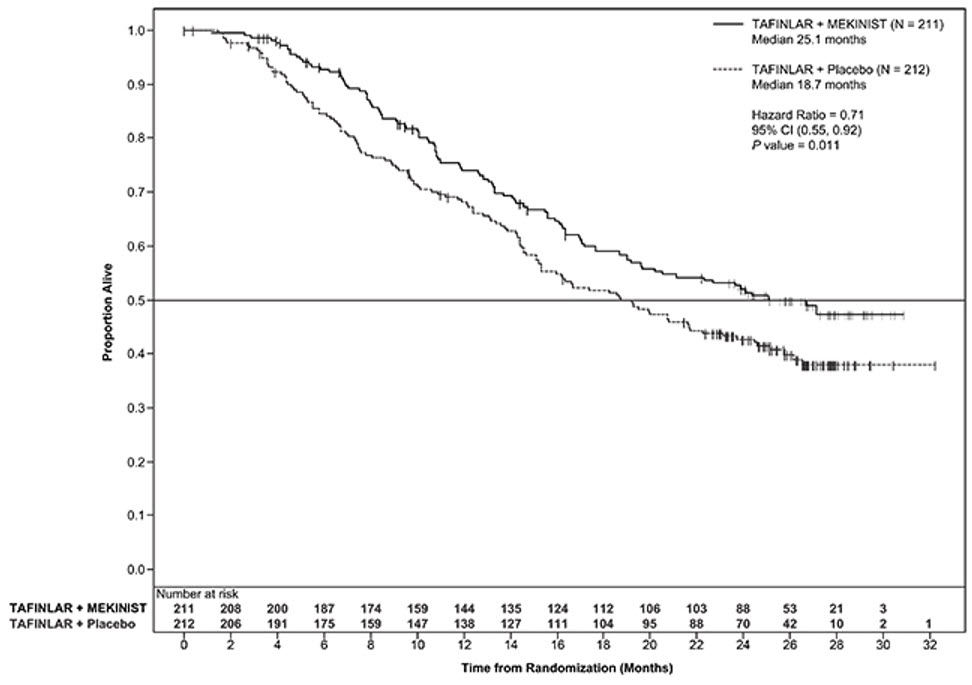
Figure 2. Kaplan-Meier Curves of Overall Survival in the COMBI-d Study
COMBI-MB Study
The activity of MEKINIST with dabrafenib for the treatment of BRAF V600E or V600K mutation-positive melanoma, metastatic to the brain, was evaluated in a non-randomized, open-label, multi-center, multi-cohort trial (the COMBI-MB study; NCT02039947). Eligible patients were required to have at least one measurable intracranial lesion and to have no leptomeningeal disease, parenchymal brain metastasis greater than 4 cm in diameter, ocular melanoma, or primary mucosal melanoma. Patients received MEKINIST 2 mg orally once daily and dabrafenib 150 mg orally twice daily until disease progression or unacceptable toxicity. The major efficacy outcome measure was intracranial response rate, defined as the percentage of patients with a confirmed intracranial response per RECIST v1.1, modified to allow up to five intracranial target lesions at least 5 mm in diameter, as assessed by independent review.
The COMBI-MB study enrolled 121 patients with a BRAF V600E (85%) or V600K (15%) mutation. The median age was 54 years (range: 23 to 84), 58% were male, 100% were White, 8% were from the United States, 65% had normal LDH at baseline, and 97% had an ECOG performance status of 0 or 1. Intracranial metastases were asymptomatic in 87% and symptomatic in 13% of patients, 22% received prior local therapy for brain metastases, and 87% also had extracranial metastases.
The intracranial response rate was 50% (95% CI: 40, 60), with a complete response rate of 4.1% and a partial response rate of 46%. The median duration of intracranial response was 6.4 months (range: 1 to 31). Of the patients with an intracranial response, 9% had stable or progressive disease as their best overall response.
14.2 Adjuvant Treatment of BRAF V600E or V600K Mutation-Positive Melanoma
The efficacy of MEKINIST administered with dabrafenib was evaluated in an international, multi-center, randomized, double-blind, placebo-controlled trial (COMBI-AD; NCT01682083) that enrolled patients with Stage III melanoma with BRAF V600E or V600K mutations as detected by the THxID®-BRAF assay and pathologic involvement of regional lymph node(s). Enrollment required complete resection of melanoma with complete lymphadenectomy within 12 weeks prior to randomization. The trial excluded patients with mucosal or ocular melanoma, unresectable in-transit metastases, distant metastatic disease, or prior systemic anti-cancer treatment, including radiotherapy. Patients were randomized (1:1) to receive MEKINIST 2 mg once daily in combination with dabrafenib 150 mg twice daily or two placebos for up to 1 year. Randomization was stratified by BRAF mutation status (V600E or V600K) and American Joint Committee on Cancer (AJCC; 7th Edition) Stage (IIIA, IIIB, or IIIC). The major efficacy outcome measure was relapse-free survival (RFS), defined as the time from randomization to disease recurrence (local, regional, or distant metastasis), new primary melanoma, or death from any cause, whichever occurred first as assessed by the investigator. Patients underwent imaging for tumor recurrence every 3 months for the first two years and every 6 months thereafter.
In COMBI-AD, a total of 870 patients were randomized: 438 to MEKINIST in combination with dabrafenib and 432 to placebo. Median age was 51 years (range: 18 to 89), 55% were male, 99% were White, and 91% had an ECOG performance status of 0. Disease characteristics were AJCC Stage IIIA (18%), Stage IIIB (41%), Stage IIIC (40%), stage unknown (1%); BRAF V600E mutation (91%), BRAF V600K mutation (9%); macroscopic lymph nodes (65%); and tumor ulceration (41%).
The median duration of follow-up at the time of the primary analysis was 2.8 years.
COMBI-AD showed a statistically significant improvement in RFS in patients randomized to MEKINIST in combination with dabrafenib compared to those randomized to placebo. Efficacy results are presented in Table 22 and Figure 3.
Table 22. Efficacy Results in COMBI-AD in the Adjuvant Treatment of Melanoma Abbreviations: HR, hazard ratio; CI, confidence interval; NE, not estimable.
aPike estimator obtained from the stratified log-rank test.
bLog-rank test stratified by disease stage (IIIA vs. IIIB vs. IIIC) and BRAF V600 mutation type (V600E vs. V600K).Investigator-Assessed Endpoint MEKINIST plus Dabrafenib
N = 438Placebo
N = 432Relapse-Free Survival Number of events (%) 166 (38) 248 (57) Median, months (95% CI) NE (44.5, NE) 16.6 (12.7, 22.1) HR (95% CI)a 0.47 (0.39, 0.58) P-valueb < 0.0001 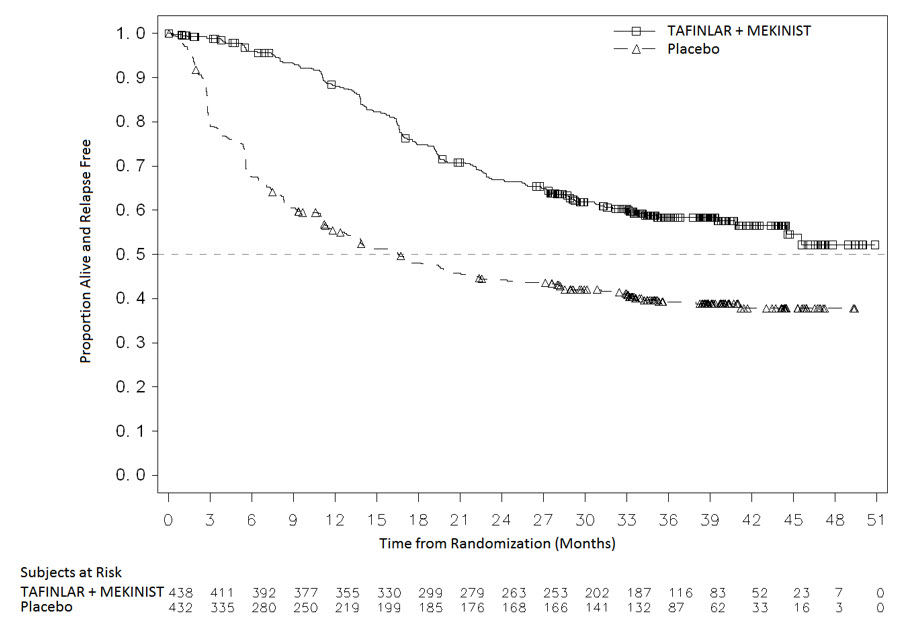
Figure 3. Kaplan-Meier Curves for Investigator-Assessed Relapse-Free Survival in COMBI-AD in the Adjuvant Treatment of Melanoma
The median duration of follow-up at the time of the final overall survival analysis was 8.0 years. The estimated hazard ratio for overall survival was 0.80 (95% CI: 0.62, 1.01; p=0.063) with 125 events (29%) in the combination arm and 136 events (31%) in the placebo arm. Median overall survival was not estimable in both arms.
14.3 BRAF V600E Mutation-Positive Metastatic Non-Small Cell Lung Cancer
The safety and efficacy of dabrafenib alone or administered with MEKINIST were evaluated in a multi-center, three-cohort, non-randomized, activity-estimating, open-label trial (Study BRF113928; NCT01336634). Key eligibility criteria were locally confirmed BRAF V600E mutation-positive metastatic NSCLC, no prior exposure to BRAF or MEK inhibitor, and absence of EGFR mutation or ALK rearrangement (unless patients had progression on prior tyrosine kinase inhibitor therapy). Patients enrolled in Cohorts A and B were required to have received at least one previous platinum-based chemotherapy regimen for NSCLC with demonstrated disease progression but no more than three prior systemic regimens. Patients in Cohort C could not have received prior systemic therapy for metastatic NSCLC. Patients in Cohort A received dabrafenib 150 mg twice daily. Patients in Cohorts B and C received MEKINIST 2 mg once daily and dabrafenib 150 mg twice daily. The major efficacy outcome measure was overall response rate (ORR) per RECIST v1.1 as assessed by independent review committee (IRC) and duration of response (DoR).
There were a total of 171 patients enrolled which included 78 patients enrolled in Cohort A, 57 patients enrolled in Cohort B, and 36 patients enrolled in Cohort C. The characteristics of the study population were: a median age of 66 years; 48% male; 81% White, 14% Asian, 3% Black, and 2% Hispanic; 60% former smokers, 32% never smokers, and 8% current smokers; 27% had ECOG performance status (PS) of 0, 63% had ECOG PS of 1, and 11% had ECOG PS of 2; 99% had metastatic disease of which 6% had brain metastasis at baseline and 14% had liver metastasis at baseline; 11% had systemic anti-cancer therapy in the adjuvant setting and 58% of the 135 previously treated patients had only one line of prior systemic therapy for metastatic disease; 98% had non-squamous histology.
Efficacy results are summarized in Table 23.
Table 23. Efficacy Results Based on Independent Review in Study BRF113928 Abbreviations: CI, confidence interval; DoR, duration of response; ORR, overall response rate.
aRepresents final analysis results (cutoff date of 24 Feb 2021) for the primary analysis responder cohorts.Treatment Dabrafenib MEKINIST plus Dabrafenib Population Previously Treated
N = 78Previously Treated
N = 57Treatment Naïve
N = 36Overall Response Ratea ORR (95% CI) 27% (18%, 38%) 61% (48%, 74%) 61% (44%, 77%) Complete response 1% 5% 8% Partial response 26% 56% 53% Duration of Responsea n = 21 n = 35 n = 22 Median DoR, months (95% CI) 18.0 (4.2, 40.1) 9.0 (5.8, 26.2) 15.2 (7.8, 23.5) In a subgroup analysis of patients with retrospectively centrally confirmed BRAF V600E mutation-positive NSCLC with the Oncomine™ Dx Target Test, the ORR results were similar to those presented in Table 23.
14.4 BRAF V600E Mutation-Positive Locally Advanced or Metastatic Anaplastic Thyroid Cancer
The safety and efficacy of MEKINIST administered with dabrafenib was evaluated in an activity-estimating, nine-cohort, multi-center, non-randomized, open-label trial (Study BRF117019; NCT02034110) in patients with rare cancers with the BRAF V600E mutation, including locally advanced, unresectable, or metastatic ATC with no standard locoregional treatment options. Trial BRF117019 excluded patients who could not swallow or retain the medication; who received prior treatment with BRAF or MEK inhibitors; with symptomatic or untreated CNS metastases; or who had airway obstruction. Patients received MEKINIST 2 mg once daily and dabrafenib 150 mg twice daily. The major efficacy outcome measure was overall response rate (ORR) per RECIST v1.1 as assessed by independent review committee (IRC) and duration of response (DoR).
Thirty-six patients were enrolled and were evaluable for response in the ATC cohort. The median age was 71 years (range: 47 to 85); 44% were male, 50% White, 44% Asian; and 94% had ECOG performance status of 0 or 1. Prior anti-cancer treatments included surgery and external beam radiotherapy (83% each), and systemic therapy (67%).
Efficacy results are summarized in Table 24.
Table 24. Efficacy Results in the ATC Cohort Based on Independent Review of Study BRF117019 Abbreviations: ATC, anaplastic thyroid cancer; CI, confidence interval; DoR, duration of response; NE, not estimable; ORR, overall response rate. ATC Cohort Population N = 36 Overall Response Rate ORR (95% CI) 53% (35.5%, 69.6%) Complete response 6% Partial response 47% Duration of Response n = 19 Median DoR, months (95% CI) 13.6 (3.8, NE) % with DoR ≥ 6 months 68% % with DoR ≥ 12 months 53% 14.5 Lack of Clinical Activity in Metastatic Melanoma Following BRAF-Inhibitor Therapy
The clinical activity of MEKINIST as a single agent was evaluated in a single-arm, multi-center, international trial in 40 patients with BRAF V600E or V600K mutation-positive, unresectable or metastatic melanoma who had received prior treatment with a BRAF inhibitor. All patients received MEKINIST at a dose of 2 mg orally once daily until disease progression or unacceptable toxicity.
The median age was 58 years, 63% were male, all were White, 98% had baseline ECOG PS of 0 or 1, and the distribution of BRAF V600 mutations was V600E (83%), V600K (10%), and the remaining patients had multiple V600 mutations (5%), or unknown mutational status (2%). No patient achieved a confirmed partial or complete response as determined by the clinical investigators.
14.6 BRAF V600E Mutation-Positive Unresectable or Metastatic Solid Tumors
The safety and efficacy of MEKINIST in combination with dabrafenib for the treatment of BRAF V600E mutation-positive unresectable or metastatic solid tumors were evaluated in Trials BRF117019, NCI-MATCH, and CTMT212X2101, and supported by results in COMBI-d, COMBI-v [see Clinical Studies (14.2)], and BRF113928 [see Clinical Studies (14.4)]. In adult studies, patients received MEKINIST 2 mg once daily and dabrafenib 150 mg twice daily. The major efficacy outcome measures were ORR per RECIST v1.1, RANO [HGG] or modified RANO [LGG] criteria and duration of response (DoR).
BRF117019 Study and NCI-MATCH Study
Study BRF117019 (NCT02034110) [see Clinical Studies (14.5)] is a multi-cohort, multi-center, non-randomized, open-label trial in adult patients with selected tumors with the BRAF V600E mutation, including high-grade glioma (HGG) (n = 45), biliary tract cancer (BTC) (n = 43), low-grade glioma (LGG) (n = 13), adenocarcinoma of small intestine (ASI) (n = 3), gastrointestinal stromal tumor (GIST) (n = 1), and anaplastic thyroid cancer [see Clinical Studies (14.5)]. Patients were enrolled based on local assessments of BRAF V600E mutation status; a central laboratory confirmed the BRAF V600E mutation in 93 of 105 patients.
Arm H (EAY131-H) of the NCI-MATCH study (NCT02465060) is a single-arm, open-label study that enrolled patients with a BRAF V600E mutation. Patients with melanoma, thyroid cancer, or CRC were excluded. BRAF V600E mutation status for enrollment was determined either by central or local laboratory test. The study included adult patients with solid tumors including gastrointestinal tumors (n = 14), lung tumors (n = 7), gynecologic or peritoneal tumors (n = 6), CNS tumors (n = 4), and ameloblastoma of mandible (n = 1).
Among the 131 patients enrolled in BRF117019 and NCI-MATCH with the tumor types shown in Table 25, the baseline characteristics were: median age of 51 years with 20% age 65 or older; 56% female; 85% White, 9% Asian, 3% Black, 3% other; and 37% ECOG PS of 0, 56% ECOG PS of 1, and 6% ECOG PS of 2. Of the 131 patients, 90% received prior systemic therapy.
Efficacy results in patients with solid tumors are summarized in Table 25.
Table 25. Efficacy Results Based on Independent Review in Study BRF117019 and NCI-MATCH Arm H Abbreviations: NA, not applicable; PR, partial response.
aExcludes NSCLC (n = 6) and ATC (n = 36) (previously approved tumor types for MEKINIST in combination with dabrafenib).
bMedian DoR 9.8 months (95% CI: 5.3, 20.4).
cMedian DoR 13.6 months (95% CI: 5.5, 26.7).
dDenotes a right-censored DoR.Tumor Typea N Objective Response Rate Duration of Response % 95% CI Range (months) Biliary tract cancerb 48 46 (31, 61) 1.8d, 40d High-grade gliomac 48 33 (20, 48) 3.9, 44 Glioblastoma 32 25 (12, 43) 3.9, 27 Anaplastic pleomorphic xanthoastrocytoma 6 67 (22, 96) 6, 43 Anaplastic astrocytoma 5 20 (0.5, 72) 15 Astroblastoma 2 100 (16, 100) 15, 23d Undifferentiated 1 PR (2.5, 100) 6 Anaplastic ganglioglioma 1 0 NA NA Anaplastic oligodendroglioma 1 0 NA NA Low-grade glioma 14 50 (23, 77) 6, 29d Astrocytoma 4 50 (7, 93) 7, 23 Ganglioglioma 4 50 (7, 93) 6, 13 Pleomorphic xanthoastrocytoma 2 50 (1.3, 99) 6 Pilocytic astrocytoma 2 0 NA NA Choroid plexus papilloma 1 PR (2.5, 100) 29d Gangliocytoma/ganglioglioma 1 PR (2.5, 100) 18d Low-grade serous ovarian carcinoma 5 80 (28, 100) 12, 42d Adenocarcinoma small intestine 4 50 (7, 93) 7, 8 Adenocarcinoma pancreas 3 0 NA NA Mixed ductal/adenoneuroendocrine carcinoma 2 0 NA NA Neuroendocrine carcinoma of colon 2 0 NA NA Ameloblastoma of mandible 1 PR (2.5, 100) 30 Combined small cell-squamous carcinoma of lung 1 PR (2.5, 100) 5 Mucinous-papillary serous adenocarcinoma of peritoneum 1 PR (2.5, 100) 8 Adenocarcinoma of anus 1 0 NA NA Gastrointestinal stromal tumor 1 0 NA NA CTMT212X2101 (X2101) Study
Study X2101 (NCT02124772) was a multi-center, open-label, multi-cohort study in pediatric patients with refractory or recurrent solid tumors. Part C was a dose escalation of MEKINIST in combination with dabrafenib in patients with a BRAF V600E mutation. Part D was a cohort expansion phase of MEKINIST in combination with dabrafenib in patients with LGG with a BRAF V600E mutation. The major efficacy outcome measure was ORR as assessed by independent review committee per RANO criteria.
The efficacy of MEKINIST in combination with dabrafenib was evaluated in 48 pediatric patients, including 34 patients with LGG and 2 patients with HGG.
For patients with BRAF V600E mutation-positive LGG and HGG in Parts C and D, the median age was 10 years (range: 1 to 17); 50% were male, 75% White, 8% Asian, 3% Black; and 58% had Karnofsky/Lansky performance status of 100. Prior anti-cancer treatments included surgery (83%), external beam radiotherapy (2.8%), and systemic therapy (92%). The ORR was 25% (95% CI: 12%, 42%). For the 9 patients who responded, DoR was ≥ 6 months for 78% of patients and ≥ 24 months for 44% of patients.
CDRB436G2201 (G2201) Study – High-Grade Glioma Cohort
Study G2201 (NCT02684058) was a multi-center, randomized, open-label, Phase II study of dabrafenib and trametinib in chemotherapy-naïve pediatric patients with BRAF V600E mutant low-grade glioma (LGG) and patients with relapsed or progressive BRAF V600E mutant HGG. Patients with HGG were enrolled in a single-arm cohort. The major efficacy outcome measure for the HGG cohort was ORR as assessed by independent review committee per RANO 2010 criteria.
The efficacy of MEKINIST in combination with dabrafenib was evaluated in 41 pediatric patients with relapsed or progressive HGG.
For patients with BRAF V600E mutant HGG enrolled in the HGG cohort, the median age was 13 years (range: 2 to 17); 56% were female, 61% White, 27% Asian, 2.4% Black, and 37% had Karnofsky/Lansky performance status of 100. Prior anti-cancer treatments included surgery (98%), radiotherapy (90%), and chemotherapy (81%). The ORR was 56% (95% CI: 40, 72). The median DoR was not reached (95% CI: 9.2, NE). For the 23 patients who responded in the HGG cohort, DoR was ≥ 6 months for 78% of patients, ≥ 12 months for 48% of patients, and ≥ 24 months for 22% of patients.
14.7 BRAF V600E Mutation-Positive Low-Grade Glioma
CDRB436G2201 (G2201) Study – Low-Grade Glioma Cohort
The safety and efficacy of MEKINIST in combination with dabrafenib for the treatment of BRAF V600E mutation-positive low-grade glioma (LGG) in pediatric patients aged 1 to < 18 years of age were evaluated in the multi-center, open-label trial (Study CDRB436G2201; NCT02684058). Patients with LGG (WHO grades 1 and 2) who required first systemic therapy were randomized in a 2:1 ratio to dabrafenib plus trametinib (D + T) or carboplatin plus vincristine (C + V).
BRAF mutation status was identified prospectively via a local assessment or a central laboratory test. In addition, retrospective testing of available tumor samples by the central laboratory was performed to evaluate BRAF V600E mutation status.
Patients received age- and weight-based dosing of MEKINIST and dabrafenib until loss of clinical benefit or until unacceptable toxicity. Carboplatin and vincristine were dosed based on body surface area at doses 175 mg/m2 and 1.5 mg/m2 (0.05 mg/kg for patients < 12 kg), respectively, as one 10-week induction course followed by eight 6-week cycles of maintenance therapy.
The major efficacy outcome measure was overall response rate (ORR) by independent review based on RANO LGG (2017) criteria. Additional efficacy outcome measures were progression-free survival and overall survival. The primary analysis was performed when all patients had completed at least 32 weeks of therapy.
In the LGG cohort, 110 patients were randomized to D + T (n = 73) or C + V (n = 37). Median age was 9.5 years (range: 1 to 17); 60% were female. Study G2201 showed a statistically significant improvement in ORR and PFS in patients with LGG randomized to D + T compared to those randomized to C + V. Efficacy results are shown in Table 26.
Table 26. Efficacy Results Based on Independent Review in Study G2201 (LGG cohort) Abbreviations: CI, confidence interval; NE, not estimable.
aBased on Clopper-Pearson exact confidence interval
bBased on Kaplan-Meier method
cBased on proportional hazards modelMEKINIST plus Dabrafenib
N = 73Carboplatin plus Vincristine
N = 37Overall Response Rate ORR% (95% CI)a 46.6 (34.8, 58.6) 10.8 (3.0, 25.4) P-value < 0.001 Complete response, n (%) 2 (2.7) 1 (2.7) Partial response, n (%) 32 (44) 3 (8) Duration of Response Median (95% CI)b, months 23.7 (14.5, NE) NE (6.6, NE) % with observed DoR ≥ 12 months 56 50 % with observed DoR ≥ 24 months 15 25 Progression-Free Survival Median (95% CI)b, months 20.1 (12.8, NE) 7.4 (3.6, 11.8) Hazard ratio (95% CI)c 0.31 (0.17, 0.55) P-value < 0.001 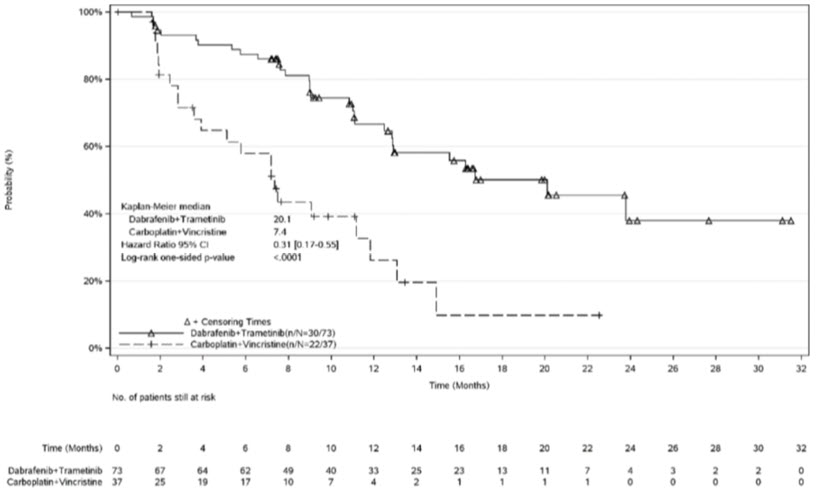
Figure 4. Kaplan-Meier Curves for Progression-Free Survival in Study G2201 (LGG cohort)
At the time of the interim analysis of overall survival (OS), conducted when all patients had completed at least 32 weeks of treatment or had discontinued earlier, there was one death on the C + V arm. The OS results at interim analysis did not reach statistical significance.
-
16
HOW SUPPLIED/STORAGE AND HANDLING
MEKINIST Tablets:
0.5 mg tablets: Yellow, ovaloid, biconvex, unscored film-coated tablets with beveled edges and with the Novartis logo debossed on one side and ‘TT’ on the other side; available in bottles of 30 (NDC 0078-1105-15).
2 mg tablets: Pink, round, biconvex, unscored film-coated tablets with beveled edges and with the Novartis logo debossed on one side and ‘LL’ on the other side; available in bottles of 30 (NDC 0078-1112-15).
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Dispense in original bottle. Do not remove desiccant. Protect from moisture and light. Do not place medication in pill boxes.
MEKINIST for Oral Solution:
White or almost white powder in amber glass bottles, co-packaged with a press-in bottle adapter and an oral syringe. Each bottle contains 4.7 mg of trametinib equivalent to 5.3 mg trametinib dimethyl sulfoxide. Each mL of reconstituted strawberry flavored trametinib solution contains 0.05 mg of trametinib non-solvated parent. (NDC 0078-1161-47).
Store refrigerated at 2°C to 8°C (36°F to 46°F). Store in the original carton to protect from light and moisture.
After reconstitution, store in the original bottle below 25°C (77°F) and do not freeze.
Discard any unused solution 35 days after reconstitution.
-
17
PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
New Primary Cutaneous and Non-cutaneous Malignancies
Advise patients that MEKINIST administered with dabrafenib can result in the development of new primary cutaneous and non-cutaneous malignancies. Advise patients to contact their healthcare provider immediately for any new lesions, changes to existing lesions on their skin, or other signs and symptoms of malignancies [see Warnings and Precautions (5.1)].
Hemorrhage
Advise patients that MEKINIST administered with dabrafenib increases the risk of intracranial and gastrointestinal hemorrhage. Advise patients to contact their healthcare provider to seek immediate medical attention for signs or symptoms of unusual bleeding or hemorrhage [see Warnings and Precautions (5.2)].
Colitis and Gastrointestinal Perforation
Advise patients that MEKINIST can cause colitis and gastrointestinal perforation and to contact their healthcare provider for signs or symptoms of colitis or gastrointestinal perforation [see Warnings and Precautions (5.3)].
Venous Thromboembolic Events
Advise patients that MEKINIST administered with dabrafenib increases the risks of PE and DVT. Advise patients to seek immediate medical attention for sudden onset of difficulty breathing, leg pain, or swelling [see Warnings and Precautions (5.4)].
Cardiomyopathy
Advise patients that MEKINIST can cause cardiomyopathy and to immediately report any signs or symptoms of heart failure to their healthcare provider [see Warnings and Precautions (5.5)].
Ocular Toxicities
Advise patients that MEKINIST can cause severe visual disturbances that can lead to blindness and to contact their healthcare provider if they experience any changes in their vision [see Warnings and Precautions (5.6)].
Interstitial Lung Disease/Pneumonitis
Advise patients that MEKINIST can cause ILD (or pneumonitis). Advise patients to contact their healthcare provider as soon as possible if they experience signs, such as cough or dyspnea [see Warnings and Precautions (5.7)].
Serious Febrile Reactions
Advise patients that MEKINIST administered with dabrafenib can cause serious febrile reactions. Instruct patients to contact their healthcare provider if they develop a fever while taking MEKINIST with dabrafenib [see Warnings and Precautions (5.8)].
Serious Skin Toxicities
Advise patients that MEKINIST can cause serious skin toxicities, which may require hospitalization, and to contact their healthcare provider for progressive or intolerable rash. Advise patients to contact their healthcare provider immediately if they develop signs and symptoms of a severe skin reaction [see Warnings and Precautions (5.9)].
Hypertension
Advise patients that MEKINIST can cause hypertension and that they need to undergo blood pressure monitoring and to contact their healthcare provider if they develop symptoms of hypertension, such as severe headache, blurry vision, or dizziness [see Adverse Reactions (6.1)].
Diarrhea
Advise patients that MEKINIST often causes diarrhea which may be severe in some cases. Inform patients of the need to contact their healthcare provider if severe diarrhea occurs during treatment [see Adverse Reactions (6.1)].
Embryo-Fetal Toxicity
- Advise pregnant women and males of reproductive potential of the potential risk to a fetus [see Warnings and Precautions (5.13), Use in Specific Populations (8.1, 8.3)].
- Advise females to contact their healthcare provider of a known or suspected pregnancy.
- Advise females of reproductive potential to use effective contraception during treatment with MEKINIST and for 4 months after the last dose.
- Advise male patients with female partners of reproductive potential to use condoms during treatment with MEKINIST and for 4 months after the last dose.
Lactation
Advise women not to breastfeed during treatment with MEKINIST and for 4 months after the last dose [see Use in Specific Populations (8.2)].
Infertility
Advise females of reproductive potential of the potential risk for impaired fertility [see Use in Specific Populations (8.3)].
Administration
- Instruct patients to take MEKINIST tablets on an empty stomach (at least 1 hour before or 2 hours after a meal) [see Dosage and Administration (2.3)].
- When administering MEKINIST for oral solution as a single agent, instruct patients to take the oral solution with a low-fat meal or on an empty stomach [see Dosage and Administration (2.3)].
- When coadministering with dabrafenib, instruct patients to take the MEKINIST oral solution on an empty stomach (at least 1 hour before or 2 hours after a meal). Breastfeeding and/or baby formula may be given on demand if a pediatric patient is unable to tolerate the fasting conditions [see Dosage and Administration (2.3)].
THxID® BRAF assay is a trademark of bioMérieux.
Oncomine™ Dx Target Test is a trademark of Life Technologies Corporation, a part of Thermo Fisher Scientific Inc.
Distributed by:
Novartis Pharmaceuticals Corporation
East Hanover, New Jersey 07936© Novartis
T2025-25
-
PATIENT PACKAGE INSERT
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: March 2025 Patient Information MEKINIST® (MEK-in-ist)
(trametinib)
tabletsMEKINIST® (MEK-in-ist)
(trametinib)
for oral solutionImportant information: If your healthcare provider prescribes MEKINIST to be taken or given with dabrafenib, also read the Medication Guide that comes with dabrafenib. What is the most important information I should know about MEKINIST?
MEKINIST may cause serious side effects, including:
Risk of new skin cancers. MEKINIST, when used with dabrafenib, may cause skin cancers, called cutaneous squamous cell carcinoma, keratoacanthoma, basal cell carcinoma, or melanoma.
Talk to your healthcare provider about your risk for these cancers.
Check your skin and tell your healthcare provider right away about any skin changes, including a:- new wart
- skin sore or reddish bump that bleeds or does not heal
- change in size or color of a mole
Your healthcare provider should also check for cancers that may not occur on the skin. Tell your healthcare provider about any new symptoms that develop during treatment with MEKINIST with dabrafenib.
See "What are the possible side effects of MEKINIST?" for more information about side effects.What is MEKINIST?
MEKINIST is a prescription medicine used:
- alone or in combination with a medicine called dabrafenib to treat a type of skin cancer called melanoma:
○ that has spread to other parts of the body or cannot be removed by surgery, and
○ that has a certain type of abnormal “BRAF” gene.
- in combination with dabrafenib, to help prevent melanoma that has a certain type of abnormal “BRAF” gene from coming back after the cancer has been removed by surgery.
- in combination with dabrafenib to treat a type of lung cancer called non-small cell lung cancer (NSCLC):
○ that has spread to other parts of the body, and
○ that has a certain type of abnormal “BRAF” gene.
- in combination with dabrafenib to treat a type of thyroid cancer called anaplastic thyroid cancer (ATC):
○ that has spread to other parts of the body and you have no satisfactory treatment options, and
○ that has a certain type of abnormal “BRAF” gene.
- in combination with dabrafenib to treat solid tumors in adults and children 1 year and older:
○ that cannot be removed by surgery or have spread to other parts of the body, and that have gotten worse (progressed), and you have no satisfactory treatment options, and
○ that have a certain type of abnormal “BRAF” gene.
- in combination with dabrafenib to treat a type of brain tumor called glioma in children 1 year and older:
○ that is low-grade glioma (LGG), and
○ that have a certain type of abnormal “BRAF” gene, and
○ who require a medicine by mouth or injection (systemic therapy).
Your healthcare provider will perform a test to make sure that MEKINIST is right for you.
It is not known if MEKINIST used in combination with dabrafenib is safe and effective in children less than 1 year of age.
It is not known if MEKINIST used alone is safe and effective in children.Before you take or give MEKINIST, tell your healthcare provider about all of your medical conditions, including if you:
- have had bleeding problems or blood clots
- have stomach problems
- have inflammation of the colon
- have heart problems
- have eye problems
- have lung or breathing problems
- have high blood pressure (hypertension)
- have liver or kidney problems
- have diabetes
- are pregnant or plan to become pregnant. MEKINIST can harm your unborn baby.
Females who are able to become pregnant:
○ Your healthcare provider will do a test to see if you are pregnant before starting treatment with MEKINIST.
○ You should use effective birth control (contraception) during treatment with MEKINIST and for 4 months after your last dose of MEKINIST.
○ Talk to your healthcare provider about birth control methods that may be right for you during this time.
○ Tell your healthcare provider right away if you become pregnant or think you might be pregnant during treatment with MEKINIST.
Males (including those who have had a vasectomy) with a female partner who is able to become pregnant:
○ Use condoms during sexual intercourse during treatment with MEKINIST and for at least 4 months after your last dose of MEKINIST. - are breastfeeding or plan to breastfeed. It is not known if MEKINIST passes into your breast milk.
○ Do not breastfeed during treatment and for 4 months after your last dose of MEKINIST. Talk to your healthcare provider about the best way to feed your baby during this time.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.How should I take or give MEKINIST? - Take or give MEKINIST exactly as your healthcare provider tells you to take or give it. Do not change your dose or stop MEKINIST unless your healthcare provider tells you.
- Your healthcare provider may change your dose of MEKINIST, temporarily stop, or completely stop your treatment with MEKINIST if you develop certain side effects.
- If you miss a dose of MEKINIST, take or give it as soon as you remember. Do not take or give a missed dose of MEKINIST if it is less than 12 hours before your next scheduled dose. Just skip the missed dose and take or give the next dose of MEKINIST at the regular time.
- If vomiting happens after taking or giving a dose of MEKINIST, do not take or give an additional dose. Take or give the next dose of MEKINIST at the regular time.
- Take MEKINIST 1 time each day, at the same time each day, about every 24 hours.
- Take MEKINIST on an empty stomach (at least 1 hour before or 2 hours after a meal).
- Take MEKINIST tablets whole. Do not crush or break MEKINIST tablets.
- If MEKINIST tablets are prescribed for your child weighing at least 57 pounds (26 kg), your child’s healthcare provider will adjust their dose as your child grows.
- Tell your healthcare provider if you or your child is not able to swallow MEKINIST tablets whole.
- MEKINIST for oral solution should only be given by a caregiver.
- If MEKINIST for oral solution is prescribed for your child, your child’s healthcare provider will adjust their dose as your child grows.
- See the “Instructions for Use” that comes with the medicine for instructions on how to correctly give a dose of MEKINIST.
- MEKINIST for oral solution can be given using an oral syringe or feeding tube (4 French gauge or larger).
- Give the MEKINIST oral solution 1 time each day, at the same time each day, about every 24 hours.
- Give the MEKINIST oral solution with a low-fat meal or on an empty stomach when giving MEKINIST alone.
- When giving MEKINIST in combination with dabrafenib, give the MEKINIST oral solution on an empty stomach (at least 1 hour before or 2 hours after a meal). If necessary, breastfeeding or baby formula may be given on demand.
What are the possible side effects of MEKINIST?
MEKINIST may cause serious side effects, including:
-
See “What is the most important information I should know about MEKINIST?”
-
bleeding problems. MEKINIST can cause serious bleeding problems, especially in your brain or stomach, that can lead to death. Call your healthcare provider and get medical help right away if you have any signs of bleeding, including:
○ headaches, dizziness, or feeling weak
○ cough up blood or blood clots
○ vomit blood or your vomit looks like “coffee grounds”
○ red or black stools that look like tar
-
inflammation of the intestines, or tears (perforation) of the stomach or intestines. MEKINIST can cause inflammation of your intestines, or tears in the stomach or intestines that can lead to death. Tell your healthcare provider right away if you have any of the following symptoms:
○ bleeding. See “bleeding problems” above.
○ diarrhea (loose stools) or more bowel movements than usual
○ stomach-area (abdominal) pain or tenderness
○ fever
○ nausea
-
blood clots. MEKINIST can cause blood clots in your arms or legs, which can travel to your lungs and can lead to death. Get medical help right away if you have the following symptoms:
○ chest pain
○ sudden shortness of breath or trouble breathing
○ pain in your legs with or without swelling
○ swelling in your arms or legs
○ a cool, pale arm or leg -
heart problems, including heart failure. Your healthcare provider should check your heart function before and during treatment with MEKINIST. Call your healthcare provider right away if you have any of the following signs and symptoms of a heart problem:
○ feeling like your heart is pounding, racing, or beating irregularly
○ shortness of breath
○ swelling of your ankles and feet
○ feeling lightheaded
-
eye problems. MEKINIST can cause severe eye problems that might lead to blindness. Call your healthcare provider right away if you get these symptoms of eye problems:
○ blurred vision, loss of vision, or other vision changes
○ see color dots
○ halo (seeing blurred outline around objects)
○ eye pain, swelling, or redness
-
lung or breathing problems. MEKINIST can cause lung or breathing problems. Tell your healthcare provider if you have any new or worsening symptoms of lung or breathing problems, including:
○ shortness of breath
○ cough
-
fever. Fever is common during treatment with MEKINIST and dabrafenib, but it may also be serious. When taking MEKINIST with dabrafenib, fever may happen more often or may be more severe. In some cases, chills or shaking chills, too much fluid loss (dehydration), low blood pressure, dizziness, or kidney problems may happen with the fever.
Call your healthcare provider right away if you get a fever during treatment with MEKINIST.
Your healthcare provider may temporarily or permanently stop your treatment or change your dose of MEKINIST with dabrafenib if you have fevers. Your healthcare provider will treat you as needed for your fever and any signs and symptoms of infection and should check your kidney function during and after you have had severe fever. -
serious skin reactions. Skin rash is a common side effect of MEKINIST. MEKINIST can also cause other skin reactions. In some cases, these rashes and other skin reactions can be severe or serious and may need to be treated in a hospital or lead to death.
Tell your healthcare provider if you get a skin rash or acne that bothers you or worsens.
Tell your healthcare provider right away if you develop any new or worsening symptoms of a severe skin reaction, including:
○ blisters or peeling of your skin
○ mouth sores○ blisters on your lips, or around your mouth or eyes
○ high fever or flu-like symptoms
○ enlarged lymph nodes-
increased blood sugar (hyperglycemia). Some people may develop high blood sugar or worsening diabetes during treatment with MEKINIST and dabrafenib. If you are diabetic, your healthcare provider should check your blood sugar levels closely during treatment with MEKINIST and dabrafenib. Your diabetes medicine may need to be changed. Tell your healthcare provider if you have any of the following symptoms of severe high blood sugar:
○ increased thirst
○ urinating more often than normal or urinating an increased amount of urine
-
hemophagocytic lymphohistiocytosis (HLH). MEKINIST when taken or given with dabrafenib may increase the risk of a type of overactivity of the immune system (hemophagocytic lymphohistiocytosis) that can cause fever, swollen glands, bruising, or skin rash. If you experience a combination of these symptoms, call your healthcare provider right away.
- rash
- diarrhea. Call your healthcare provider if you get severe diarrhea.
- swelling of the face, arms, or legs
The most common side effects of MEKINIST when taken with dabrafenib in people with melanoma that has spread to other parts of the body or cannot be removed by surgery include: • fever • diarrhea • nausea • vomiting • rash • high blood pressure (hypertension) • chills • swelling of the face, arms, or legs The most common side effects of MEKINIST when taken with dabrafenib to help prevent melanoma from coming back after the cancer has been removed by surgery include: • fever • chills • tiredness • diarrhea • nausea • vomiting • headache • joint aches • rash • muscle aches The most common side effects of MEKINIST when taken with dabrafenib in people with NSCLC include: • fever • rash • tiredness • swelling of your face, arms, and legs • nausea • chills • vomiting • bleeding • diarrhea • cough • dry skin • shortness of breath • decreased appetite The most common side effects of MEKINIST when taken with dabrafenib in adults with solid tumors that cannot be removed by surgery or have spread to other parts of the body include: • fever • bleeding • tiredness • cough • nausea • vomiting • rash • constipation • chills • diarrhea • headache • muscle and joint aches • swelling of your arms and legs The most common side effects of MEKINIST when given with dabrafenib in children 1 year of age and older with solid tumors that cannot be removed by surgery or have spread to other parts of the body include: • fever • acne • rash • headache • vomiting • stomach-area (abdominal) pain • tiredness • nausea • dry skin • bleeding • cough • constipation • diarrhea • skin infection around fingernails or toenails The most common side effects of MEKINIST when given with dabrafenib in children 1 year of age and older with low-grade glioma include: • fever • diarrhea • rash • dry skin • headache • nausea • vomiting • bleeding • muscle and bone pain • stomach-area (abdominal) pain • tiredness • acne MEKINIST can cause new or worsening high blood pressure (hypertension). Your healthcare provider should check your blood pressure during treatment with MEKINIST. Call your healthcare provider right away if you develop high blood pressure, your blood pressure worsens, or you have severe headache, lightheadedness, blurry vision, or dizziness.
MEKINIST may cause fertility problems in females. This could affect your ability to become pregnant. Talk to your healthcare provider if this is a concern for you.
These are not all the possible side effects of MEKINIST.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Novartis Pharmaceuticals Corporation at 1-888-669-6682.How should I store MEKINIST?
MEKINIST tablets:- Store MEKINIST tablets at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep MEKINIST tablets dry and away from moisture and light.
- The bottle of MEKINIST tablets contains a drying agent (desiccant packet) to help keep your medicine dry. Do not throw away (dispose of) the desiccant packet.
- Keep MEKINIST tablets in the original bottle. Do not place tablets in a pill box.
- Safely throw away MEKINIST tablets that are out of date or no longer needed.
- Store MEKINIST for oral solution in the original amber bottle at room temperature below 77°F (25°C). Do not freeze.
- Keep MEKINIST for oral solution in the carton it comes in and away from direct moisture and light.
- Throw away unused MEKINIST for oral solution after the expiration or “discard after” date written on the amber bottle label.
General information about the safe and effective use of MEKINIST
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use MEKINIST for a condition for which it was not prescribed. Do not give MEKINIST to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about MEKINIST that is written for health professionals.What are the ingredients in MEKINIST?
MEKINIST tablets:
Active ingredient: trametinib
Inactive ingredients:
Tablet Core: colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate (vegetable source), mannitol, microcrystalline cellulose, sodium lauryl sulfate.
Tablet Coating: hypromellose, iron oxide red (2 mg tablets), iron oxide yellow (0.5 mg tablets), polyethylene glycol, polysorbate 80 (2 mg tablets), titanium dioxide.
MEKINIST for oral solution:
Active ingredient: trametinib
Inactive ingredients: betadex sulfobutyl ether sodium, citric acid monohydrate, dibasic sodium phosphate, methylparaben, potassium sorbate, sucralose, and strawberry flavor.
Distributed by: Novartis Pharmaceuticals Corporation, East Hanover, New Jersey 07936
© Novartis
For more information, go to www.us.tafinlarmekinist.com or call 1-888-669-6682.
T2025-21
- new wart
-
INSTRUCTIONS FOR USE
This Instructions for Use has been approved by the U.S. Food and Drug Administration. Revised: March 2025 INSTRUCTIONS FOR USE
MEKINIST® (MEK-in-ist)
(trametinib)
for oral solutionThis “Instructions for Use” contains information on how to give MEKINIST oral solution. Important information you need to know before giving MEKINIST oral solution - MEKINIST oral solution should only be given by a caregiver.
- Read this “Instructions for Use” carefully before you give MEKINIST oral solution for the first time and each time you get a refill. There may be new information.
- This “Instructions for Use” does not take the place of talking with your healthcare provider about your or your child’s medical condition and treatment.
- You will receive the MEKINIST oral solution in an amber-colored bottle that your pharmacist has already mixed.
- If you receive MEKINIST as a powder, contact your healthcare provider or pharmacist right away. Do not mix the MEKINIST powder on your own.
- Your pharmacist prepared the MEKINIST oral solution to an exact concentration. Do not add more water to the oral solution you receive from the pharmacy.
- Your healthcare provider or pharmacist should show the caregiver how to measure and give a dose of MEKINIST oral solution correctly. Always give MEKINIST oral solution exactly as your healthcare provider tells you to.
- If you have any questions about how to measure and give a dose of MEKINIST oral solution, talk to your healthcare provider or pharmacist.
- If at any time MEKINIST oral solution gets on your or your child’s skin, wash the area well with soap and water.
- If at any time MEKINIST oral solution gets in your or your child’s eyes, rinse the eyes well with cool water.
- Ask your healthcare provider or pharmacist about how to safely throw away (dispose of) MEKINIST oral solution.
- When MEKINIST is prepared as an oral solution, it can be used for 35 days. Throw away any remaining oral solution after 35 days.
- If you spill any MEKINIST oral solution, follow the instructions at the end of this "Instructions for Use" in the section called “How to clean up any spilled MEKINIST oral solution.”
The MEKINIST pack should contain: 
Reusable oral syringe parts: 
Storage of MEKINIST oral solution - Store the amber bottle of MEKINIST oral solution at room temperature below 77°F (25°C).
- Do not freeze MEKINIST oral solution.
- Store the amber bottle of MEKINIST oral solution upright with the cap tightly closed.
- Keep MEKINIST oral solution in the carton it comes in and away from direct moisture and light.
- When MEKINIST is prepared as an oral solution, it can be used for 35 days. Throw away any remaining oral solution after 35 days.
- Do not use MEKINIST oral solution after the expiration or “discard after” date written or printed on the label by the pharmacist.
- Keep MEKINIST oral solution and all medicines out of the reach of children.

Storage of oral syringe - Store the oral syringe with the MEKINIST oral solution.
- Keep oral syringe out of the reach of children.

Section A. Measuring and giving a dose of MEKINIST oral solution - Gather your supplies:
- 1 bottle adapter (already inserted into the bottle neck)
- 1 amber bottle containing MEKINIST oral solution
- 1 reusable oral syringe
- If any of the MEKINIST oral solution comes into contact with your skin or eyes when you are following the steps below, follow the instructions in the section “Important information you need to know before giving MEKINIST oral solution” above.
- If any MEKINIST oral solution spills, follow the instructions in the section “How to clean up any spilled MEKINIST oral solution” at the end of this "Instructions for Use."
Step 1. Wash and dry your hands before measuring and giving a dose of MEKINIST oral solution. Step 2. Place your supplies on a clean, flat work surface. Step 3. Check if you have powder or solution in the amber bottle. - If you have powder, do not give MEKINIST and contact your healthcare provider or pharmacist. Do not mix the MEKINIST powder on your own.
- If you have solution, continue to Step 4 below.
- Your pharmacist prepared the MEKINIST oral solution to an exact concentration. Do not add more water to the oral solution you receive from the pharmacy.
Step 4. Check the expiration or “discard after” date of the MEKINIST oral solution that is handwritten or printed on the amber bottle label. - Do not give MEKINIST oral solution if the expiration or “discard after” date has passed or there is no date on the amber bottle label.
- Note: If you are unsure of the expiration or “discard after” date, contact your healthcare provider or pharmacist.
Step 5. Gently swirl the amber bottle for 30 seconds to mix the MEKINIST oral solution. - If foam appears, put the amber bottle on a flat surface and let it sit there until the foam disappears.
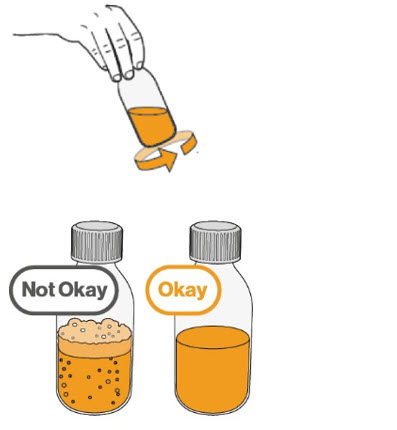
Step 6. When the foam has disappeared inside the amber bottle, remove the child-resistant cap by pushing down on the cap and turning it in the direction of the arrow (counter-clockwise), as shown. Then place the amber bottle back on your flat work surface. 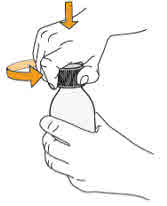
Step 7. Check if there is a bottle adapter already inserted in the bottle neck. Do not remove the bottle adapter. If not inserted, separate the bottle adapter from the oral syringe and insert it.
Contact your healthcare provider if you are unsure or the bottle adapter is missing.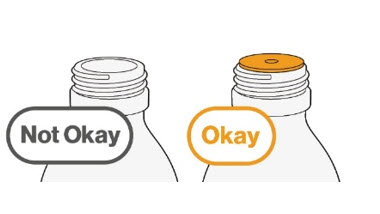
Step 8. Next, pick up the oral syringe.
Push the plunger up into the oral syringe as far as it will go to remove all the air inside.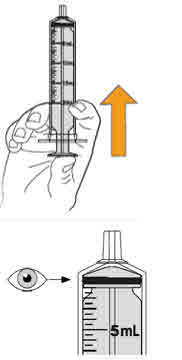
Step 9. Use one hand to hold the amber bottle containing the prepared MEKINIST oral solution steady.
With your other hand, insert the tip of the oral syringe into the opening of the bottle adapter.
Make sure the oral syringe is securely attached.
Important: Due to air pressure, the plunger may move by itself when you measure your dose during Step 10.
Hold the end of the plunger to prevent it from moving.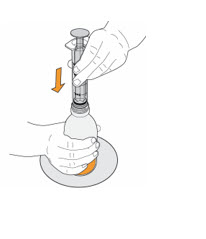
Step 10. Carefully turn the amber bottle upside down and pull down on the plunger to draw the MEKINIST oral solution into the oral syringe.
To measure your dose, keep the tip of the oral syringe facing up. Pull down on the plunger until the top of the black stopper lines up with your prescribed dose in mLs on the oral syringe barrel.
If large air bubbles appear in the oral syringe, push the MEKINIST oral solution back into the amber bottle and then pull down on the plunger again to draw up your dose.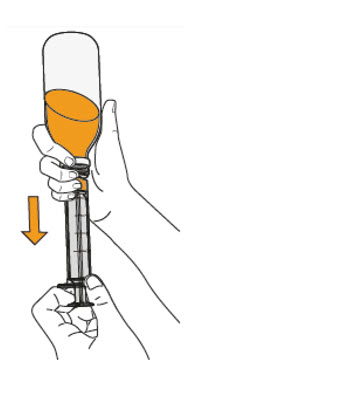
Keep doing this until there are no large air bubbles present. Small air bubbles are okay. 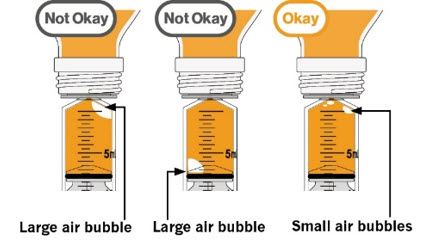
Step 11. Continue to hold the plunger in place, and carefully turn the amber bottle upright. Put the amber bottle onto your flat work surface again.
While still holding the plunger and barrel, remove the oral syringe from the bottle by gently pulling straight up.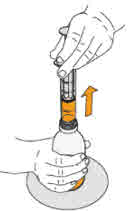
Check again to be sure that the top of the black stopper is at your prescribed dose. If not, repeat Steps 9, 10 and 11 again.
Note: Your dose may be different than the dose shown in this figure.
If you are giving a dose of MEKINIST oral solution by mouth, move to Step 12.
If you are giving a dose of MEKINIST oral solution through a feeding tube, go to Section B.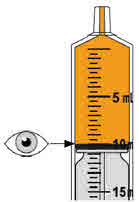
Step 12. Important: If giving a dose of MEKINIST oral solution to a child, make sure they are sitting upright. Place the tip of the oral syringe inside the mouth. The tip should touch the inside of either cheek.
Slowly push the plunger all the way down to give the full dose of MEKINIST oral solution.
Warning: Giving MEKINIST oral solution directly into the throat or pushing down on the plunger too fast may cause choking.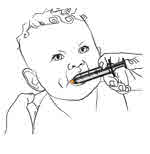
Step 13. Check to make sure that there is no MEKINIST oral solution left in the oral syringe.
If there is any MEKINIST oral solution left in the oral syringe, give it.
Note: If your dose in mLs is more than the oral syringe can hold, repeat Steps 8 through 14 until the total prescribed dose is given.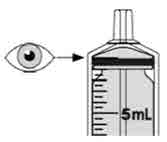
Step 14. When finished, close the amber bottle.
Do not remove the bottle adapter.
Place the cap back on the amber bottle and turn it in the direction of the arrow (clockwise), as shown, to close it.
Make sure the cap is securely attached onto the amber bottle.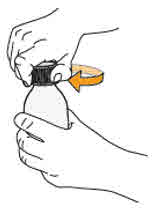
Step 15. Clean the reusable oral syringe. See the instructions in Section C “Cleaning the reusable oral syringe.” 
Section B. Giving a dose of MEKINIST oral solution through a feeding tube This section is for use only if you are going to give a dose of MEKINIST oral solution through a feeding tube.
Before giving a dose of MEKINIST oral solution through a feeding tube, carefully read the following information, then move to Step 1.
- MEKINIST oral solution may be given through a feeding tube, as directed by your healthcare provider.
- Only use a nasogastric (NG) or gastric (G-tube) feeding tube with a size of 4 French gauge or larger.
- Always use the 20 mL oral syringe provided in this pack to give a dose of MEKINIST oral solution.
- You may need an ENFIT adapter (not included in this pack) to connect the 20 mL oral syringe to the feeding tube.
Step 1. Flush the feeding tube according to the manufacturer’s instructions right away before giving a dose of MEKINIST oral solution. Step 2. Follow Steps 1 through 11 in Section A, then move to Step 3 in this section. Step 3. Connect the 20 mL oral syringe containing MEKINIST oral solution to the feeding tube. You may need an ENFIT adapter to connect the oral syringe to the feeding tube. Step 4. Apply steady pressure to the plunger to give the dose of MEKINIST oral solution through the feeding tube. Step 5. Check to be sure that there is no MEKINIST oral solution left in the oral syringe.
If there is any MEKINIST oral solution left in the oral syringe, repeat Steps 3 through 5.Step 6. Flush the feeding tube again according to the manufacturer’s instructions. Step 7. Go to Section C for instructions on “Cleaning the reusable oral syringe.” Section C. Cleaning the reusable oral syringe Note: Clean the oral syringe separate from other kitchen items. Step 1. Fill a glass with warm, soapy water. 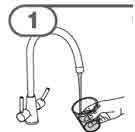
Step 2. Place the tip of the oral syringe into the glass of warm, soapy water.
Pull up and then push down on the plunger to pull the soapy water in and out of the oral syringe 4 to 5 times.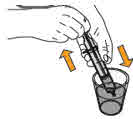
Step 3. Remove the plunger from the barrel. 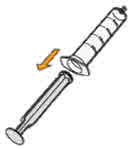
Step 4. Rinse the glass, plunger, and barrel under warm tap water. 
Step 5. Leave the oral syringe plunger and barrel on a clean paper towel to air dry. When your oral syringe is dry, store it with the amber bottle of MEKINIST oral solution.
Always keep the oral syringe out of the reach of children.
Note: Use a new oral syringe for each new amber bottle of MEKINIST oral solution.
How to throw away MEKINIST oral solution that is expired or no longer needed, or old oral syringes - Throw away unused MEKINIST oral solution into the trash. Do not pour MEKINIST oral solution down the drain.
- Ask your healthcare provider or pharmacist about how to safely throw away MEKINIST oral solution if you are not sure.
- Use a new oral syringe for each new amber bottle of MEKINIST oral solution. Throw away the old oral syringe into the trash.
- Ask your healthcare provider or pharmacist how to safely throw away the oral syringe if you are not sure.
How to clean up any spilled MEKINIST oral solution If you accidentally spill any MEKINIST oral solution, clean up the spill as follows:
1. Put on plastic gloves.
2. Soak up the spilled MEKINIST oral solution completely using an absorbent material, such as paper towels.
3. Place the used absorbent material into a sealable plastic bag, such as a food storage bag.
4. Wipe all surfaces exposed to MEKINIST oral solution with alcohol wipes or pour rubbing alcohol onto a paper towel and then wipe the exposed surfaces with the paper towel.
5. Place the bag, gloves and used alcohol wipes or paper towel into another second plastic bag and seal.
6. Throw away the bags into the trash.
7. Wash your hands well with soap and water.Distributed by:
Novartis Pharmaceuticals Corporation
East Hanover, New Jersey 07936
© NovartisT2025-13
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
PRINCIPAL DISPLAY PANEL
NDC 0078-1161-47
Rx only
Mekinist®
(trametinib)
for Oral Solution0.05 mg per mL*
90 mL (when reconstituted)
To Pharmacist: Reconstitute before
dispensing*. Dispense with the enclosed
Patient Information and Instructions
for Use.For administration by caregivers only.
Discard any unused solution 35 days after reconstitution.
NOVARTIS

-
INGREDIENTS AND APPEARANCE
MEKINIST
trametinib tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0078-1105 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRAMETINIB DIMETHYL SULFOXIDE (UNII: BSB9VJ5TUT) (TRAMETINIB - UNII:33E86K87QN) TRAMETINIB .5 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM LAURYL SULFATE (UNII: 368GB5141J) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color YELLOW Score no score Shape OVAL (biconvex, with beveled edges) Size 9mm Flavor Imprint Code IMPRINT;TT Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0078-1105-15 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/22/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204114 03/17/2016 MEKINIST
trametinib tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0078-1112 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRAMETINIB DIMETHYL SULFOXIDE (UNII: BSB9VJ5TUT) (TRAMETINIB - UNII:33E86K87QN) TRAMETINIB 2 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) MANNITOL (UNII: 3OWL53L36A) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM LAURYL SULFATE (UNII: 368GB5141J) FERRIC OXIDE RED (UNII: 1K09F3G675) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color PINK Score no score Shape ROUND (biconvex, with beveled edges) Size 8mm Flavor Imprint Code IMPRINT;LL Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0078-1112-15 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/22/2021 2 NDC:0078-1112-94 14 in 1 BOTTLE; Type 0: Not a Combination Product 12/22/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA204114 03/17/2016 MEKINIST
trametinib powder, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0078-1161 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRAMETINIB DIMETHYL SULFOXIDE (UNII: BSB9VJ5TUT) (TRAMETINIB - UNII:33E86K87QN) TRAMETINIB 0.05 mg in 1 mL Inactive Ingredients Ingredient Name Strength BETADEX SULFOBUTYL ETHER SODIUM (UNII: 2PP9364507) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM PHOSPHATE, DIBASIC, UNSPECIFIED FORM (UNII: GR686LBA74) METHYLPARABEN (UNII: A2I8C7HI9T) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SUCRALOSE (UNII: 96K6UQ3ZD4) STRAWBERRY (UNII: 4J2TY8Y81V) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0078-1161-47 90 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/16/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA217513 03/16/2023 Labeler - Novartis Pharmaceuticals Corporation (002147023)





